Abstract
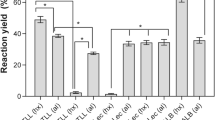
Lipase from Brevibacillus agri 52 was found stable up to 90% diethylenglycol (DEG), glycerol (GLY), and 1,2 propanediol (1,2 PRO) at 37 °C for 1 h and the stability was reduced only approximately 20% after 12 h incubation, but in 40% dimethylsulfoxide (DMSO), lipase activity was stable only for 1 h. Inhibition of the biocatalysts with dimethylformamide (DMF) was detected at 20% solvent concentration. In water immiscible systems, the stability of lipase in n-hexane, n-tetradecane and n-heptane resembles the water activity, but in the presence of isobutanol, 1-hexanol, and butylbutirate, the stability was significantly reduced. Lipase 52 precipitates in the presence of 50% acetone or ethanol/water mixtures, but enzymatic activity was partially recovered by adding 20% GLY, DEG, 1,2 PRO, or DMSO to the reaction mixture. Furthermore, by increasing DEG in 70% DMF/DEG mixtures, the lipase activity was protected. Encapsulation of lipase in pectin gels cross-linked with calcium ions brings three to four times more enzymatic activity in 70% water miscible organic solvents compared to aqueous systems.







Similar content being viewed by others
References
Vulfson, E. N., Halling, P. J., & Holland, H. L. (2001). Enzymes in non-aqueous solvents. Totowa, NJ: Humana.
Jaeger, K. E., & Reetz, M. T. (1998). Tibtech, 16, 396–403.
Pandey, A., Benjamin, S., Soccol, C. R., Nigam, P., Krieger, N., & Soccol, V. T. (1999). Biotechnology and Applied Biochemistry, 29, 119–131.
Haba, E., Breco, O., Ferrer, C., Marqués, A., Busquets, M., & Manresa, A. (2000). Biotechnology and Applied Biochemistry, 26, 40–44.
Torres, S., & Castro, G. R. (2004). Food Technology and Biotechnology, 42, 271–277.
Hotta, Y., Ezaki, S., Atomi, H., & Imanaka, T. (2002). Applied and Environmental Microbiologys, 68, 3925–393.
Carrea, G., & Riva, S. (2000). Angewandte Chemie. International Edition in English, 39, 2226–2254.
Reetz, M. T. (1997). Advanced Materials, 9, 943–954.
Van Buren, J. P. (1991). Function of pectin in plant tissue structure and firmness. In H. W. Reginald (Ed.), The chemistry and technology of pectin (pp. 3–4). New York: Academic.
Ryden, P., & Selvendram, R. R. (1990). Carbohydrate Research, 195, 257–272.
Lootens, D., Capel, F., Durand, D., Nicolai, T., Boulenguer, P., & Langendorff, V. (2003). Food Hydrocolloid, 17, 237–244.
Gupta, N., Rathi, P., & Gupta, R. (2002). Analytical Biochemistry, 311, 98–99.
Bradford, M. M. (1976). Analytical Biochemistry, 78, 248–254.
Laane, C., Boeren, S., Vos, K., & Veeger, C. (1987). Biotechnology and Bioengineering, 30, 81–87.
Hudson, E. P., Eppler, R. K., & Clark, D. S. (2005). Current Opinion in Biotechnology, 16, 637–643.
Ferst, A. (1999). Structure and mechanism in protein science. New York: Freeman.
Castro, G. R., & Knubovets, T. (2003). Critical Reviews in Biotechnology, 23, 195–231.
Lima, V. M. G., Krieger, N., Mitchell, D. A., Baratti, J. C., De Felippis, I., & Fontana, J. D. (2004). Journal of Molecular Catalysis. B, Enzymatic, 31, 53–61.
Zhao, L. -L., Xu, J. -H., Zhao, J., Pan, J., & Wang, Z. -L. (2008). Process Biochemistry, 43, 626–633.
Leščic, I., Vukelic, B., Majeric-Elenkov, M., Saenger, W., & Abramic, M. (2001). Enzyme and Microbial Technology, 29, 548–553.
Pahujani, S., Kanwar, S. S., Chauhan, G., & Gupta, R. (2008). Bioresource Technology, 99, 2566–2570.
Tsuzuki, W., Ue, A., & Nagao, A. (2003). Bioscience, Biotechnology, and Biochemistry, 67, 1660–1666.
Acknowledgments
The authors would like to thank M.D. Baigorí for helpful discussions and providing for the strain. The authors gratefully acknowledge the financial support from Pew Charitable Trust (USA), CONICET (PIP 6203/06, Argentina) and ANPCyT (BID no. 1728/ OC-AR, Argentina).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Costas, L., Bosio, V.E., Pandey, A. et al. Effects of Organic Solvents on Immobilized Lipase in Pectin Microspheres. Appl Biochem Biotechnol 151, 578–586 (2008). https://doi.org/10.1007/s12010-008-8233-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-008-8233-0