Abstract
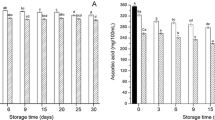
Various enzymes are responsible for the quality and color degradation of sugarcane juice and need to be inactivated through suitable interventions. In this study, microfluidization of sugarcane juice was attempted over a pressure range of 50–200 MPa with 1–7 processing cycles to remediate the problem. The impact of microfluidization on the activity of polyphenol oxidase (PPO), peroxidase (POD), and sucrose neutral invertase (SNI) along with the sugar, color, and chlorophyll content of sugarcane juice was assessed. Results showed that microfluidization reduced the PPO activity by 39.4–64.7% and POD activity by 16.4–75.0%. However, SNI activity was restricted only at specific processing conditions. Reducing sugar increased with microfluidization but was found comparable to control, while total sugars decreased by 11–54%. Chlorophyll content increased up to 100 MPa pressure and showed no degradation at higher pressures. Juice color was relatively stable up to a pressure of 150 MPa with distinct high intensity of greenness. Fourier transform infrared (FTIR) spectra of the processed juice revealed that amide II band in the structure of PPO and POD was more susceptible to pressure treatment, while SNI can be more effectively reduced at 50 and 100 MPa. Genetic algorithm-based optimization showed 159 MPa pressure/1 cycle as suitable processing condition for maintaining the quality of sugarcane juice. Although microfluidization significantly reduced the activity of deteriorative enzymes in sugarcane juice while maintaining its color characteristics, complete inactivation could not be achieved which could hinder long-term preservation. In this regard, microfluidization-assisted technologies could be a future breakthrough for juice preservation.







Similar content being viewed by others
Data Availability
All data has been provided within the article. Additionally, supplementary data has been provided along with this manuscript on the effect of microfluidization processing conditions (pressure, 50–200 MPa; cycles, 1–7) on the polyphenol and antioxidant potential of sugarcane juice.
References
AOAC (2011). Method 2002.02 and method 2009.01. In W. Horwitz & G. Latimer (Eds.), Rev 4 (18th ed.). Maryland: AOAC International, Gaithersburg.
Augusto, P. E., Tribst, A. A., & Cristianini, M. (2018). High hydrostatic pressure and high-pressure homogenization processing of fruit juices. In Fruit Juices, 393–421 Academic Press.
Azodi, M., Falamaki, C., & Mohsenifar, A. (2011). Sucrose hydrolysis by invertase immobilized on functionalized porous silicon. Journal of Molecular Catalysis B: Enzymatic, 69(3-4), 154–160.
Baltacıoğlu, H., Bayındırlı, A., Severcan, M., & Severcan, F. (2015). Effect of thermal treatment on secondary structure and conformational change of mushroom polyphenol oxidase (PPO) as food quality related enzyme: A FTIR study. Food Chemistry, 187, 263–269.
Brochier, B., Mercali, G. D., & Marczak, L. D. F. (2018). Effect of ohmic heating parameters on peroxidase inactivation, phenolic compounds degradation and color changes of sugarcane juice. Food and Bioproducts Processing, 111, 62–71.
Bucheli, C. S., & Robinson, S. P. (1994). Contribution of enzymic browning to color in sugarcane juice. Journal of Agricultural and Food Chemistry, 42(2), 257–261.
Chauhan, O. P., Ravi, N., Roopa, N., Kumar, S., & Raju, P. S. (2017). High pressure, temperature and time-dependent effects on enzymatic and microbial properties of fresh sugarcane juice. Journal of Food Science and Technology, 54(12), 4135–4138.
de Medeiros, J. K., Sarkis, J. R., Jaeschke, D. P., & Mercali, G. D. (2020). Thermosonication for peroxidase inactivation in sugarcane juice. LWT, 110730.
Dubois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. T., & Smith, F. (1956). Colorimetric method for determination of sugars and related substances. Analytical Chemistry, 28(3), 350-356.
Eisenmenger, M. J., & Reyes-De-Corcuera, J. I. (2009). High pressure enhancement of enzymes: A review. Enzyme and Microbial Rechnology, 45(5), 331–347.
Etzbach, L., Pfeiffer, A., Schieber, A., & Weber, F. (2019). Effects of thermal pasteurization and ultrasound treatment on the peroxidase activity, carotenoid composition, and physicochemical properties of goldenberry (Physalis peruviana L.) puree. LWT- Food Science and Technology, 100, 69–74.
G. L. Miller, (1959) Use of Dinitrosalicylic Acid Reagent for Determination of Reducing Sugar. Analytical Chemistry, 31 (3):426-428.
Hanover, L. M., & White, J. S. (1993). Manufacturing, composition, and applications of fructose. The American Journal of Clinical Nutrition, 58(5), 724S–732S.
Huang, H. W., Chang, Y. H., & Wang, C. Y. (2015). High pressure pasteurization of sugarcane juice: Evaluation of microbiological shelf life and quality evolution during refrigerated storage. Food and Bioprocess Technology, 8(12), 2483–2494.
Iqbal, A., Murtaza, A., Hu, W., Ahmad, I., Ahmed, A., & Xu, X. (2019). Activation and inactivation mechanisms of polyphenol oxidase during thermal and non-thermal methods of food processing. Food and Bioproducts Processing, 117, 170–182.
Ivanov, Y. D., Pleshakova, T. O., Shumov, I. D., Kozlov, A. F., Valueva, A. A., Ivanova, I. A., Ershova, M. O., Larionov, D. I., Repnikov, V. V., Ivanova, N. D., Tatur, V. Y., Stepanov, I. N., & Ziborov, V. S. (2021). AFM and FTIR investigation of the effect of water flow on horseradish peroxidase. Molecules, 26(2), 306.
Kohli, G., Jain, G., Bisht, A., Upadhyay, A., Kumar, A., & Dabir, S. (2019). Effect of non-thermal hurdles in shelf life enhancement of sugarcane juice. LWT, 112, 108233.
Kumar, D., Tarafdar, A., Kumar, Y., & Badgujar, P. C. (2019). Intelligent modelling and detailed analysis of drying, hydration, thermal and spectral characteristics for convective drying of chicken breast slices. Journal of Food Process Engineering, e13087.
Li, R., Wang, Y., Ling, J., & Liao, X. (2017). Effects of high pressure processing on activity and structure of soluble acid invertase in mango pulp, crude extract, purified form and model systems. Food Chemistry, 231, 96–104.
Liu, W., Liu, J., Xie, M., Liu, C., Liu, W., & Wan, J. (2009a). Characterization and high-pressure microfluidization-induced activation of polyphenoloxidase from Chinese pear (Pyrus pyrifolia Nakai). Journal of Agricultural and Food Chemistry, 57(12), 5376–5380.
Liu, W., Liu, J., Liu, C., Zhong, Y., Liu, W., & Wan, J. (2009b). Activation and conformational changes of mushroom polyphenoloxidase by high pressure microfluidization treatment. Innovative Food Science & Emerging Technologies, 10(2), 142–147.
Mao, L. C., Xu, Y. Q., & Que, F. (2007). Maintaining the quality of sugarcane juice with blanching and ascorbic acid. Food Chemistry, 104(2), 740–745.
Oliete, B., Potin, F., Cases, E., & Saurel, R. (2019). Microfluidization as homogenization technique in pea globulin-based emulsions. Food and Bioprocess Technology, 12(5), 877–882.
Qudsieh, H. Y. M., Yusof, S., Osman, A., & Rahman, R. A. (2002). Effect of maturity on chlorophyll, tannin, color, and polyphenol oxidase (PPO) activity of sugarcane juice (Saccharum officinarum Var. Yellow Cane). Journal of Agricultural and Food Chemistry, 50(6), 1615–1618.
Saxena, J., Makroo, H. A., Bhattacharya, S., & Srivastava, B. (2018). Kinetics of the inactivation of polyphenol oxidase and formation of reducing sugars in sugarcane juice during Ohmic and conventional heating. Journal of Food Process Engineering, 41(3), e12671.
Sirohi, R., Singh, A., Tarafdar, A., & Shahi, N. C. (2018). Application of genetic algorithm in modelling and optimization of cellulase production. Bioresource Technology, 270, 751–754.
Sreedevi, P., Jayachandran, L. E., & Rao, P. S. (2019). Kinetic modeling of high-pressure induced inactivation of polyphenol oxidase in sugarcane juice (Saccharum officinarum). Journal of the Science of Food and Agriculture, 99(5), 2365–2374.
Stuart, B. H. (2004). Infrared spectroscopy: Fundamentals and applications (pp. 244). Wiley.
Sugarcane Breeding Institute. (2020). Sugarcane, sugar and molasses production at a glance. ICAR, 1–4.
Sun, C., Yang, J., Liu, F., Yang, W., Yuan, F., & Gao, Y. (2016). Effects of dynamic high-pressure microfluidization treatment and the presence of quercetagetin on the physical, structural, thermal, and morphological characteristics of zein nanoparticles. Food and Bioprocess Technology, 9(2), 320–330.
Tabora, V. (2018). Understanding chroma and luminance in digital imaging. https://medium.com/hd-pro/understanding-chroma-and-luminance-in-digital-imaging-f0b4d97ee157. Accessed on 8 October 2020.
Tarafdar, A., & Shahi, N. C. (2018). Application and comparison of genetic and mathematical optimizers for freeze-drying of mushrooms. Journal of Food Science and Technology, 55(8), 2945–2954.
Tarafdar, A., Nair, S. G., & Kaur, B. P. (2019). Identification of microfluidized conditions for quality retention of sugarcane juice using genetic algorithm. Food and Bioprocess Technology, 12(11), 1874–1886.
Tarafdar, A., Kaur, B. P., Nema, P. K., Babar, O. A., & Kumar, D. (2020). Using a combined neural network ─ Genetic algorithm approach for predicting the complex rheological characteristics of microfluidized sugarcane juice. LWT- Food Science and Technology, 123, 109058.
Tarafdar, A., Kumar, Y., Kaur, B. P., & Badgujar, P. C. (2021). High-pressure microfluidization of sugarcane juice − Effect on total phenols, total flavonoids, antioxidant activity and microbiological quality. Journal of Food Processing and Preservation, e15428.
Taranto, F., Pasqualone, A., Mangini, G., Tripodi, P., Miazzi, M. M., Pavan, S., & Montemurro, C. (2017). Polyphenol oxidases in crops: Biochemical, physiological and genetic aspects. International Journal of Molecular Sciences, 18(2), 377.
Acknowledgement
The first author thanks the University Grants Commission of India for providing financial assistance for this work vide grant no. F1-17.1/2017-18/RGNF-2017-18-SC-RAJ-37149. The authors also thank Dr. Prarabdh Badgujar, Department of Food Science and Technology, NIFTEM, Sonipat, for providing valuable resources for carrying out this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 39 kb)
Rights and permissions
About this article
Cite this article
Tarafdar, A., Kaur, B.P. & Pareek, S. Effect of Microfluidization on Deteriorative Enzymes, Sugars, Chlorophyll, and Color of Sugarcane Juice. Food Bioprocess Technol 14, 1375–1385 (2021). https://doi.org/10.1007/s11947-021-02651-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-021-02651-w