Abstract
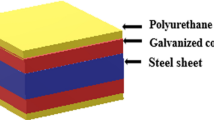
A newly developed, polyurethane-based, marine antifouling coating, containing 2% immobilized Econea, was examined in terms of its anticorrosion performance. The novelty of the experimental formulation arises from the immobilization of the biocide which minimizes leaching and was accomplished via a newly developed functionalization method, based on reaction of the biocide with highly reactive isocyanate functionality. The painting system was applied on steel specimens, then scribed with a sharp cutter and examined for 12 weeks in cyclic salt spray exposure. Identification of the rust morphologies was performed with XRD, Raman spectroscopy, SEM, and EDS methods. The absence of paint deformation during the experiment led to the formation of compact corrosion products, firmly adherent to the substrate, allowing transformation to more protective forms, such as oxides (hematite, maghemite, magnetite) and the least harmful of the oxyhydroxides (goethite, feroxyhyte), found in the mixture, ensuring sufficient corrosion protection. The unscratched part of the paint served as a barrier to corrosion product expansion beyond the scribed areas. An acrylic-based antifouling system was also examined for reasons of comparison. The experimental formulation exhibited superior anticorrosion performance overall, since the acrylic system presented extended material loss, blistering, checking, and extensive substrate rust coverage beneath the multilayer coat, implying unsatisfactory corrosion protection.










Similar content being viewed by others
References
Callow, JA, Callow, ME, “Trends in the Development of Environmentally Friendly Fouling-Resistant Marine Coatings.” Nat. Commun., 2 244 (2011)
Schultz, MP, Swain, GW, “The Influence of Biofilms on Skin Friction Drag.” Biofouling, 15 (1–3) 129–139 (2000)
Abbott, A, Abel, PD, Arnold, DW, Milne, A, “Cost Benefit Analysis of the Use of TBT: The Case for a Treatment Approach.” Sci. Total Environ., 258 (1–2) 5–19 (2000)
Champ, MA, “A Review of Organotin Regulatory Strategies, Pending Actions, Related Costs and Benefits.” Sci. Total Environ., 258 (1–2) 21–71 (2000)
Dürr, S, Thomason, JC, Biofouling. Wiley-Blackwell, UK (2010)
Omae, I, “Organotin Antifouling Paints and their Alternatives.” Appl. Organomet. Chem., 17 81–105 (2003)
Davies, P, Evrard, G, “Accelerated Ageing of Polyurethanes for Marine Applications.” Polym. Degrad. Stabil., 92 1455–1464 (2007)
Quaiser, S, Lynam, I, Wind, M, Celik, A, Kampf, G, “Fouling Resistant Polyurethanes, in Particular for Maritime Use.” US Patent 2008/0090938 A1, 2008
Brady, RF, Aronson, CL, “Elastomeric Fluorinated Polyurethane Coatings for Nontoxic Fouling Control.” Biofouling, 19 59–62 (2003)
Webster, DC, Chisholm, BJ, Stafslien, SJ, “Mini Review: Combinatorial Approaches for the Design of Novel Coating Systems.” Biofouling, 23 179–192 (2007)
Rahman, MM, Chun, HH, “Waterborne Polysiloxane-Urethane-Urea for Potential Marine Coatings.” J. Coat. Technol. Res., 8 (3) 389–399 (2011)
Verborgt, J, Webb, A, “Solvent-Free, Self-Polishing Polyurethane Matrix for Use in Solvent-Free Antifoulings.” US Application Patent, 2007014753, 2007
Bierwagen, G, Tallman, D, Li, J, He, L, Jeffcoate, C, “EIS Studies of Coated Metals in Accelerated Exposure.” Prog. Org. Coat., 46 (2) 149–158 (2003)
Yang, X, Vang, C, Tallman, D, Bierwagen, G, Croll, S, Rohlik, S, “Weathering Degradation of a Polyurethane Coating.” Polym. Degrad. Stabil., 74 (2) 341–351 (2001)
Yang, XF, Tallman, D, Bierwagen, G, Croll, S, Rohlik, S, “Blistering and Degradation of Polyurethane Coatings under Different Accelerated Weathering Tests.” Polym. Degrad. Stabil., 77 (1) 103–109 (2002)
Kumar, SA, Balakrishnan, T, Alagar, M, Denchev, Z, “Development and Characterization of Silicone/Phosphorus Modified Epoxy Materials and Their Application as Anticorrosion and Antifouling Coatings.” Prog. Org. Coat., 55 (3) 207–217 (2006)
Bierwagen, GP, “Reflections on Corrosion Control by Organic Coatings.” Prog. Org. Coat., 28 (1) 43–48 (1996)
Skerry, B, Alavi, A, Lindgren, K, “Environmental and Electrochemical Test Methods for the Evaluation of Protective Organic Coatings.” J. Coat. Technol., 60 (765) 97–106 (1988)
Yebra, DM, Kiil, S, Dam-Johansen, K, “Antifouling Technology—Past, Present and Future Steps Towards Efficient and Environmentally Friendly Antifouling Coatings.” Prog. Org. Coat., 50 (2) 75–104 (2004)
Kiosidou, ED, Karantonis, A, Pantelis, DI, Silva, ER, Bordado, JCM, “Rust Morphology Characterization of Silicone-Based Marine Antifouling Paints after Salt Spray Test on Scribed Specimens.” J. Coat. Technol. Res., 14 (2) 333–345 (2017)
Antunes, RA, Costa, I, De Faria, DLA, “Characterization of Corrosion Products Formed on Steels in the First Months of Atmospheric Exposure.” Mat. Res., 6 (3) 403–408 (2003)
Ma, Y, Li, Y, Wang, F, “The Effect of β-FeOOH on the Corrosion Behavior of Low Carbon Steel Exposed in Tropic Marine Environment.” Mater. Chem. Phys., 112 (3) 844–852 (2008)
Saha, JK, Corrosion of Constructional Steels in Marine and Industrial Environment: Frontier Work in Atmospheric Corrosion, Springer India (2013)
Dubois, F, Mendibide, C, Pagnier, T, Perrard, F, Duret, C, “Raman Mapping of Corrosion Products Formed onto Spring Steels during Salt Spray Experiments. A Correlation between the Scale Composition and the Corrosion Resistance.” Corros. Sci., 50 (12) 3401–3409 (2008)
Morcillo, M, Alcántara, J, Díaz, I, Chico, B, Simancas, J, De La Fuente, D, “Marine Atmospheric Corrosion of Carbon Steels.” Rev. Metal. Madrid, 51 (2) 1–18 (2015)
Oh, SJ, Cook, DC, Townsend, HE, “Atmospheric Corrosion of Different Steels in Marine, Rural and Industrial Environments.” Corros. Sci., 41 (9) 1687–1702 (1999)
Silva, ER, Ferreira, O, Bordado, JC, “Functionalization. Process for Biocides Immobilisation in Polymeric Matrices”, Patent application, WO2016/093719 A1, 2016
Silva, ER, Ferreira, O, Tulcidas, AV, Bayon, R, Igartua, A, Mendoza, G, Areitioaurtena, O, Bordado, JC, “Non-Releasing Biocidal Coatings: A New Eco-Friendly Strategy to Prevent Marine Biofouling”. Proc. Shipping in Changing Climates Conference, Glasgow, Scotland, November, 2015
Kim, W, Suh, CY, Cho, SW, Roh, KM, Kwon, H, Song, K, Shon, IJ, “A New Method for the Identification and Quantification of Magnetite-Maghemite Mixture Using Conventional X-Ray Diffraction Technique.” Talanta, 94 348–352 (2012)
De Faria, DLA, Venâncio Silva, S, De Oliveira, MT, “Raman Microspectroscopy of Some Iron Oxides and Oxyhydroxides.” J. Raman Spectrosc., 28 (11) 873–878 (1997)
Oh, SJ, Cook, D, Townsend, H, “Characterization of Iron Oxides Commonly Formed as Corrosion Products on Steel.” Hyperfine Interact., 112 (1) 59–66 (1998)
Legodi, M, De Waal, D, “The Preparation of Magnetite, Goethite, Hematite and Maghemite of Pigment Quality from Mill Scale Iron Waste.” Dyes Pigments, 74 (1) 161–168 (2007)
Boucherit, N, Hugot-Le Goff, A, Joiret, S, Beranger, G, Chaudanson, H, “Study of Thin Oxidation Films on Iron Alloys by Raman Spectroscopy.” Thin Solid Films, 174 111–116 (1989)
Bellot-Gurlet, L, Neff, D, Reguer, S, Monnier, J, Saheb, M, Dillmann, P, “Raman Studies of Corrosion Layers Formed on Archaeological Irons in Various Media.” J. Nano Res., 8 147–156 (2009)
Li, S, Hihara, L, “A Micro-Raman Spectroscopic Study of Marine Atmospheric Corrosion of Carbon Steel: The Effect of Akaganeite.” J. Electrochem. Soc., 162 (9) C495–C502 (2015)
Dünnwald, J, Otto, A, “An Investigation of Phase Transitions in Rust Layers Using Raman Spectroscopy.” Corros. Sci., 29 (9) 1167–1176 (1989)
Das, S, Hendry, MJ, “Application of Raman Spectroscopy to Identify Iron Minerals Commonly Found in Mine Wastes.” Chem. Geol., 290 (3) 101–108 (2011)
Guzonas, DA, Rochefort, PA, Turner, CW, “Corrosion Product Characterisation by Fibre Optic Raman Spectroscopy”, Proc. Corrosion ‘98 Conference, San Diego, CA,USA, March 1998
Thibeau, RJ, Brown, CW, Heidersbach, RH, “Raman Spectra of Possible Corrosion Products of Iron.” Appl. Spectrosc., 32 (6) 532–535 (1978)
Colomban, P, “Potential and Drawbacks of Raman (Micro) Spectrometry for the Understanding of Iron and Steel Corrosion.” In: Chiaberge, M (ed.) New Trends and Developments in Automotive System Engineering, pp. 567–584. INTECH Open Access Publisher (2011)
Rémazeilles, C, Refait, P, “On the Formation of β-FeOOH (Akaganéite) in Chloride-Containing Environments.” Corros. Sci., 49 (2) 844–857 (2007)
Hanesch, M, “Raman Spectroscopy of Iron Oxides and (Oxy) Hydroxides at Low Laser Power and Possible Applications in Environmental Magnetic Studies.” Geophys. J. Int., 177 (3) 941–948 (2009)
Schwertmann, U, Cornell, RM, Iron Oxides in the Laboratory: Preparation and Characterization. Wiley, London (2008)
De Faria, DLA, Lopes, FN, “Heated Goethite and Natural Hematite: Can Raman Spectroscopy be Used to Differentiate Them?” Vib. Spectrosc., 45 (2) 117–121 (2007)
Antunes, RA, Ichikawa, RU, Martinez, LG, Costa, I, “Characterization of Corrosion Products on Carbon Steel Exposed to Natural Weathering and to Accelerated Corrosion Tests.” Int. J. Corr., 2014 1–9 (2014)
Bano, H, “Morphology and Composition of Corrosion Products Formed on Painted Mild Steel in Marine Atmosphere after 4, 8 and 12 Months Exposure.” Int. J. Adv. Res., 3 (4) 318–325 (2015)
Raman, A, Nasrazadani, S, Sharma, L, “Morphology of Rust Phases Formed on Weathering Steels in Various Laboratory Corrosion Tests.” Metallography, 22 (1) 79–96 (1989)
Gilbert, F, Refait, P, Lévêque, F, Remazeilles, C, Conforto, E, “Synthesis of Goethite from Fe(OH)2 Precipitates: Influence of Fe(II) Concentration and Stirring Speed.” J. Phys. Chem. Solids, 69 (8) 2124–2130 (2008)
Hallam, PM, Gómez-Mingot, M, Kampouris, DK, Banks, CE, “Facile Synthetic Fabrication of Iron Oxide Particles and Novel Hydrogen Superoxide Supercapacitors.” RSC Advances, 2 (16) 6672–6679 (2012)
Ma, Y, Li, Y, Wang, F, “Corrosion of Low Carbon Steel in Atmospheric Environments of Different Chloride Content.” Corros. Sci., 51 (5) 997–1006 (2009)
Koleva, D, van Breugel, K, De Wit, JHW, Boshkov, N, Fraaij, ALA, “Composition and Morphology of Product Layers in the Steel/Cement Paste Interface in Conditions of Corrosion and Cathodic Protection in Reinforced Concrete.” ECS Transactions, 2 (9) 127–139 (2007)
Oh, SJ, Cook, DC, Carpio, JJ, “Characterization of the Corrosion Products Formed on Carbon Steel in a Marine Environment.” J. Korean Phys. Soc., 36 (2) 106–110 (2000)
De La Fuente, D, Díaz, I, Simancas, J, Chico, B, Morcillo, M, “Long-Term Atmospheric Corrosion of Mild Steel.” Corros. Sci., 53 (2) 604–617 (2011)
Morales, A, “An X-Ray Diffraction Study of Corrosion Products from Low Carbon Steel,” Rev. Metal. Madrid., 39 (extra) 28–31 (2003)
Cook, DC, “Spectroscopic Identification of Protective and Non-protective Corrosion Coatings on Steel Structures in Marine Environments.” Corros. Sci., 47 (10) 2550–2570 (2005)
Keiser, JT, Brown, CW, Heidersbach, RH, “Characterization of the Passive Film Formed on Weathering Steels.” Corros. Sci., 23 (3) 251–259 (1983)
Acknowledgments
The experimental paint investigated in this work was developed in the framework of the European collaborative project FP7 FOUL-X-SPEL (Grant Agreement 285552). The authors would also like to thank Ms. E. Rosolymou and Ms. E. Siranidi for their support on obtaining the XRD and Raman measurements, respectively.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kiosidou, E.D., Karantonis, A., Pantelis, D.I. et al. Rust morphology characterization of polyurethane and acrylic-based marine antifouling paints after salt spray test on scribed specimens. J Coat Technol Res 14, 1381–1395 (2017). https://doi.org/10.1007/s11998-017-9939-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11998-017-9939-0