Abstract
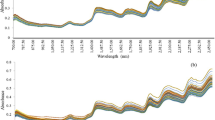
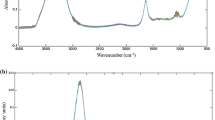
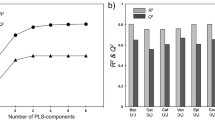
Determining phenolic compounds of wood and its extractability to the hydroalcoholic medium is important in the oenological industry. The method proposed in this study copes with this issue in an in situ, non-destructive, and fast way. For this purpose, a number of oak by-product samples spectrally representative have been selected. Selected spectral data have been correlated with oak wood extractable polyphenols (extractable total phenolic content and extractable ellagitannin content) by modified partial least squares regression (MPLS) obtaining coefficients of determination (RSQ) greater than 0.9 and standard errors of prediction (SEP) between 13.68 and 23.51% for all parameters evaluated. The obtained results are comparable with those obtained using bench-top devices and present the advantage of its eventual friendly use out of lab. Development of applicable models in situ will allow a greater versatility and efficiency for the decision-making in the winemaking process on the adequacy and/or dosage of these by-products according to the requirements of the wine. The use of cooperage by-products as the source of copigments for wine leads to a sustainable and competitive cooperage industry, through waste reduction and by-product valorization.



Similar content being viewed by others
References
Abu Izneid, B., Fadhel, M. I., Al-Kharazi, T., Ali, M., & Miloud, S. (2014). Design and develop a nondestructive infrared spectroscopy instrument for assessment of mango (Mangifera indica) quality. Journal of Food Science and Technology-Mysore, 51(11), 3244–3252.
Antonucci, F., Pallottino, F., Paglia, G., Palma, A., D’Aquino, S., & Menesatti, P. (2011). Non-destructive estimation of mandarin maturity status through portable Vis-NIR spectrophotometer. Food and Bioprocess Technology, 4(5), 809-813
Baca-Bocanegra, B., Nogales-Bueno, J., Hernández-Hierro, J. M., & Heredia, F. J. (2018). Evaluation of extractable polyphenols released to wine from cooperage byproduct by near infrared hyperspectral imaging. Food Chemistry, 244, 206–212.
Balík, J., Híc, P., Kulichová, J., Novotná, P., Tříska, J., Vrchotová, N., et al. (2017). Musts with increased lignan content through addition of lignan extracts. Food and Bioprocess Technology, 10(7), 1367–1373.
Boulton, R. (2001). The copigmentation of anthocyanins and its role in the color of red wines. A critical review. American Journal of Enology and Viticulture, 52, 67–87.
Brereton, R. G. (2003). Chemometrics: data analysis for the laboratory and chemical plant. Chichester, West Sussex, England: J. Wiley.
Camps, C., Simone, C., & Gilli, C. (2012). Assessment of tomato quality using portable NIR spectroscopy and PLSR with wavelengths selection. Acta Horticulturae, 936, 437–442.
Cozzolino, D., Dambergs, R. G., Janik, L., Cynkar, W. U., & Gishen, M. (2006). Analysis of grapes and wine by near infrared spectroscopy. Journal of Near Infrared Spectroscopy, 14(5), 279–289.
Chassaing, S., Lefeuvre, D., Jacquet, R., Jourdes, M., Ducasse, L., Galland, S., et al. (2010). Physicochemical studies of new anthocyano-ellagitannin hybrid pigments: about the origin of the influence of oak C-glycosidic ellagitannins on wine color. European Journal of Organic Chemistry, 1, 55–63.
Chira, K., & Teissedre, P.-L. (2013). Extraction of oak volatiles and ellagitannins compounds and sensory profile of wine aged with French winewoods subjected to different toasting methods: behaviour during storage. Food Chemistry, 140(1-2), 168–177.
Del Álamo, M., Nevares, I., Gallego, L., Martin, C., & Merino, S. (2008). Aging markers from bottled red wine aged with chips, staves and barrels. Analytica Chimica Acta, 621(1), 86–99.
Dhanoa, M. S., Lister, S. J. and Barnes, R. J. (1995). On the scales associated with near-infrared reflectance difference spectra. Applied Spectroscopy 49: 765–772.
Doussot, F., De Jéso, B., Quideau, S., & Pardon, P. (2002). Extractives content in cooperage oak wood during natural seasoning and toasting; influence of tree species, geographic location, and single-tree effects. Journal of Agricultural and Food Chemistry, 50, 5955–5961.
Escribano-Bailón, M. T., & Santos-Buelga, C. (2012). Anthocyanin copigmentation - evaluation, mechanisms and implications for the colour of red wines. [Article]. Current Organic Chemistry, 16(6), 715–723.
Ferrer-Gallego, R., Hernández-Hierro, J. M., Rivas-Gonzalo, J. C., & Escribano-Bailón, M. T. (2010). Feasibility study on the use of near infrared spectroscopy to determine flavanols in grape seeds. Talanta, 82(5), 1778–1783.
Ferrer-Gallego, R., Hernández-Hierro, J. M., Rivas-Gonzalo, J. C., & Escribano-Bailón, M. T. (2011). Determination of phenolic compounds of grape skins during ripening by NIR spectroscopy. LWT - Food Science and Technology, 44(4), 847–853.
García-Estévez, I., Alcalde-Eon, C., Le Grottaglie, L., Rivas-Gonzalo, J. C., & Escribano-Bailón, M. T. (2015). Understanding the ellagitannin extraction process from oak wood. Tetrahedron, 71(20), 3089–3094.
García-Estévez, I., Alcalde-Eon, C., María Martínez-Gil, A., Rivas-Gonzalo, J. C., Teresa Escribano-Bailón, M., Nevares, I., et al. (2017). An approach to the study of the interactions between ellagitannins and oxygen during oak wood aging. Journal of Agricultural and Food Chemistry, 65(31), 6369–6378.
García-Estévez, I., Escribano-Bailón, M. T., Rivas-Gonzalo, J. C., & Alcalde-Eon, C. (2010). Development of a fractionation method for the detection and identification of oak ellagitannins in red wines. Analytica Chimica Acta, 660(1-2), 171–176.
García-Estévez, I., Escribano-Bailón, M. T., Rivas-Gonzalo, J. C., & Alcalde-Eon, C. (2012). Validation of a mass spectrometry method to quantify oak ellagitannins in wine samples. [Article]. Journal of Agricultural and Food Chemistry, 60(6), 1373–1379.
García-Estévez, I., Gavara, R., Alcalde-Eon, C., Rivas-Gonzalo, J. C., Quideau, S., Escribano-Bailón, M. T., et al. (2013a). Thermodynamic and kinetic properties of a new myrtillin–vescalagin hybrid pigment. Journal of Agricultural and Food Chemistry, 61(47), 11569–11578.
García-Estévez, I., Jacquet, R., Alcalde-Eon, C., Rivas-Gonzalo, J. C., Escribano-Bailón, M. T., & Quideau, S. (2013b). Hemisynthesis and structural and chromatic characterization of delphinidin 3-O-glucoside-vescalagin hybrid pigments. Journal of Agricultural and Food Chemistry, 61(47), 11560–11568.
Geladi, P., MacDougall, D. and Martens, H. (1985). Linearization and scatter-correction for near-infrared reflectance spectra of meat. Appl. Spectrosc. 39, 491–500.
Giordanengo, T., Charpentier, J. P., Boizot, N., Roussel, S., Roger, J. M., Chaix, G., et al. (2009). Oakscan: procédé de mesure rapide et non destructif des polyphénols du bois de chêne de tonnellerie. Revue Francaise d'Oenologie, 10–15.
González-Caballero, V., Sánchez, M.-T., Fernández-Novales, J., López, M.-I., & Pérez-Marín, D. (2012). On-vine monitoring of grape ripening using near-infrared spectroscopy. Food Analytical Methods, 5(6), 1377–1385.
Guidetti, R., Beghi, R., & Bodria, L. (2010). Evaluation of frape quality parameters by a simple VIS/NIR system. Transactions of the ASABE, 53(2), 477–484.
Hernández-Hierro, J. M., Nogales-Bueno, J., Rodríguez-Pulido, F. J., & Heredia, F. J. (2013). Feasibility study on the use of near-infrared hyperspectral imaging for the screening of anthocyanins in intact grapes during ripening. Journal of Agricultural and Food Chemistry, 61(41), 9804–9809.
Jourdes, M., Michel, J., Saucier, C., Quideau, S., & Teissedre, P. L. (2011). Identification, amounts, and kinetics of extraction of C-glucosidic ellagitannins during wine aging in oak barrels or in stainless steel tanks with oak chips. Analytical and Bioanalytical Chemistry, 401(5), 1531–1539.
Kemps, B., Leon, L., Best, S., De Baerdemaeker, J., & De Ketelaere, B. (2010). Assessment of the quality parameters in grapes using VIS/NIR spectroscopy. Biosystems Engineering, 105(4), 507-513
Larrain, M., Guesalaga, A. R., & Agosin, E. (2008). A multipurpose portable instrument for determining ripeness in wine grapes using NIR spectroscopy. IEEE Transactions on Instrumentation and Measurement, 57(2), 294–302.
Masson, G., Moutounet, M., & Puech, J. L. (1995). Ellagitannin content of oak wood as a function of species and of sampling position in the tree. American Journal of Enology and Viticulture, 46(2), 262–268.
Michel, J., Jourdes, M., Le Floch, A., Giordanengo, T., Mourey, N., & Teissedre, P. L. (2013). Influence of wood barrels classified by NIRS on the ellagitannin content/composition and on the organoleptic properties of wine. Journal of Agricultural and Food Chemistry, 61(46), 11109–11118.
Nevares, I., Del Alamo, M., Cárcel, L. M., Crespo, R., Martin, C., & Gallego, L. (2008). Measure the dissolved oxygen consumed by red wines in aging tanks. Food and Bioprocess Technology, 2(3), 328–336.
Nogales-Bueno, J., Baca-Bocanegra, B., Rodríguez-Pulido, F. J., Heredia, F. J., & Hernández-Hierro, J. M. (2015). Use of near infrared hyperspectral tools for the screening of extractable polyphenols in red grape skins. Food Chemistry, 172, 559–564.
Osborne, B. G., Fearn, T., Hindle, P. T., & Osborne, B. G. (1993). Practical NIR spectroscopy with applications in food and beverage analysis. Harlow, Essex. In England. New York: Longman Scientific & Technical ; Wiley.
Páscoa, R. N. M. J., Machado, S., Magalhães, L. M., & Lopes, J. A. (2014). Value adding to red grape pomace exploiting eco-friendly FT-NIR spectroscopy technique. Food and Bioprocess Technology, 8(4), 865–874.
Peng, S., Scalbert, A., & Monties, B. (1991). Insoluble ellagitannins in Castanea sativa and Quercus petraea woods. Phytochemistry, 30(3), 775–778.
Ribéreau-Gayon, P., Dubourdieu, D., Doneche, B., Lonvaud, A., Glories, Y., Maujean, A., et al. (2006). Handbook of enology, the microbiology of wine and vinifications (Vol. v. 1). West Sussex, England: J. Wiley & Sons.
Rodriguez-Pulido, F. J., Hernández-Hierro, J. M., Nogales-Bueno, J., Gordillo, B., Gonzalez-Miret, M. L., & Heredia, F. J. (2014). A novel method for evaluating flavanols in grape seeds by near infrared hyperspectral imaging. Talanta, 122, 145–150.
Shenk, J. S., & Westerhaus, M. O. (1995). Routine operation,calibration, development and network system management manual. Silver Spring, Maryland: NIRSystems.
Siesler, H. W., Ozaky, Y., Kawata, S., & Heise, H. M. (2002). Near infrared spectroscopy: principles, instruments, applications. Weinheim, Germany: Wiley-VCH.
Singleton, V. L., & Rossi, J. A. (1965). Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. American Journal of Enology and Viticulture, 16, 144–158.
Snakkers, G., Nepveu, G., Guilley, E., & Cantagrel, R. (2000). Geographic, silvicultural and individual variabilities of extractive content for French sessile oaks (Quercus petraea Liebl.): polyphenols, octalactones and volatile phenols. Annals of Forest Science, 57(3), 251–260.
Viriot, C., Scalbert, A., Hervé du Penhoat, C. L. M., & Moutounet, M. (1994). Ellagitannins in woods of sessile oak and sweet chestnut dimerization and hydrolysis during wood ageing. Phytochemistry, 36(5), 1253–1260.
Vivas, N., & Glories, Y. (1996). Role of oak wood ellagitannins in the oxidation process of red wines during aging. American Journal of Enology and Viticulture, 47(1), 103–107.
Zahri, S., Moubarik, A., Charrier, F., Chaix, G., Bailleres, H., Nepveu, G., et al. (2008). Quantitative assessment of total phenol contents of European oak (Quercus petraea and Quercus robur) by diffuse reflectance NIR spectroscopy on solid wood surfaces. Holzforschung, 62(6), 679–687.
Acknowledgments
The authors thank the technical staff of the Biology Service (Servicios Generales de Investigación (SGI), Universidad de Sevilla). The authors also thank Tonelería Salas S.L. (Bollulos Par del Condado, Huelva, Spain) for supplying the cooperage by-product samples.
Funding
This work is financially supported by the Spanish Ministerio de Economía y Competitividad under the project AGL2017-84793-C2. B. Baca-Bocanegra and J. Nogales-Bueno received funding from the Universidad de Sevilla under the predoctoral grant (VPPI-II.2) and postdoctoral grant (VPPI-II.4), respectively. I. García-Estévez received funding from the FEDER-Interreg España-Portugal Programme (project ref. 0377_IBERPHENOL_6_E) under his postdoctoral grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(DOCX 93 kb)
Rights and permissions
About this article
Cite this article
Baca-Bocanegra, B., Nogales-Bueno, J., García-Estévez, I. et al. Screening of Wine Extractable Total Phenolic and Ellagitannin Contents in Revalorized Cooperage By-products: Evaluation by Micro-NIRS Technology. Food Bioprocess Technol 12, 477–485 (2019). https://doi.org/10.1007/s11947-018-2225-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-018-2225-z