Abstract
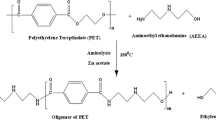
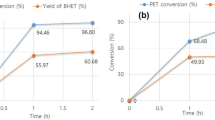
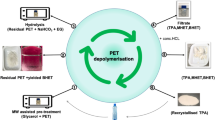
Polyethylene terephthalate (PET) waste recycling has become a worldwide research interest for industries and academic institutes due its inevitable environmental impact. The main objective of current research work is to target efficient recycling of PET waste from mineral water bottles by the glycolysis method and subsequent use of the recycled product for value-added coating application. In the present study, we report on MP-diol (2-methyl-1,3-propanediol) which is not explored much for the chemical recycling of PET, having a branched aliphatic diol with two primary hydroxyls, for glycolysis reaction. The reaction parameters were optimized for microwave-assisted technique by varying the ratio of raw materials, reaction time, temperature, and power. The reaction parameters were optimized, and the recycled oligomeric product (OPETMPD) was separated, purified, and characterized by chemical and spectroscopic methods. Subsequently, dimethacrylated oligoesters of PET oligomer (UV oligomer) were synthesized by methacrylation of the glycolyzed PET product (OPETMPD). The synthesized UV oligomer was evaluated using chemical and spectroscopic methods. Ultraviolet (UV) radiation-curable formulations were prepared using synthesized UV oligomer and applied on wooden panels. The coatings were cured using UV-curing machine and evaluated for their performance properties. The partial replacement of UV oligomer in UV formulations exhibited comparative coating performance properties with respect to conventional UV formulation.










Similar content being viewed by others
References
Nikles, DE, Farahat, MS, “New Motivation for the Depolymerization Products Derived from Poly (Ethylene Terephthalate) (PET) Waste: A Review.” Macromol. Mater. Eng., 290 13–30 (2005)
Paszun, D, Spychaj, T, “Chemical Recycling of Poly(ethylene terephthalate).” Ind. Eng. Chem. Res., 36 (4) 1373–1383 (1997)
Achilias, D, Karayannidis, G, “The Chemical Recycling of PET in the Framework of Sustainable Development.” Water Air Soil Pollut. Focus, 4 385–396 (2004)
Sinha, V, Patel, M, Patel, J, “Pet Waste Management by Chemical Recycling: A Review.” J. Polym. Environ., 18 8–25 (2010)
Al-Sabagh, AM, Yehia, FZ, Eshaq, Gh, Rabie, AM, El Metwally, AE, “Greener Routes for Recycling of Polyethylene Terephthalate.” Egypt. J. Petrol, 25 (1) 53–64 (2016)
Geyer, B, Lorenz, G, Kandelbauer, A, “Recycling of Poly (ethylene terephthalate)—A Review Focusing on Chemical Methods.” eXPRESS Poly Lett., 10 (7) 559–586 (2016)
Radenkov, P, Radenkov, M, Grancharov, G, Troev, K, “Direct Usage of Products of Poly(ethylene terephthalate) Glycolysis for Manufacturing of Glass-Fibre-Reinforced Plastics.” Eur. Polym. J., 39 1223–1228 (2003)
Colomines, G, Robin, J, Tersac, G, “Study of the Glycolysis of PET by Oligoesters.” Polymer, 46 3230–3247 (2005)
Dutt, K, Soni, RK, “A Review on Synthesis of Value Added Products from Polyethylene Terephthalate (PET) Waste.” Polym. Sci. Ser. B, 55 430–452 (2013)
Hoogenboom, R, Schubert, U, “Microwave-Assisted Polymer Synthesis: Recent Developments in a Rapidly Expanding Field of Research.” Macromol. Rapid Commun., 28 368–386 (2007)
Surati, M, Jauhari, S, Desai, KR, “A Brief Review: Microwave Assisted Organic Reaction.” Arch. Appl. Sci. Res., 4 (1) 645–661 (2012)
Jamdar, V, Kathalewar, M, Jagtap, RN, Dubey, KA, Sabnis, A, “Effect of γ-Irradiation on Glycolysis of PET Waste and Preparation of Ecofriendly Coatings Using Bio-Based and Recycled Materials.” Polym. Eng. Sci., 55 (11) 2653–2660 (2015)
Karayannidis, GP, Nikolaidis, AK, Sideridou, ID, Bikiaris, DN, Achilias, DS, “Chemical Recycling of PET by Glycolysis: Polymerization and Characterization of the Dimethacrylated Glycolysate.” Macromol. Mater. Eng., 291 1338–1347 (2006)
Soni, RK, Teotia, M, Dutt, K, “Studies on Synthesis and Characterization of a Novel Acrylic Aromatic Amide Oligomer of Aminolysed End Products Generated from Pet Waste with Hydrazine Monohydrate and Its Photocuring with Acrylate Monomers.” J. Appl. Polym. Sci., 118 638–645 (2010)
ASTM D-1980, 1987 (reapproved 1998), Standard Test Method for Acid Value of Fatty Acids and Polymerized Fatty Acids, United States
ASTM D 1957-86 (Reapproved 2001), Standard Test Method for Hydroxyl Value of Fatty Oils and Acids
ASTM D-1959, 1997, Standard Test Method for Iodine Value of Drying Oils and Fatty Acids, United States
ASTM D 523-67, Standard Test Method for Specular Gloss.
ASTM D-3359-02, Standard Test Methods for Measuring Adhesion by Tape Test.
ASTM D-3363-00, Standard Test Method for Film Hardness by Pencil Test.
ASTM D-4752-03, Standard Test Method for Measuring MEK Resistance of Ethyl Silicate (Inorganic) Zinc Rich Primers by Solvent Rub.
Kathalewar, M, Sabnis, A, Waghoo, G, “Effect of Incorporation of Surface Treated Zinc Oxide on Non-isocyanate Polyurethane Based Nano-composite Coatings.” Prog. Org. Coat., 76 (9) 1215–1229 (2013)
ASTM D 570-98 (2010), Standard Test Method for Water Absorption of Plastics
ASTM D 3023-98, Standard Practice for Determination of Resistance of Factory-Applied Coatings on Wood Products to Stains and Reagents
Pavia, DL, Lampman, GM, Kriz, GS, Introduction to Spectroscopy, 4th ed. Brooks/Cole, Cengage Learning Inc., United States (2009)
Silverstein, RM, Webster, FX, Kiemle, DJ, Spectrometric Identification of Organic Compounds, pp. 127–227. Wiley, NE York (2005)
Acknowledgments
This research work was supported by the Board of Research in Nuclear Sciences (BRNS), India.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jamdar, V., Kathalewar, M. & Sabnis, A. Glycolytic depolymerization of PET waste using MP-diol and utilization of recycled product for UV-curable wood coating. J Coat Technol Res 15, 259–270 (2018). https://doi.org/10.1007/s11998-017-9992-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11998-017-9992-8