Abstract
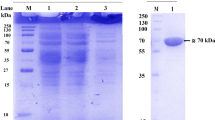
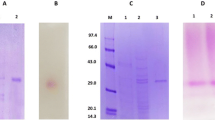
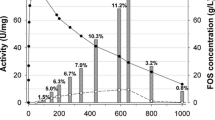
The present work was carried out with the aim to investigate some properties of an extracellular fructofuranosidase enzyme, with high transfructosylating activity, from Candida sp. LEB-I3 (Laboratory of Bioprocess Engineering, Unicamp, Brazil). The enzyme was produced through fermentation, and after cell separation from the fermented medium, the enzyme was concentrated by ethanol precipitation and than purified by anion exchange chromatography. The enzyme exhibited both fructofuranosidase (FA) and fructosyltransferase (FTA) activities on a low and high sucrose concentration. With sucrose as the substrate, the data fitted the Michaellis–Menten model for FA, showing rather a substrate inhibitory shape for fructosyltransferase activity. The K m and v max values were shown to be 13.4 g L−1 and 21.0 μmol mL−1 min−1 and 25.5 g L−1 and 52.5 μmol mL−1 min−1 for FA and FTA activities, respectively. FTA presented an inhibitory factor K i of 729.8 g L−1. The optimum conditions for FA activity were found to be pH 3.25–3.5 and temperatures around 69 °C, while for FTA, the optimum condition were 65 °C (±2 °C) and pH 4.00 (±0.25). Both activities were very stable at temperatures below 60 °C, while for FA, the best stability occurred at pH 5.0 and for FTA at pH 4.5–5.0. Despite the strong fructofuranosidase activity, causing hydrolysis of the fructooligosaccharides (FOS), the high transfructosilating activity allows a high FOS production from sucrose (44%).







Similar content being viewed by others
References
Bekers, M., Laukevics, J., Upite, D., Kaminska, E., Vigants, A., Viesturs, U., et al. (2002). Fructooligosaccharide and levan producing activity of Zymomonas mobilis extracellular levansucrase. Process Biochemistry, 38, 701–706.
Belcarz, A., Ginalska, G., Lobarzewski, J., & Penel, C. (2002). The novel non-glycosylated invertase from Candida utilis (the properties and the conditions of production and purification). Biochimica et Biophysica Acta, 1594, 40–53.
Breuil, C., & Saddler, J. N. (1985). Comparison of the 3,5-dinitrosalicylic acid and Nelson–Somogyi methods of assaying for reducing sugars and determining cellulase activity. Enzyme and Microbial Technology, 7, 327–332.
Carvalho, W., Santos, J. C., Canilha, L., Silva, S. S., Perego, P., & Converti, A. (2005). Xylitol production from sugarcane bagasse hydrolysate: Metabolic behaviour of Candida guilliermondii cells entrapped in Ca-alginate. Biochemical Engineering Journal, 25(1), 25–31.
Chen, W., & Liu, C. (1996). Production of B-fructofuranosidase by Aspergillus japonicus. Enzyme and Microbial Technology, 18, 153–160.
Chien, C., Lee, W., & Lin, T. (2001). Immobilization of Aspergillus japonicus by entrapping cells in gluten for production of fructooligosaccharides. Enzyme and Microbial Technology, 29, 252–257.
Gibson, G. R., & Roberfroid, M. D. (1995). Dietary modulation of the colonic microbiota: Introducing the concept of prebiotics. Journal of Nutrition, 125, 1401–1412.
Haaland, P. D. (1989). Experimental design in biotechnology. New York: Marcel Dekker.
Hawksworth, D. L. (2002). Why study tropical fungi? In R. Watling, J. Frankland, M. Ainsworth, S. Isaac, & C. Robinson (Eds.), Tropical mycology: Micromycetes Vol. 2 (p. 320). Cambridge: CABI.
Hidaka, H., Hirayama, M., & Sumi, N. (1988). A fructooligosaccharide-producing enzyme from Aspergillus niger ATCC 20611. Agricultural and Biology Chemistry, 52(05), 1181–1187.
Kalil, S. J., Maugeri, F., & Rodrigues, M. I. (2000). Response surface analysis and simulation as a tool for bioprocess design and optimization. Process Biochemistry, 35, 539–550.
L’Hocine, L., Wang, Z., Jiang, B., & Xu, S. (2000). Purification and partial characterization of fructosyltransferase and invertase from Aspergillus niger AS0023. Journal of Biotechnology, 81, 73–84.
Laemmli, U. K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 277, 680–685.
Lowry, O. H., Rosebroght, N. J., Farr, A. I., & Randall, R. J. (1951). Protein measurement with the folin phenol reagent. Journal of Biological Chemistry, 193, 265–275.
Maugeri, F., & Hernalsteens, S. (2007). Screening of yeast strains for transfructosylating activity. Journal of Molecular Catalysis B: Enzymatic, 49, 43–49.
Monsan, P., & Paul, F. (1995). Enzymatic synthesis of oligosaccharides. FEMS Microbiology Reviews, 16(2–3), 187–192.
Onishi, N., & Tanaka, T. (1996). Purification and properties of galacto- and gluco-oligosaccharide-producing b-glycosidase from Rhodotorula minuta IFO879. Journal of Fermentation Bioengineering, 82, 439–443.
Park, J., Oh, T., & Yun, J. W. (2001). Purification and characterization of a novel transfructosylating enzyme from Bacillus macerans EG-6. Process Biochemistry, 37, 471–476.
Rajoka, M. I., Khan, S. H., Jabbar, M. A., Awan, M. S., & Hashmi, A. S. (2006). Kinetics of batch single cell protein production from rice polishings with Candida utilis in continuously aerated tank reactors. Bioresource Technology, 97(15), 1934–1941.
Risso, F. V. A. (2004). Síntese de oligossacarídeos em meio orgânico e aquoso utilizando—Se inulinase livre e imobilizada produzida por K. marxianus ATCC 16045 e NRRL Y 7571. Ph.D. thesis, Faculty of Food Engineer, UNICAMP—Campinas, Brazil.
Rubio, M. C., Runco, R., & Navarro, A. R. (2002). Invertase from a strain of Rhodotorula glutinis. Phytochemistry, 61, 605–609.
Sageetha, P. T., Ramesh, M. N., & Prapulla, S. G. (2005). Recent trends in the microbial production, analysis and application of fructooligosaccharides. Trends in Food Science & Technology, 16, 442–457.
Trinder, R. (1969). Determination of glucose in blood using glucose with alternative oxygen acceptor. Annals of Clinical Biochemistry, 6, 27–27.
Vranešić, D., Kurtanjek, Ž., Santos, A. M. P., & Maugeri, F. (2002). Optimization of inulinase production by Kluyveromyces bulgaricus. Food Technology and Biotechnology, 40(1), 67–73.
Wang, X., & Rakshit, S. K. (2000). Iso-oligosaccharide production by multiple forms of transferase enzymes from Aspergillus foetidus. Process Biochemistry, 35, 771–775.
Yun, J. W. (1996). Fructooligosaccharides: Occurrence, preparation and application. Enzyme and Microbial Technology, 19, 107–117.
Acknowledgment
We would like to thank FAPESP, CNPq, and CAPES for the financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hernalsteens, S., Maugeri, F. Partial Purification and Characterization of Extracellular Fructofuranosidase with Transfructosylating Activity from Candida sp.. Food Bioprocess Technol 3, 568–576 (2010). https://doi.org/10.1007/s11947-008-0089-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-008-0089-3