Abstract
Purpose of review
The purpose of this paper is to provide an up-to-date review of the current knowledge on immune-mediated neuropathies secondary to antibodies against nodal and paranodal proteins.
Recent findings
Antibodies against neurofascins and contactin have recently been linked to characteristic neuropathy presentations including a more acute/subacute presentation, involvement of cranial nerves, presence of tremor, and refractory to typical first-line treatments for immune-mediated neuropathies.
Summary
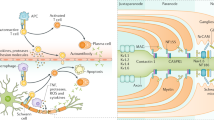
Nodopathies are a group of disorders associated with humoral autoimmunity to antigens of the node, paranode, and juxtaparanode. The clinical syndromes most commonly associated with these antibodies include acute inflammatory demyelinating polyneuropathy, chronic inflammatory demyelinating polyneuropathy, and combined central and peripheral demyelination. Treatment response is variable, and though most data reported is on NF155, there appears to be a better response to plasma exchange and rituximab followed by prednisone and then IVIG.


Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Kuwabara S, Misawa S, Mori M. Nodopathy: chronic inflammatory demyelinating polyneuropathy with anti-neurofascin 155 antibodies. J Neurol Neurosurg Psychiatry. 2017;88:459–9. https://doi.org/10.1136/jnnp-2016-315170.
Oh SJ. Nodal conduction block: a unifying concept. Muscle Nerve. 2021;63:178–80. https://doi.org/10.1002/mus.27115.
Uncini A, Kuwabara S. Nodopathies of the peripheral nerve: an emerging concept. J Neurol Neurosurg Psychiatry. 2015;86:1186–95. https://doi.org/10.1136/jnnp-2014-310097.
Stathopoulos P, Alexopoulos H, Dalakas MC. Autoimmune antigenic targets at the node of Ranvier in demyelinating disorders. Nat Rev Neurol Nature Publishing Group. 2015;11:143–56. https://doi.org/10.1038/nrneurol.2014.260.
Querol L, Devaux J, Rojas-Garcia R, Illa I. Autoantibodies in chronic inflammatory neuropathies: diagnostic and therapeutic implications. Nat Rev Neurol Nature Publishing Group. 2017;13:533–47. https://doi.org/10.1038/nrneurol.2017.84.
Wolbert J, Cheng MI, Horste GM z, Su MA. Deciphering immune mechanisms in chronic inflammatory demyelinating polyneuropathies. JCI Insight. 2020;5. https://doi.org/10.1172/jci.insight.132411.
Vural A, Doppler K, Meinl E. Autoantibodies against the node of ranvier in seropositive chronic inflammatory demyelinating polyneuropathy: diagnostic, pathogenic, and therapeutic relevance. Front Immunol. 2018;9. https://doi.org/10.3389/fimmu.2018.01029.
Fokke C, van den Berg B, Drenthen J, Walgaard C, van Doorn PA, Jacobs BC. Diagnosis of Guillain-Barre syndrome and validation of Brighton criteria. Brain. 2014;137:33–43. https://doi.org/10.1093/brain/awt285.
Desforges JF, Ropper AH. The Guillain–Barré syndrome. N Engl J Med. 1992;326:1130–6. https://doi.org/10.1056/NEJM199204233261706.
Saperstein DS, Katz JS, Amato AA, Barohn RJ. Clinical spectrum of chronic acquired demyelinating polyneuropathies. Muscle Nerve. 2001;24:311–24. https://doi.org/10.1002/1097-4598(200103)24:3<311::AID-MUS1001>3.0.CO;2-A.
Ruts L, van Koningsveld R, van Doorn PA. Distinguishing acute-onset CIDP from Guillain-Barre syndrome with treatment related fluctuations. Neurology. 2005;65:138–40. https://doi.org/10.1212/01.wnl.0000167549.09664.b8.
Kawamura N, Yamasaki R, Yonekawa T, Matsushita T, Kusunoki S, Nagayama S, et al. Anti-neurofascin antibody in patients with combined central and peripheral demyelination. Neurology. 2013;81:714–22. https://doi.org/10.1212/WNL.0b013e3182a1aa9c.
Doneddu PE, Cocito D, Manganelli F, Fazio R, Briani C, Filosto M, et al. Atypical CIDP: diagnostic criteria, progression and treatment response. Data from the Italian CIDP Database. J Neurol Neurosurg Psychiatry. 2019;90:125–32. https://doi.org/10.1136/jnnp-2018-318714.
Katz JS, Saperstein DS, Gronseth G, Amato AA, Barohn RJ. Distal acquired demyelinating symmetric neuropathy. Neurology. 2000;54:615–5. https://doi.org/10.1212/WNL.54.3.615.
Saperstein DS, Amato AA, Wolfe GI, Katz JS, Nations SP, Jackson CE, et al. Multifocal acquired demyelinating sensory and motor neuropathy: the Lewis-Sumner syndrome. Muscle Nerve. 1999;22:560–6. https://doi.org/10.1002/(SICI)1097-4598(199905)22:5<560::AID-MUS2>3.0.CO;2-Q.
Lewis RA, Sumner AJ, Brown MJ, Asbury AK. Multifocal demyelinating neuropathy with persistent conduction block. Neurology. 1982;32:958–8. https://doi.org/10.1212/WNL.32.9.958.
Querol L, Nogales-Gadea G, Rojas-Garcia R, Martinez-Hernandez E, Diaz-Manera J, Suárez-Calvet X, et al. Antibodies to contactin-1 in chronic inflammatory demyelinating polyneuropathy. Ann Neurol. 2013;73:370–80. https://doi.org/10.1002/ana.23794.
Delmont E, Manso C, Querol L, Cortese A, Berardinelli A, Lozza A, et al. Autoantibodies to nodal isoforms of neurofascin in chronic inflammatory demyelinating polyneuropathy. Brain. 2017;140:1851–8. https://doi.org/10.1093/brain/awx124.
Querol L, Nogales-Gadea G, Rojas-Garcia R, Diaz-Manera J, Pardo J, Ortega-Moreno A, et al. Neurofascin IgG4 antibodies in CIDP associate with disabling tremor and poor response to IVIg. Neurology. 2014;82:879–86. https://doi.org/10.1212/WNL.0000000000000205.
Devaux JJ, Miura Y, Fukami Y, Inoue T, Manso C, Belghazi M, et al. Neurofascin-155 IgG4 in chronic inflammatory demyelinating polyneuropathy. Neurology. 2016;86:800–7. https://doi.org/10.1212/WNL.0000000000002418.
Dubey D, Honorat JA, Shelly S, Klein CJ, Komorowski L, Mills JR, et al. Contactin-1 autoimmunity: Serologic, neurologic, and pathologic correlates. Neurol Neuroimmunol neuroinflammation. 2020;7:e771. https://doi.org/10.1212/NXI.0000000000000771.
Ogata H, Yamasaki R, Hiwatashi A, Oka N, Kawamura N, Matsuse D, et al. Characterization of IgG4 anti-neurofascin 155 antibody-positive polyneuropathy. Ann Clin Transl Neurol. 2015;2:960–71. https://doi.org/10.1002/acn3.248.
Martinez-Martinez L, Lleixà MC, Boera-Carnicero G, Cortese A, Devaux J, Siles A, et al. Anti-NF155 chronic inflammatory demyelinating polyradiculoneuropathy strongly associates to HLA-DRB15. J Neuroinflammation. 2017;14:224. https://doi.org/10.1186/s12974-017-0996-1.
Doppler K, Appeltshauser L, Wilhelmi K, Villmann C, Dib-Hajj SD, Waxman SG, et al. Destruction of paranodal architecture in inflammatory neuropathy with anti-contactin-1 autoantibodies. J Neurol Neurosurg Psychiatry. 2015;86:720–8. https://doi.org/10.1136/jnnp-2014-309916.
•• Burnor E, Yang L, Zhou H, Patterson KR, Quinn C, Reilly MM, et al. Neurofascin antibodies in autoimmune, genetic, and idiopathic neuropathies. Neurology. 2018;90:e31–8. https://doi.org/10.1212/WNL.0000000000004773 A thorough description of the temporal evolution and isoform specificity of neurofascin antibodies in patients with autoimmune peripheral neuropathies and its implication in therapy.
Querol L, Illa I. Paranodal and other autoantibodies in chronic inflammatory neuropathies. Curr Opin Neurol. 2015;28:474–9. https://doi.org/10.1097/WCO.0000000000000233.
Querol L, Siles AM, Alba-Rovira R, Jáuregui A, Devaux J, Faivre-Sarrailh C, et al. Antibodies against peripheral nerve antigens in chronic inflammatory demyelinating polyradiculoneuropathy. Sci Rep. 2017;7:14411. https://doi.org/10.1038/s41598-017-14853-4.
Doppler K, Appeltshauser L, Villmann C, Martin C, Peles E, Krämer HH, et al. Auto-antibodies to contactin-associated protein 1 (Caspr) in two patients with painful inflammatory neuropathy. Brain. 2016;139:2617–30. https://doi.org/10.1093/brain/aww189.
Breiner A, Brannagan TH. Comparison of sensitivity and specificity among 15 criteria for chronic inflammatory demyelinating polyneuropathy. Muscle Nerve. 2014;50:40–6. https://doi.org/10.1002/mus.24088.
Van den Bergh PYK, Hadden RDM, Bouche P, Cornblath DR, Hahn A, Illa I, et al. European Federation of Neurological Societies/Peripheral Nerve Society Guideline on management of chronic inflammatory demyelinating polyradiculoneuropathy: report of a joint task force of the European Federation of Neurological Societies and the Peripher. Eur J Neurol. 2010;17:356–63. https://doi.org/10.1111/j.1468-1331.2009.02930.x.
Magda P, Latov N, Brannagan TH, Weimer LH, Chin RL, Sander HW. Comparison of electrodiagnostic abnormalities and criteria in a cohort of patients with chronic inflammatory demyelinating polyneuropathy. Arch Neurol. 2003;60:1755. https://doi.org/10.1001/archneur.60.12.1755.
Al-Shekhlee A, Hachwi RN, Preston DC, Katirji B. New criteria for early electrodiagnosis of acute inflammatory demyelinating polyneuropathy. Muscle Nerve. 2005;32:66–72. https://doi.org/10.1002/mus.20342.
Yiannikas C, Vucic S. Utility of somatosensory evoked potentials in chronic acquired demyelinating neuropathy. Muscle Nerve. 2008;38:1447–54. https://doi.org/10.1002/mus.21078.
Devic P, Petiot P, Mauguiere F. Diagnostic utility of somatosensory evoked potentials in chronic polyradiculopathy without electrodiagnostic signs of peripheral demyelination. Muscle Nerve. 2016;53:78–83. https://doi.org/10.1002/mus.24693.
Kouton L, Boucraut J, Devaux J, Rajabally YA, Adams D, Antoine JC, et al. Electrophysiological features of chronic inflammatory demyelinating polyradiculoneuropathy associated with IgG4 antibodies targeting neurofascin 155 or contactin 1 glycoproteins. Clin Neurophysiol. 2020;131:921–7. https://doi.org/10.1016/j.clinph.2020.01.013 Largest cohort of patients with neurofascin-155 antibodies describing electrophysiologic findings.
Barohn RJ. Chronic Inflammatory Demyelinating Polyradiculoneuropathy. Arch Neurol. 1989;46:878. https://doi.org/10.1001/archneur.1989.00520440064022.
Di Pasquale A, Morino S, Loreti S, Bucci E, Vanacore N, Antonini G. Peripheral nerve ultrasound changes in CIDP and correlations with nerve conduction velocity. Neurology. 2015;84:803–9. https://doi.org/10.1212/WNL.0000000000001291.
Scheidl E, Böhm J, Simó M, Rózsa C, Bereznai B, Kovács T, et al. Ultrasonography of MADSAM neuropathy: focal nerve enlargements at sites of existing and resolved conduction blocks. Neuromuscul Disord. 2012;22:627–31. https://doi.org/10.1016/j.nmd.2012.03.005.
Goedee HS, Herraets IJT, Visser LH, Franssen H, Asseldonk JH, Pol WL, et al. Nerve ultrasound can identify treatment-responsive chronic neuropathies without electrodiagnostic features of demyelination. Muscle Nerve. 2019;60:415–9. https://doi.org/10.1002/mus.26629.
Herraets IJT, Goedee HS, Telleman JA, van Eijk RPA, van Asseldonk JT, Visser LH, et al. Nerve ultrasound improves detection of treatment-responsive chronic inflammatory neuropathies. Neurology. 2020;94:e1470–9. https://doi.org/10.1212/WNL.0000000000008978 Important paper that demonstrates the role of ultrasonography in the evaluation of inflammatory neuropathies.
Herraets IJT, Goedee HS, Telleman JA, van Eijk RPA, Verhamme C, Saris CGJ, et al. Nerve ultrasound for diagnosing chronic inflammatory neuropathy. Neurology. 2020;95:e1745–53. https://doi.org/10.1212/WNL.0000000000010369.
Goedee HS, van der Pol WL, van Asseldonk J-TH, Franssen H, Notermans NC, Vrancken AJFE, et al. Diagnostic value of sonography in treatment-naive chronic inflammatory neuropathies. Neurology. 2017;88:143–51. https://doi.org/10.1212/WNL.0000000000003483.
Garg N, Park SB, Yiannikas C, Vucic S, Howells J, Noto Y-I, et al. Neurofascin-155 IGG4 Neuropathy: pathophysiological insights, spectrum of clinical severity and response to treatment. Muscle Nerve. 2018;57:848–51. https://doi.org/10.1002/mus.26010.
Lozeron P, Lacour M-C, Vandendries C, Théaudin M, Cauquil C, Denier C, et al. Contribution of plexus MRI in the diagnosis of atypical chronic inflammatory demyelinating polyneuropathies. J Neurol Sci. 2016;360:170–5. https://doi.org/10.1016/j.jns.2015.11.048.
Hughes RA, Donofrio P, Bril V, Dalakas MC, Deng C, Hanna K, et al. Intravenous immune globulin (10% caprylate-chromatography purified) for the treatment of chronic inflammatory demyelinating polyradiculoneuropathy (ICE study): a randomised placebo-controlled trial. Lancet Neurol. 2008;7:136–44. https://doi.org/10.1016/S1474-4422(07)70329-0.
Rajabally YA, Afzal S. Clinical and economic comparison of an individualised immunoglobulin protocol vs. standard dosing for chronic inflammatory demyelinating polyneuropathy. J Neurol. 2019;266:461–7. https://doi.org/10.1007/s00415-018-9157-4.
van Schaik IN, Bril V, van Geloven N, Hartung H-P, Lewis RA, Sobue G, et al. Subcutaneous immunoglobulin for maintenance treatment in chronic inflammatory demyelinating polyneuropathy (PATH): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Neurol. 2018;17:35–46. https://doi.org/10.1016/S1474-4422(17)30378-2.
Mahdi-Rogers M, Brassington R, Gunn AA, van Doorn PA, Hughes RA. Immunomodulatory treatment other than corticosteroids, immunoglobulin and plasma exchange for chronic inflammatory demyelinating polyradiculoneuropathy. Cochrane Database Syst Rev. 2017;5:CD003280. https://doi.org/10.1002/14651858.CD003280.pub5.
Querol L, Rojas-García R, Diaz-Manera J, Barcena J, Pardo J, Ortega-Moreno A, et al. Rituximab in treatment-resistant CIDP with antibodies against paranodal proteins. Neurol - Neuroimmunol Neuroinflammation. 2015;2:e149. https://doi.org/10.1212/NXI.0000000000000149.
Ng JKM, Malotka J, Kawakami N, Derfuss T, Khademi M, Olsson T, et al. Neurofascin as a target for autoantibodies in peripheral neuropathies. Neurology. 2012;79:2241–8. https://doi.org/10.1212/WNL.0b013e31827689ad.
Kadoya M, Kaida K, Koike H, Takazaki H, Ogata H, Moriguchi K, et al. IgG4 anti-neurofascin155 antibodies in chronic inflammatory demyelinating polyradiculoneuropathy: clinical significance and diagnostic utility of a conventional assay. J Neuroimmunol. 2016;301:16–22. https://doi.org/10.1016/j.jneuroim.2016.10.013.
Miura Y, Devaux JJ, Fukami Y, Manso C, Belghazi M, Wong AHY, et al. Contactin 1 IgG4 associates to chronic inflammatory demyelinating polyneuropathy with sensory ataxia. Brain. 2015;138:1484–91. https://doi.org/10.1093/brain/awv054.
Doppler K, Stengel H, Appeltshauser L, Grosskreutz J, Man Ng JK, Meinl E, et al. Neurofascin-155 IgM autoantibodies in patients with inflammatory neuropathies. J Neurol Neurosurg Psychiatry. 2018;89:1145–51. https://doi.org/10.1136/jnnp-2018-318170 First and only report of IgM neurofascin-155 antibodies and their potential role in immune-mediated neuropathies.
Huijbers MG, Querol LA, Niks EH, Plomp JJ, van der Maarel SM, Graus F, et al. The expanding field of IgG4-mediated neurological autoimmune disorders. Eur J Neurol. 2015;22:1151–61. https://doi.org/10.1111/ene.12758.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Neuromuscular Disorders
Rights and permissions
About this article
Cite this article
Vizcarra, J.A., Harrison, T.B. & Garcia-Santibanez, R. Update on Nodopathies of the Peripheral Nerve. Curr Treat Options Neurol 23, 25 (2021). https://doi.org/10.1007/s11940-021-00683-3
Accepted:
Published:
DOI: https://doi.org/10.1007/s11940-021-00683-3