Abstract
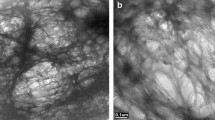
In this paper, multilayer pectin-beeswax/colophony-pectin (P-BC-P) films including different proportions of beeswax/colophony mixtures were prepared in order to reduce the water vapor permeability. FTIR, XRD, DSC, polarized light microscopy (PLM), and water vapor permeation assays were performed. Characterization techniques showed (i) polar interactions between beeswax and colophony at the amorphous phase, (ii) changes in beeswax crystalline phase from sponge-like to needle-like structure, and (iii) formation of a eutectic mixture at BC3 70/30 ratio which guides the epitaxial crystallization of beeswax. Pure pectin films showed low resistance to the water vapor permeation (361 × 10−13 g m m−2 s−1 Pa−1), while multilayer films showed major control over the transport process. P-BC3-P showed one of the lowest water vapor permeability (WVP) values (56 × 10−13 g m m−2 s−1 Pa−1) and the closest WVP value to that of polyethylene films (LDPE 5.8 × 10−13 g m m−2 s−1 Pa−1). This result was attributed to the ordered crystalline structure reached by the epitaxial crystallization of beeswax within the hydrophobic phase.





Similar content being viewed by others
References
Abdelhak, M. (2019). A review: Application of biopolymers in the pharmaceutical formulation, (1), 15–25.
Abdikheibari, S., Parvizi, R., Moayed, M. H., Zebarjad, S. M., & Sajjadi, S. A. (2015). Beeswax-colophony blend: A novel green organic coating for protection of steel drinking water storage tanks. Metals, 5(3), 1645–1664.
Alves, V. D., Costa, N., & Coelhoso, I. M. (2010). Barrier properties of biodegradable composite films based on kappa-carrageenan/pectin blends and mica flakes. Carbohydrate Polymers, 79(2), 269–276.
ASTM, E. (n.d.) (96). Standard test methods for water vapor transmission of materials. ASTM International: West, Conshohocken, PA.
Attama, A. A., Schicke, B. C., & Müller-Goymann, C. C. (2006). Further characterization of theobroma oil–beeswax admixtures as lipid matrices for improved drug delivery systems. European Journal of Pharmaceutics and Biopharmaceutics, 64(3), 294–306.
Basch, C. Y., Jagus, R. J., & Flores, S. K. (2013). Physical and antimicrobial properties of tapioca starch-HPMC edible films incorporated with nisin and/or potassium sorbate. Food and Bioprocess Technology, 6(9), 2419–2428.
Borchman, D. (2014). From the bench to the bedside: Infrared spectroscopy and the diagnosis and treatment of dry eye and cataracts. Spectroscopy, 29(2), 38–52.
Borchman, D., Foulks, G. N., Yappert, M. C., & Ho, D. V. (2007). Temperature-induced conformational changes in human tearlipids hydrocarbon chains. Biopolymers: Original Research on Biomolecules, 87(2–3), 124–133.
Cruces, F., García, M. G., & Ochoa, N. A. (2019). Can the maximum volume fraction ensure optimum reinforcement in short-fiber composites? Journal of Applied Polymer Science, 136(31), 47821.
Crucitti, V. C., Migneco, L. M., Piozzi, A., Taresco, V., Garnett, M., Argent, R. H., & Francolini, I. (2018). Intermolecular interaction and solid state characterization of abietic acid/chitosan solid dispersions possessing antimicrobial and antioxidant properties. European Journal of Pharmaceutics and Biopharmaceutics, 125, 114–123.
Da Silva, D. C., Lopes, I. A., Da Silva, L. J. S., Lima, M. F., Barros Filho, A. K. D., Villa-Vélez, H. A., & Santana, A. A. (2019). Physical properties of films based on pectin and babassu coconut mesocarp. International journal of biological macromolecules, 130, 419–428.
Debeaufort, F., Quezada-Gallo, J.-A., Delporte, B., & Voilley, A. (2000). Lipid hydrophobicity and physical state effects on the properties of bilayer edible films. Journal of Membrane Science, 180(1), 47–55.
Dorset, D. L. (1999). Development of lamellar structures in natural waxes-an electron diffraction investigation. Journal of Physics D: Applied Physics, 32(11), 1276–1280.
Dorset, D. L. (2013). Structural electron crystallography. Springer Science & Business Media.
Favvas, E. P., Kouvelos, E. P., Papageorgiou, S. K., Tsanaktsidis, C. G., & Mitropoulos, A. C. (2015). Characterization of natural resin materials using water adsorption and various advanced techniques. Applied Physics A, 119(2), 735–743.
Fraga, S. C., Azevedo, M. A., Coelhoso, I. M., Brazinha, C., & Crespo, J. G. (2018). Steady-state and transient transport studies of gas permeation through dense membranes using on-line mass spectrometry. Separation and Purification Technology, 197, 18–26.
Gaillard, Y., Mija, A., Burr, A., Darque-Ceretti, E., Felder, E., & Sbirrazzuoli, N. (2011). Green material composites from renewable resources: Polymorphic transitions and phase diagram of beeswax/rosin resin. Thermochimica acta, 521(1–2), 90–97.
Jana, S. (2016). Crystallization behavior of waxes. Utah State University.
Jana, S., & Martini, S. (2014). Effect of high-intensity ultrasound and cooling rate on the crystallization behavior of beeswax in edible oils. Journal of agricultural and food chemistry, 62(41), 10192–10202.
Jana, S., & Martini, S. (2016). Phase behavior of binary blends of four different waxes. Journal of the American Oil Chemists’ Society, 93(4), 543–554.
Jenning, V., & Gohla, S. (2000). Comparison of wax and glyceride solid lipid nanoparticles (SLN®). International journal of pharmaceutics, 196(2), 219–222.
Jiménez, A., Fabra, M. J., Talens, P., & Chiralt, A. (2012). Edible and biodegradable starch films: A review. Food and Bioprocess Technology, 5(6), 2058–2076.
Jo, C., Kang, H., Lee, N. Y., Kwon, J. H., & Byun, M. W. (2005). Pectin-and gelatin-based film: Effect of gamma irradiation on the mechanical properties and biodegradation. Radiation Physics and Chemistry, 72(6), 745–750.
Kameda, T. (2004). Molecular structure of crude beeswax studied by solid-state 13C NMR. Journal of Insect Science, 4(1).
Kasirajan, S., & Ngouajio, M. (2012). Polyethylene and biodegradable mulches for agricultural applications: A review. Agronomy for Sustainable Development, 32(2), 501–529.
Khurana, R., Singh, K., Sapra, B., Tiwary, A. K., & Rana, V. (2014). Tamarindus indica pectin blend film composition for coating tablets with enhanced adhesive force strength. Carbohydrate polymers, 102, 55–65.
Kristo, E., Biliaderis, C. G., & Zampraka, A. (2007). Water vapour barrier and tensile properties of composite caseinate-pullulan films: Biopolymer composition effects and impact of beeswax lamination. Food chemistry, 101(2), 753–764.
Leonid, V. (1968). Elements of X-ray crystallography. McGraw-Hill.
Michaels, A. S., & Parker Jr., R. B. (1959). Sorption and flow of gases in polyethylene. Journal of Polymer Science, 41(138), 53–71.
Ochoa, T. A., Almendárez, B. E. G., Reyes, A. A., Pastrana, D. M. R., López, G. F. G., Belloso, O. M., & Regalado-González, C. (2017). Design and characterization of corn starch edible films including beeswax and natural antimicrobials. Food and bioprocess technology, 10(1), 103–114.
Pasini Cabello, S., Takara, E. A., Marchese, J., & Ochoa, N. A. (2015). Influence of plasticizers in pectin films: Microstructural changes. Materials Chemistry and Physics, 162, 491–497.
Pattanashetti, N. A., Heggannavar, G. B., & Kariduraganavar, M. Y. (2017). Smart biopolymers and their biomedical applications. Procedia Manufacturing, 12, 263–279.
Pawar, P. A. (2013). Aachal. H. Purwar. Bioderadable polymers in food packaging. American Journal of Engineering Research (AJER), 2, 151–164.
Preda, F.-M., Alegría, A., Bocahut, A., Fillot, L.-A., Long, D. R., & Sotta, P. (2015). Investigation of water diffusion mechanisms in relation to polymer relaxations in polyamides. Macromolecules, 48(16), 5730–5741.
Rebelo, R., Fernandes, M., & Fangueiro, R. (2017). Biopolymers in medical implants: A brief review. Procedia engineering, 200, 236–243.
Sängerlaub, S., Schmid, M., & Müller, K. (2018). Comparison of water vapour transmission rates of monolayer films determined by water vapour sorption and permeation experiments. Food packaging and shelf life, 17, 80–84.
Schoening, F. R. L. (1980). The X-ray diffraction pattern and deformation texture of beeswax. South African Journal of Science, 76(6), 262–265.
Shih, F. F., Daigle, K. W., & Champagne, E. T. (2011). Effect of rice wax on water vapour permeability and sorption properties of edible pullulan films. Food Chemistry, 127(1), 118–121.
Silva, H. D., Cerqueira, M. Â., & Vicente, A. A. (2012). Nanoemulsions for food applications: Development and characterization. Food and Bioprocess Technology, 5(3), 854–867.
Singh, A. V. (2011). Biopolymers in drug delivery: A review. Pharmacologyonline, 1, 666–674.
Slavutsky, A. M., & Bertuzzi, M. A. (2016). Improvement of water barrier properties of starch films by lipid nanolamination. Food Packaging and Shelf Life, 7, 41–46.
Soazo, M., Rubiolo, A. C., & Verdini, R. A. (2011). Effect of drying temperature and beeswax content on physical properties of whey protein emulsion films. Food Hydrocolloids, 25(5), 1251–1255.
Sudesh, K., & Iwata, T. (2008). Review: Sustainability of biobased and biodegradable plastics, 36, 433–442.
Svečnjak, L., Baranović, G., Vinceković, M., & Pr\djun, S., Bubalo, D., & Gajger, I. T. (2015). An approach for routine analytical detection of beeswax adulteration using FTIR-ATR spectroscopy. Journal of apicultural science, 59(2), 37–49.
Syahida, N., Fitry, I., Zuriyati, A., & Hanani, N. (2020). Effects of palm wax on the physical, mechanical and water barrier properties of fish gelatin films for food packaging application. Food Packaging and Shelf Life, 23, 100437.
Takara, E. A., Marchese, J., & Ochoa, N. A. (2015). NaOH treatment of chitosan films: Impact on macromolecular structure and film properties. Carbohydrate polymers, 132, 25–30.
Tavernier, I., Doan, C. D., Van de Walle, D., Danthine, S., Rimaux, T., & Dewettinck, K. (2017). Sequential crystallization of high and low melting waxes to improve oil structuring in wax-based oleogels. RSC advances, 7(20), 12113–12125.
Wittmann, J. C., Hodge, A. M., & Lotz, B. (1983). Epitaxial crystallization of polymers onto benzoic acid: Polyethylene and paraffins, aliphatic polyesters, and polyamides. Journal of Polymer Science: Polymer Physics Edition, 21(12), 2495–2509.
Zhang, W., Xiao, H., & Qian, L. (2014). Enhanced water vapour barrier and grease resistance of paper bilayer-coated with chitosan and beeswax. Carbohydrate polymers, 101, 401–406.
Funding
This work was supported by grants PROICO 2-2116 Universidad Nacional de San Luis and PIP-No11220130100448CO CONICET.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
This manuscript has not been published, and it is not under consideration for publication elsewhere.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cruces, F., García, M.G. & Ochoa, N.A. Reduction of Water Vapor Permeability in Food Multilayer Biopackaging by Epitaxial Crystallization of Beeswax. Food Bioprocess Technol 14, 1244–1255 (2021). https://doi.org/10.1007/s11947-021-02628-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-021-02628-9