Abstract
Purpose of review
The goal of this review is to provide the reader with a summary of the percutaneous mechanical circulatory support (MCS) devices currently used in clinical practice.
Recent findings
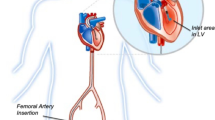
Although there are limited data for MCS including intra-aortic balloon pump (IABP), percutaneous left ventricular (LV) micro-axial pump (Impella), percutaneous left ventricular (LV) centrifugal pump (TandemHeart), VA-extracorporeal membrane oxygenation (VA-ECMO), and right ventricular (RV) support (Impella RP, Protek Duo), these devices are frequently employed in clinical practice.
Summary
Guidelines largely support the use of MCS in refractory ischemic and non-ischemic cardiogenic shock (CS). However, the literature to support MCS use is limited, especially in the setting of high-risk PCI. Ongoing randomized clinical trials of these devices will help to provide supportive data for MCS utilization in various clinical settings.
Opinion statement
Percutaneous mechanical circulatory support (MCS) has become an important adjunctive therapy in clinical practice for a variety of indications, including cardiogenic shock secondary to acute myocardial infarction (AMI-CS), cardiogenic shock secondary to acute decompensated heart failure (ADHF-CS), a bridge to advanced therapies, left ventricular (LV) unloading, high-risk PCI, and extracorporeal cardiopulmonary resuscitation (eCPR). Although there is a lack of robust data for many of these devices, MCS is recommended in an algorithmic approach in the modern era. Intra-aortic balloon pump (IABP) use is mostly limited to patients with mechanical complications of AMI, persistent ischemia in the setting of un-revascularized disease, early CS or as a bridge to durable support in non-AMI CS, or in refractory CS if other MCS devices are contraindicated. IABP support is often escalated to Impella or VA-extracorporeal membrane oxygenation (VA-ECMO) in advanced stages of CS. In cases of prolonged support as a bridge to durable treatment, Impella 5.0 or 5.5 should be considered. For high-risk PCI, Impella CP is frequently used as supportive therapy although more data are needed to clearly define its role for this indication. Impella RP and Protek Duo are commonly used devices to support the right ventricle (RV). VA-ECMO is frequently used for severe biventricular shock, but “Bi-Pella” (Impella CP/5.0/5.5 + Impella RP) remains an option when VA-ECMO is not feasible. Future randomized data will help to delineate the role for MCS devices in various clinical scenarios.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as:
• Of importance
Atti V, Narayanan MA, Patel B, Balla S, Siddique A, Lundgren S, et al. A Comprehensive review of mechanical circulatory support devices. Heart Int. 2022;16(1):37–48.
Naidu SS, Baran DA, Jentzer JC, Hollenberg SM, van Diepen S, Basir MB, et al. SCAI SHOCK stage classification expert consensus update: a review and incorporation of validation studies: This statement was endorsed by the American College of Cardiology (ACC), American College of Emergency Physicians (ACEP), American Heart Association (AHA), European Society of Cardiology (ESC) Association for Acute Cardiovascular Care (ACVC), International Society for Heart and Lung Transplantation (ISHLT), Society of Critical Care Medicine (SCCM), and Society of Thoracic Surgeons (STS) in December 2021. J Am Coll Cardiol. 2022;79(9):933–46.
Zein R, Patel C, Mercado-Alamo A, Schreiber T, Kaki A. A review of the Impella devices. Interv Cardiol. 2022;8(17):e05.
Salter BS, Gross CR, Weiner MM, Dukkipati SR, Serrao GW, Moss N, et al. Temporary mechanical circulatory support devices: practical considerations for all stakeholders. Nat Rev Cardiol. 2022;10:1–15.
Thiele H, Zeymer U, Neumann FJ, Ferenc M, Olbrich HG, Hausleiter J, et al. Intraaortic balloon support for myocardial infarction with cardiogenic shock. N Engl J Med. 2012;367(14):1287–96.
Thiele H, Zeymer U, Thelemann N, Neumann FJ, Hausleiter J, Abdel-Wahab M, et al. Intraaortic balloon pump in cardiogenic shock complicating acute myocardial infarction: long-term 6-year outcome of the randomized IABP-SHOCK II trial. Circulation. 2019;139(3):395–403.
Ahmad Y, Sen S, Shun-Shin MJ, Ouyang J, Finegold JA, Al-Lamee RK, et al. Intra-aortic balloon pump therapy for acute myocardial infarction: a meta-analysis. JAMA Intern Med. 2015;175(6):931–9.
Unverzagt S, Buerke M, Waha A, Haerting J, Pietzner D, Seyfarth M, et al. Intra-aortic balloon pump counterpulsation (IABP) for myocardial infarction complicated by cardiogenic shock. Cochrane Database Syst Rev. 2015;2015(3):CD007398.
Neumann FJ, Sousa-Uva M, Ahlsson A, Alfonso F, Banning AP, Benedetto U, et al. 2018 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J. 2019;40(2):87–165.
2013 ACCF/AHA Guideline for the management of ST-elevation myocardial infarction.
Kimman JR, Van Mieghem NM, Endeman H, Brugts JJ, Constantinescu AA, Manintveld OC, et al. Mechanical support in early cardiogenic shock: what is the role of intra-aortic balloon counterpulsation? Curr Heart Fail Rep. 2020;17(5):247–60.
Patel MR, Smalling RW, Thiele H, Barnhart HX, Zhou Y, Chandra P, et al. Intra-aortic balloon counterpulsation and infarct size in patients with acute anterior myocardial infarction without shock: the CRISP AMI randomized trial. JAMA. 2011;306(12):1329–37.
Perera D, Stables R, Thomas M, Booth J, Pitt M, Blackman D, et al. Elective intra-aortic balloon counterpulsation during high-risk percutaneous coronary intervention: a randomized controlled trial. JAMA. 2010;304(8):867–74.
Malick W, Fried JA, Masoumi A, Nair A, Zuver A, Huang A, et al. Comparison of the hemodynamic response to intra-aortic balloon counterpulsation in patients with cardiogenic shock resulting from acute myocardial infarction versus acute decompensated heart failure. Am J Cardiol. 2019;124(12):1947–53.
Jones TL, Nakamura K, McCabe JM. Cardiogenic shock: evolving definitions and future directions in management. Open Heart. 2019;6(1):e000960.
den Uil CA, Van Mieghem NM, Bastos MB, Jewbali LS, Lenzen MJ, Engstrom AE, et al. Primary intra-aortic balloon support versus inotropes for decompensated heart failure and low output: a randomised trial. EuroIntervention. 2019;15(7):586–93.
Gjesdal O, Gude E, Arora S, Leivestad T, Andreassen AK, Gullestad L, et al. Intra-aortic balloon counterpulsation as a bridge to heart transplantation does not impair long-term survival. Eur J Heart Fail. 2009;11(7):709–14.
Tanaka A, Tuladhar SM, Onsager D, Asfaw Z, Ota T, Juricek C, et al. The subclavian intraaortic balloon pump: a compelling bridge device for advanced heart failure. Ann Thorac Surg. 2015;100(6):2151–7; discussion 2157-2158.
Mandawat A, Rao SV. Percutaneous mechanical circulatory support devices in cardiogenic shock. Circ Cardiovasc Interv. 2017;10(5):e004337.
Vetrovec GW, Kaki A, Dahle TG. A review of bleeding risk with Impella-supported high-risk percutaneous coronary intervention. Heart International [Internet]. 2020. [cited 2023 Jan 22]. Available from: https://www.touchcardio.com/interventional-cardiology/journal-articles/a-review-of-bleeding-risk-with-impella-supported-high-risk-percutaneous-coronary-intervention/.
Ramzy D, Soltesz EG, Silvestry SC, Hall SA, D’Alessandro DA. Improved clinical outcomes associated with the Impella 5.5 compared to the Impella 5.0 in contemporary cardiogenic shock and heart failure patients. J Heart Lung Transplant. 2022;41(4):S31.
Balthazar T, Vandenbriele C, Verbrugge FH, Den UC, Engström A, Janssens S, et al. Managing patients with short-term mechanical circulatory support. J Am Coll Cardiol. 2021;77(9):1243–56.
Dhruva SS, Ross JS, Mortazavi BJ, Hurley NC, Krumholz HM, Curtis JP, et al. Use of mechanical circulatory support devices among patients with acute myocardial infarction complicated by cardiogenic shock. JAMA Netw Open. 2021;4(2):e2037748.
Shah M, Patnaik S, Patel B, Ram P, Garg L, Agarwal M, et al. Trends in mechanical circulatory support use and hospital mortality among patients with acute myocardial infarction and non-infarction related cardiogenic shock in the United States. Clin Res Cardiol. 2018;107(4):287–303.
Amin AP, Spertus JA, Curtis JP, Desai N, Masoudi FA, Bach RG, et al. The evolving landscape of Impella use in the United States among patients undergoing percutaneous coronary intervention with mechanical circulatory support. Circulation. 2020;141(4):273–84.
Karami M, Eriksen E, Ouweneel DM, Claessen BE, Vis MM, Baan J, et al. Long-term 5-year outcome of the randomized IMPRESS in severe shock trial: percutaneous mechanical circulatory support vs. intra-aortic balloon pump in cardiogenic shock after acute myocardial infarction. Eur Heart J Acute Cardiovasc Care. 2021;10(9):1009–15.
Seyfarth M, Sibbing D, Bauer I, Fröhlich G, Bott-Flügel L, Byrne R, et al. A randomized clinical trial to evaluate the safety and efficacy of a percutaneous left ventricular assist device versus intra-aortic balloon pumping for treatment of cardiogenic shock caused by myocardial infarction. J Am Coll Cardiol. 2008;52(19):1584–8.
• Dhruva SS, Ross JS, Mortazavi BJ, Hurley NC, Krumholz HM, Curtis JP, et al. Association of use of an intravascular microaxial left ventricular assist device vs intra-aortic balloon pump with in-hospital mortality and major bleeding among patients with acute myocardial infarction complicated by cardiogenic shock. JAMA. 2020;323(8):734–45. Findings from this study suggest no differences in mortality and higher complication rate with Impella versus IABP in AMI-CS, but this study serves as a foundation for upcoming RCTs in this population including Dan-Ger Shock and RECOVER IV.
Miller PE, Bromfield SG, Ma Q, Crawford G, Whitney J, DeVries A, et al. Clinical outcomes and cost associated with an intravascular microaxial left ventricular assist device vs intra-aortic balloon pump in patients presenting with acute myocardial infarction complicated by cardiogenic shock. JAMA Intern Med. 2022;182(9):926–33.
Moustafa A, Khan MS, Saad M, Siddiqui S, Eltahawy E. Impella support versus intra-aortic balloon pump in acute myocardial infarction complicated by cardiogenic shock: a meta-analysis. Cardiovasc Revasc Med. 2022;34:25–31.
Panuccio G, Neri G, Macrì LM, Salerno N, De Rosa S, Torella D. Use of Impella device in cardiogenic shock and its clinical outcomes: a systematic review and meta-analysis. Int J Cardiol Heart Vasc. 2022;25(40):101007.
O’Neill WW, Schreiber T, Wohns DHW, Rihal C, Naidu SS, Civitello AB, et al. The current use of Impella 2.5 in acute myocardial infarction complicated by cardiogenic shock: results from the USpella registry. J Interv Cardiol. 2014;27(1):1–11.
Del Rio-Pertuz G, Benjanuwattra J, Juarez M, Mekraksakit P, Argueta-Sosa E, Ansari MM. Efficacy of mechanical circulatory support used before versus after primary percutaneous coronary intervention in patients with cardiogenic shock from ST-elevation myocardial infarction: a systematic review and meta-analysis. Cardiovasc Revasc Med. 2022;42:74–83.
Pahuja M, Johnson A, Kabir R, Bhogal S, Wermers JP, Bernardo NL, et al. Randomized trials of percutaneous microaxial flow pump devices. J Am Coll Cardiol. 2022;80(21):2028–49.
O’Neill WW, Kleiman NS, Moses J, Henriques JPS, Dixon S, Massaro J, et al. A prospective, randomized clinical trial of hemodynamic support with Impella 2.5 versus intra-aortic balloon pump in patients undergoing high-risk percutaneous coronary intervention: the PROTECT II study. Circulation. 2012;126(14):1717–27.
O’Neill WW, Anderson M, Burkhoff D, Grines CL, Kapur NK, Lansky AJ, et al. Improved outcomes in patients with severely depressed LVEF undergoing percutaneous coronary intervention with contemporary practices. Am Heart J. 2022;248:139–49.
Bhatia K, Jain V, Hendrickson MJ, Aggarwal D, Aguilar-Gallardo JS, Lopez PD, et al. Meta-analysis comparing venoarterial extracorporeal membrane oxygenation with or without Impella in patients with cardiogenic shock. Am J Cardiol. 2022;1(181):94–101.
Fiorelli F, Panoulas V. Impella as unloading strategy during VA-ECMO: systematic review and meta-analysis. Rev Cardiovasc Med. 2021;22(4):1503–11.
• Kapur NK, Alkhouli MA, DeMartini TJ, Faraz H, George ZH, Goodwin MJ, et al. Unloading the left ventricle before reperfusion in patients with anterior ST-segment–elevation myocardial infarction. Circulation. 2019;139(3):337–46. Findings from this study suggest that there is feasibility for LV unloading 30 minutes prior to reperfusion in anterior STEMI patients, and this is the foundation for the upcoming STEMI-DTU study.
Lima B, Kale P, Gonzalez-Stawinski GV, Kuiper JJ, Carey S, Hall SA. Effectiveness and safety of the Impella 5.0 as a bridge to cardiac transplantation or durable left ventricular assist device. Am J Cardiol. 2016;117(10):1622–8.
Lo N, Magnus Ohman E. Mechanical circulatory support in ST-elevation myocardial infarction. In: Watson TJ, Ong PJ, Tcheng JE, editors. Primary Angioplasty: A Practical Guide [Internet]. Singapore: Springer; 2018 [cited 2023 Jan 24]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK543576/.
Thiele H, Sick P, Boudriot E, Diederich KW, Hambrecht R, Niebauer J, et al. Randomized comparison of intra-aortic balloon support with a percutaneous left ventricular assist device in patients with revascularized acute myocardial infarction complicated by cardiogenic shock. Eur Heart J. 2005;26(13):1276–83.
Burkhoff D, Cohen H, Brunckhorst C, O’Neill WW. A randomized multicenter clinical study to evaluate the safety and efficacy of the TandemHeart percutaneous ventricular assist device versus conventional therapy with intraaortic balloon pumping for treatment of cardiogenic shock. Am Heart J. 2006;152(3):469.e1-469.e8.
Kar B, Gregoric ID, Basra SS, Idelchik GM, Loyalka P. The percutaneous ventricular assist device in severe refractory cardiogenic shock. J Am Coll Cardiol. 2011;57(6):688–96.
Bruckner BA, Jacob LP, Gregoric ID, Loyalka P, Kar B, Cohn WE, et al. Clinical experience with the TandemHeart percutaneous ventricular assist device as a bridge to cardiac transplantation. Tex Heart Inst J. 2008;35(4):447–50.
Gregoric ID, Jacob LP, La Francesca S, Bruckner BA, Cohn WE, Loyalka P, et al. The TandemHeart as a bridge to a long-term axial-flow left ventricular assist device (bridge to bridge). Tex Heart Inst J. 2008;35(2):125–9.
Acharya D, Torabi M, Borgstrom M, Rajapreyar I, Lee K, Kern K, et al. Extracorporeal membrane oxygenation in myocardial infarction complicated by cardiogenic shock: analysis of the ELSO registry. J Am Coll Cardiol. 2020;76(8):1001–2.
Vallabhajosyula S, Prasad A, Bell MR, Sandhu GS, Eleid MF, Dunlay SM, et al. Extracorporeal membrane oxygenation use in acute myocardial infarction in the United States, 2000 to 2014. Circ Heart Fail. 2019;12(12):e005929.
Benenati S, Toma M, Canale C, Vergallo R, Bona RD, Ricci D, et al. Mechanical circulatory support in patients with cardiogenic shock not secondary to cardiotomy: a network meta-analysis. Heart Fail Rev. 2022;27(3):927–34.
• Ostadal P, Rokyta R, Karasek J, Kruger A, Vondrakova D, Janotka M, et al. Extracorporeal membrane oxygenation in the therapy of cardiogenic shock: results of the ECMO-CS randomized clinical trial. Circulation. 2022;6(147):454–64. Findings from this study showed no difference in death from any cause, resuscitated circulatory arrest, and implementation of another MCS device at 30 days in patients with early initiation of ECMO compared to a conservative strategy, but this study is the first of a few ongoing trials to evaluate effectiveness of ECMO in AMI-CS (ANCHOR, ECLS-SHOCK, and EURO-SHOCK.
Yannopoulos D, Bartos J, Raveendran G, Walser E, Connett J, Murray TA, et al. Advanced reperfusion strategies for patients with out-of-hospital cardiac arrest and refractory ventricular fibrillation (ARREST): a phase 2, single centre, open-label, randomised controlled trial. The Lancet. 2020;396(10265):1807–16.
Belohlavek J, Smalcova J, Rob D, Franek O, Smid O, Pokorna M, et al. Effect of intra-arrest transport, extracorporeal cardiopulmonary resuscitation, and immediate invasive assessment and treatment on functional neurologic outcome in refractory out-of-hospital cardiac arrest: a randomized clinical trial. JAMA. 2022;327(8):737–47.
Anderson MB, Goldstein J, Milano C, Morris LD, Kormos RL, Bhama J, et al. Benefits of a novel percutaneous ventricular assist device for right heart failure: the prospective RECOVER RIGHT study of the Impella RP device. J Heart Lung Transplant. 2015;34(12):1549–60.
Anderson M, Morris DL, Tang D, Batsides G, Kirtane A, Hanson I, et al. Outcomes of patients with right ventricular failure requiring short-term hemodynamic support with the Impella RP device. J Heart Lung Transplant. 2018;37(12):1448–58.
Kapur NK, Paruchuri V, Jagannathan A, Steinberg D, Chakrabarti AK, Pinto D, et al. Mechanical circulatory support for right ventricular failure. JACC Heart Fail. 2013;1(2):127–34.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr. Luke Kim has received fellowship grant support from Abbott and Medtronic. The other authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kanduri, J., Axman, R., Lu, D.Y. et al. Mechanical Circulatory Support: When and How to Use It. Curr Treat Options Cardio Med 25, 111–126 (2023). https://doi.org/10.1007/s11936-023-00980-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11936-023-00980-x