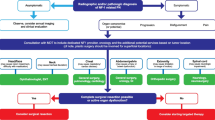
Opinion statement
Neurofibromatosis type 1 (NF1) is a genetic multisystemic disorder involving the skin, the central and peripheral nervous systems, bones, and the cardiovascular and endocrine systems. This condition is caused by inherited or de novo mutations of the NF1 gene at the 17q11.2 chromosomal region, a gene that codes for the protein neurofibromin. Neurofibromin is particularly expressed in neural cells and inhibits the RAS pathway, which regulates cellular proliferation and differentiation. The typically age-dependent emergence of diagnostic signs and the risk for severe complications in the first years of life simultaneously makes a precocious diagnosis crucial and makes the management of children with suspected NF1 challenging. Currently, no standardized specific treatments for NF1 and its complications are available. However, in recent years, increasing knowledge of the pathogenetic mechanisms has motivated the scientific search behind targeted biological agents that might change the course of the disease. Numerous clinical trials for the treatment of the most typical NF1 complications, such as plexiform neurofibromas (Ns) and NF1-related tumors, have been conducted. Consequently, encouraging in vitro and in vivo results are emerging. Insufficient efficacy and safety in in vivo data do not permit the routine use of these drugs in clinical practice. Radiotherapy appears to be indicated only for high-grade soft-tissue sarcomas, whereas surgical approaches should be considered for malignant peripheral nerve sheath tumors (MPNSTs) and Ns, optic pathway gliomas, and bone dystrophic changes because they might improve quality of life. Further prospective studies, however, are needed to confirm the efficacy, safety, and cost/benefit ratio of new therapeutic approaches and the optimal timing for their use in patients with NF1.
Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Jett K, Friedman MJ. Clinical and genetic aspects of neurofibromatosis 1. Genet Med. 2010;12:1–11.
National Institutes of Health Consensus Development Conference Statement: Neurofibromatosis. Arch Neurol (Chicago). 1988;45:575–8.
DeBella K, Szudek J, Friedman JM. Use of the national institutes of health criteria for diagnosis of neurofibromatosis 1 in children. Pediatrics. 2000;105:608–14.
Stoll C. Difficulties in the diagnosis of neurofibomatosis-1 in children. Am J Med Genet. 2002;112:422–6.
Tadini G, Milani D, Menni F, et al. Is it time to change the neurofibromatosis type 1 diagnostic criteria? Eur J Intern Med. 2014;25:506–10. An interesting paper that provides emerging criteria useful for a precocious diagnosis of NF1 in paediatric age.
Trovó-Marqui AB, Tajara EH. Neurofibromin: a general outlook. Clin Genet. 2006;70:1–13.
Sabbagh A, Pasmant E, Imbard A, Luscan A, Soares M, Blanché H, et al. NF1 molecular characterization and neurofibromatosis type I genotype–phenotype correlation: the French experience. Hum Mutat. 2013;34:1510–8.
Riccardi VM. Diagnostic and management considerations posed by multiple café au lait spots. Arch Dermatol. 2009;145:8.
Cambiaghi S, Restano L, Caputo R. Juvenile xanthogranuloma associated with neurofibromatosis 1: 14 patients without evidence of hematologic malignancies. Pediatr Dermatol. 2004;21:97–101.
Fenot M, Stalder JF, Barbarot S. Juvenile xanthogranulomas are highly prevalent but transient in young children with neurofibromatosis type 1. J Am Acad Dermatol. 2014;71:389–90.
Marque M, Roubertie A, Jaussent A, et al. Nevus anemicus in neurofibromatosis type 1: a potential new diagnostic criterion. J Am Acad Dermatol. 2013;69:768–75.
Zeller J, Wechsler J, Revuz J, et al. Blue-red macules and pseudoatrophic macules in neurofibromatosis 1. Ann Dermatol Venereol. 2002;129:180–1.
Tadini G, Brena M, Pezzani L, et al. Anemic nevus in neurofibromatosis type 1. Dermatology. 2013;226:115–8.
Rosser T, Packer RJ. Neurofibromas in children with neurofibromatosis 1. J Child Neurol. 2002;17:585.
Boyd KP, Korf BR, Theos A. Neurofibromatosis type 1. J Am Acad Dermatol. 2009;61:1.
Overdiek A, Winner U, Mayatepek E, et al. Schwann cells from human neurofibromas show increased proliferation rates under the influence of progesterone. Pediatr Res. 2008;64:40–3.
Tonsgard JH, Kwak SM, Short MP, et al. CT imaging in adults with neurofibromatosis-1: frequent asymptomatic plexiform lesions. Neurology. 1998;50:1755.
Evans DG, Baser ME, McGaughran J, et al. Malignant peripheral nerve sheath tumours in neurofibromatosis 1. J Med Genet. 2002;39:311.
Ferner RE, Gutmann DH. International consensus statement on malignant peripheral nerve sheath tumors in neurofibromatosis. Cancer Res. 2002;62:1573–7.
Listernick R, Louis DN, Packer RJ, et al. Optic pathway gliomas in children with neurofibromatosis 1: consensus statement from the NF1 optic pathway glioma task force. Ann Neurol. 1997;41:143–9.
Listernick R, Charrow J, Greenwald MJ, et al. Natural history of optic pathway tumors in children with neurofibromatosis type 1: a longitudinal study. J Pediatr. 1994;125:63–6.
Perilongo G, Moras P, Carollo C, et al. Spontaneous partial regression of low-grade glioma in children with neurofibromatosis 1: a real possibility. J Child Neurol. 1999;14:352–6.
Sharif S, Upadhyaya M, Ferner R, et al. A molecular analysis of individuals with neurofibromatosis type 1 (NF1) and optic pathway gliomas (OPGs), and an assessment of genotype-phenotype correlations. J Med Genet. 2011;48:256–60.
Fisher MJ, Loguidice M, Gutmann DH, et al. Visual outcomes in children with neurofibromatosis type 1-associated optic pathway glioma following chemotherapy: a multicenter retrospective analysis. Neuro Oncol. 2012;14:790–7.
Habiby R, Silverman B, Listernick R, et al. Precocious puberty in children with neurofibromatosis type 1. J Pediatr. 1995;126:364–7.
Korones DN, Padowski J, Factor BA, et al. Do children with optic pathway tumors have an increased frequency of other central nervous system tumors? Neuro Oncol. 2003;5:116–20.
Avery RA, Hardy KK. Vision specific quality of life in children with optic pathway gliomas. J Neurooncol. 2014;116:341–7.
Ferner RE, Huson SM, Thomas N, et al. Guidelines for the diagnosis and management of individuals with neurofibromatosis 1. J Med Genet. 2007;44:81–8.
Avery RA, Ferner RE, Listernick R, et al. Visual acuity in children with low grade gliomas of the visual pathway: implications for patient care and clinical research. J Neurooncol. 2012;110:1–7.
Gutmann DH, Parada LF, Silva AJ, et al. Neurofibromatosis type 1: modeling CNS dysfunction. J Neurosci. 2012;32:14087–93.
Margariti PN, Blekas K, Katzioti FG, et al. Magnetization transfer ratio and volumetric analysis of the brain in macrocephalic patients with neurofibromatosis type 1. Eur Radiol. 2007;17:433–8.
Steen RG, Taylor JS, Langston JW, et al. Prospective evaluation of the brain in asymptomatic children with neurofibromatosis type 1: relationship of macrocephaly to T1 relaxation changes and structural brain abnormalities. Am J Neuroradiol. 2001;22:810–7.
Ostendorf AP, Gutmann DH, Weisenberg JLZ. Epilepsy in individuals with neurofibromatosis type 1. Epilepsia. 2013;54:1810–4.
Hsieh HY, Fung HC, Wang CJ, et al. Epileptic seizures in neurofibromatosis type 1 are related to intracranial tumors but not to neurofibromatosis bright objects. Seizure. 2011;20:606–11.
Hervey-Jumper SL, Singla N, Gebarski SS, et al. Diffuse pontine lesions in children with neurofibromatosis type 1: making a case for unidentified bright objects. Pediatr Neurosurg. 2013;49:55–9.
Hyman SL, Shores EA, North KN. The nature and frequency of cognitive deficits in children with neurofibromatosis type 1. Neurology. 2005;65:1037–44.
Lehtonen A, Howi E, Trump D, et al. Behaviour in children with neurofibromatosis type 1: cognition, executive function, attention, emotion, and social competence. Dev Med Child Neurol. 2013;55:111–25.
Dilts CV, Carey JC, Kircher JC, et al. Children and adolescents with neurofibromatosis 1: a behavioral phenotype. J Dev Behav Pediatr. 1996;17:229–39.
Krab LC, Aarsen FK, de Goede-Bolder A, et al. Impact of neurofibromatosis type 1 on school performance. J Child Neurol. 2008;23:1002–10.
Garg S, Green J, Leadbitter K, et al. Neurofibromatosis type 1 and autism spectrum disorder. Pediatrics. 2013;132:1642–8.
Arrington DK, Danehy AR, Peleggi A, et al. Calvarial defects and skeletal dysplasia in patients with neurofibromatosis type 1. J Neurosurg Pediatr. 2013;11:410–6.
Riccardi VM. A controlled multiphase trial of ketotifen to minimize neurofibroma-associated pain and itching. Arch Dermatol. 1993;129:577–81.
Jakacki RI, Dombi E, Potter DM, et al. Phase I trial of pegylated interferon-alpha-2b in young patients with plexiform neurofibromas. Neurology. 2011;76:265–72.
Kebudi R, Cakir FB, Gorgun O. Interferon-α for unresectable progressive and symptomatic plexiform neurofibromas. J Pediatr Hematol Oncol. 2013;35:e115–7.
Packer RJ, Gutmann DH, Rubenstein A, et al. Plexiform neurofibromas in NF1: toward biologic-based therapy. Neurology. 2002;58:1461–70.
Gupta A, Cohen B, Ruggierri P, et al. A phase I study of thalidomide for the treatment of plexiform neurofibroma in patients with neurofibromatosis 1 (NF1). Neurology. 2000;54:12–3.
Widemann BC, Salzer WL, Arceci RJ, et al. Phase I trial and pharmacokinetic study of the arnesyltransferase inhibitor tipifarnib in children with refractory solid tumors or neurofibromatosis type I and plexiform neurofibromas. J Clin Oncol. 2006;24:507–16.
Widemann BC, Dombi E, Gillespie A, et al. Phase 2 randomized, flexible crossover, double-blinded, placebo-controlled trial of the farnesyltransferase inhibitor tipifarnib in children and young adults with neurofibromatosis type 1 and progressive plexiform neurofibromas. Neuro Oncol. 2014;16:707–18. A study showing that tipifarnib was well tolerated but did not significantly prolong time of tumor progression of plexiform Ns compared with placebo.
Demestre M, Herzberg J, Holtkamp N, et al. Imatinib mesylate (Glivec) inhibits Schwann cell viability and reduces the size of human plexiform neurofibroma in a xenograft model. J Neurooncol. 2010;98:11–9.
Robertson KA, Nalepa G, Yang FC, et al. Imatinib mesylate for plexiform neurofibromas in patients with neurofibromatosis type 1: a phase 2 trial. Lancet Oncol. 2012;13:1218–24. This trial shows the possible role of imatinib mesylate in the treatment of clinically significant plexiform Ns in children and adults with NF1; it is significant for understanding the importance of a targeted therapy based on pathogenetical mechanisms.
Wie J, Freytag M, Schobe Y, et al. Nilotinib is more potent than imatinib for treating plexiform neurofibroma in vitro and in vivo. PLoS ONE. 2014;9:e107760. An interesting study suggesting that nilotinib may be more potent than imatinib for treating plexiform Ns and may also be better tolerated.
Jiang W, Schnabel C, Spyra M, et al. Efficacy and selectivity of nilotinib on NF1-associated tumors in vitro. J Neurooncol. 2014;116:231–6.
Widemann BC, Babovic-Vuksanovic D, Dombi E, et al. Phase II trial of pirfenidone in children and young adults with neurofibromatosis type 1 and progressive plexiform neurofibromas. Pediatr Blood Cancer. 2014;61:1598–602.
Weiss B, Widemann BC, Wolters P, et al. Sirolimus for non-progressive NF1-associated plexiform neurofibromas: an NF clinical trials consortium phase II study. Pediatr Blood Cancer. 2014;61:982–6.
Hua C, Zehou O, Ducassou S, et al. Sirolimus improves pain in NF1 patients with severe plexiform neurofibromas. Pediatrics. 2014;133:e1792–7. A study showing that sirolimus for inoperable symptomatic plexiform Ns in patients with NF1 permitted stabilization of mass and produced unpredictable and important alleviation of pain in all cases with good tolerance.
Aerang K, Dombi E, Tepas K, et al. Phase I trial and pharmacokinetic study of sorafenib in children with neurofibromatosis type I and plexiform neurofibromas. Pediatr Blood Cancer. 2013;60:396–401.
Wu J, Dombi E, Jousma E, et al. Preclinical testing of sorafenib and RAD001 in the Nf(flox/flox); DhhCre mouse model of plexiform neurofibroma using magnetic resonance imaging. Pediatr Blood Cancer. 2012;58:173–80.
Kinebuchi Y, Noguchi W, Igawa Y, et al. Recurrent retroperitoneal malignant nerve sheath tumor associated with neurofibromatosis type 1 responding to carboplatin and etoposide combined chemotherapy. Int J Clin Oncol. 2005;10:353–6.
Steins MB, Serve H, Zuhlsdorf M, et al. Carboplatin/etoposide induces remission of metastasised malignant peripheral nerve tumours (malignant schwannoma) refractory to first-line therapy. Oncol Rep. 2002;9:627–30.
Brosius SN, Turk AN, Byer SJ, et al. Combinatorial therapy with tamoxifen and trifluoperazine effectively inhibits malignant peripheral nerve sheath tumor growth by targeting complementary signaling cascades. J Neuropathol Exp Neurol. 2014;73:1078–90.
Reena PT, Gibbs IC, Xu LW, et al. Treatment options for optic pathway gliomas. Curr Treat Options Neurol. 2015;17:2.
Torres KE, Liu J, Young E, et al. Expression of ‘drugable’ tyrosine kinase receptors in malignant peripheral nerve sheath tumour: potential molecular therapeutic targets for a chemoresistant cancer. Histopathology. 2011;59:143–61.
Stricker TP, Henriksen KJ, Tonsgard JH, et al. Expression profiling of 519 kinase genes in matched malignant peripheral nerve sheath tumor/plexiform neurofibroma samples is discriminatory and identifies mitotic regulators BUB1B, PBK and NEK2 as overexpressed with transformation. Mod Pathol. 2013;26:930–43.
Kaul A, Toonen JA, Cimino PJ, et al. Akt- or MEK-mediated mTOR inhibition suppresses Nf1 optic glioma growth. Neuro Oncol. 2014. This paper discusses possible biological targets for specific therapy of NF1-related OPGs.
Chabernaud C, Mennes M, Kardel PG, et al. Lovastatin regulates brain spontaneous low-frequency brain activity in neurofibromatosis type 1. Neurosci Lett. 2012;515:28–33.
Mainberger F, Nikolai HJ, Zenker M, et al. Lovastatin improves impaired synaptic plasticity and phasic alertness in patients with neurofibromatosis type 1. BMC Neurol. 2013;13:131.
van der Vaart T, Plasschaert E, Rietman AB, et al. Simvastatin for cognitive deficits and behavioural problems in patients with neurofibromatosis type 1 (NF1-SIMCODA): a randomised, placebo-controlled trial. Lancet Neurol. 2013;12:1076–83.
Créange A, Zeller J, Rostaing-Rigattieri S, et al. Neurological complications of neurofibromatosis type 1 in adulthood. Brain. 1999;122:473–81.
Carotenuto M, Esposito M. Nutraceuticals safety and efficacy in migraine without aura in a population of children affected by neurofibromatosis type 1. Neurol Sci. 2013;34:1905–9.
Widemann BC. Current status of sporadic and neurofibromatosis type 1-associated malignant peripheral nerve sheath tumors. Curr Oncol Rep. 2009;11:322–8.
Carli M, Ferrari A, Mattke A, et al. Pediatric malignant peripheral nerve sheath tumor: the Italian and German soft tissue sarcoma cooperative group. J Clin Oncol. 2005;23:8422–30.
Kawabata S, Watanabe K, Hosogane N, et al. Surgical correction of severe cervical kyphosis in patients with neurofibromatosis type 1. J Neurosurg Spine. 2013;8:274–9.
Stoker GE, Lenke LG, Dorward IG. Posterior vertebral column resection for the treatment of dystrophic kyphosis associated with type-1 neurofibromatosis. Spine. 2012;37:E1659–64.
Compliance with Ethics Guidelines
Conflict of Interest
Caterina Sabatini, Donatella Milani, Francesca Menni, Gianluca Tadini, and Susanna Esposito declare that they have no conflicts of interest.
Human and Animal Rights and Informed Consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation and with the Helsinki Declaration of 1975, as subsequently revised.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Pediatric Neurology
Rights and permissions
About this article
Cite this article
Sabatini, C., Milani, D., Menni, F. et al. Treatment of Neurofibromatosis Type 1. Curr Treat Options Neurol 17, 26 (2015). https://doi.org/10.1007/s11940-015-0355-4
Published:
DOI: https://doi.org/10.1007/s11940-015-0355-4