Abstract
Purpose of Review
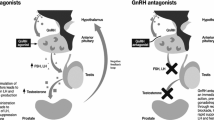
Androgen deprivation therapy (ADT) is a mainstay of treatment for advanced prostate cancer. Several studies have reported an association between ADT and an increase in cardiovascular events, especially in those receiving gonadotropin-releasing hormone (GnRH) agonists compared to GnRH antagonists. We review the body of literature reporting the association of ADT and cardiovascular morbidity, and discuss the proposed mechanism of cardiovascular disease due to ADT including metabolic changes that may promote atherosclerosis and local hormonal effects that may increase plaque rupture and thrombosis.
Recent Findings
GnRH agonists appear to increase the risk of cardiovascular morbidity by 20–25% in men on these agents compared those who do not receive ADT. GnRH antagonists may appear to have halve this risk while improving PSA progression-free survival.
Summary
GnRH antagonists may be superior to GnRH agonists for patients with significant cardiovascular disease, significant metastatic disease burden, or severe lower urinary tract symptoms.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
WHO. Globocan 2012. http://globocan.iarc.fr/Default.aspx
Grönberg H. Prostate cancer epidemiology. Lancet. 2003;361(9360):859–64.
Huggins C. Effect of orchiectomy and irradiation on cancer of the prostate. Ann Surg. 1942;115(6):1192–200.
National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in OncologyTM. Prostate cancer. National Comprehensive Cancer Network: Fort Washington, PA, Version 1. 2017. Available from: https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf. Accessed January 2017.
Mariani A, Glover M, Arita S. Medical versus surgical androgen suppression therapy for prostate cancer: a 10-year longitudinal cost study. J Urol. 2001;165(1):104–7.
Drudge-Coates L. GnRH blockers: a changing paradigm in the management of prostate cancer. Int J Urol Nurs. 2009;3:85–92.
Klotz L, Boccon–Gibod L, Shore N, Andreou C, Persson B, Cantor P, et al. The efficacy and safety of degarelix: a 12-month, comparative, randomized, open-label, parallel-group phase III study in patients with prostate cancer. BJU Int. 2008;102:1531–8.
Shore N. Experience with degarelix in the treatment of prostate cancer. Ther Adv Urol. 2013;5:11–24.
Keating N, O’Malley A, Smith M. Diabetes and cardiovascular disease during androgen deprivation therapy for prostate cancer. J Clin Oncol. 2006;23:4448–56.
Tsai H, D’Amico A, Sadetsky N, Chen M, Carrol P. Androgen deprivation therapy for localized prostate cancer and the risk of cardiovascular mortality. J Natl Cancer Inst. 2007;99:1516–24.
Saigal C, Gore J, Krupski T, Hanley J, Schonlau M, Litwin M. Androgen deprivation therapy increases cardiovascular morbidity in men with prostate cancer. Cancer. 2007;110:1493–500.
D’Amico A, Denham J, Crook J, Chen M, Goldhaber S, Lamb D, Joseph D, Tai K, Malone S, Ludgate C, Steigler A, Kantoff P. Influence of androgen suppression therapy for prostate cancer on the frequency and timing of fatal myocardial infarctions. J Clin Oncol. 2007;25:2420–5.
D’Amico A, Chen M, Renshaw A, Loffredo M, Kantoff P. Causes of death in men undergoing androgen suppression therapy for newly diagnosed localized or recurrent prostate cancer. Cancer. 2008;113:3290–7.
Levine G, D’Amico A, Berger P, Clark P, Eckel R, Keating N, Milani R, Sagalowsky A, Smith M, Zakai N. Androgen-deprivation therapy in prostate cancer and cardiovascular risk. A science advisory from the American Heart Association, American Cancer Society, and American Urologic Association: endorsed by the American Society for Radiation Oncology. Circulation. 2010;121:833–40.
Studer U, Whelan P, Albrecht W, Casselman J, de Reijke T, Hauri D, Loidl W, Isorna S, Sundaram S, Debois M, Collette L. Immediate or deferred androgen deprivation for patients with prostate cancer not suitable for local treatment with curative intent: European Organisation for Research and Treatment of Cancer (EORTC) Trial 30891. J Clin Oncol. 2006;24:1868–76.
Efstathiou J, Bae K, Shipley W, Hanks G, Pilepich M, Sandler H, Smith M. Cardiovascular mortality after androgen deprivation therapy for locally advanced prostate cancer: analysis of RTOG 92-02. Eur Urol. 2008;54:816–23.
Roach III M, Bae K, Speight J, Wolkov H, Rubin P, Lee R, Lawton C, Valicenti R, Grignon D, Pilepich M. Short-term neoadjuvant androgen deprivation therapy and external beam radiotherapy for locally advanced prostate cancer: long-term results of RTOG 8610. J Clin Oncol. 2008;26:585–91.
Efstathiou J, Bae K, Shipley W, Hanks G, Pilepich M, Sandler H, Smith M. Cardiovascular mortality after androgen deprivation therapy for locally advanced prostate cancer: RTOG 85-31. J Clin Oncol. 2009;27:92–9.
Bolla M, de Reijke T, Van Tienhoven G, Van den Bergh A, Oddens J, Poortmans P, Gez E, Kil P, et al. EORTC Radiation Oncology Group and Genito-Urinary Tract Cancer Group. Duration of androgen suppression in the treatment of prostate cancer. N Engl J Med. 2009;360:2516–27.
• Klotz L, Miller K, Crawford D, Shore N, Tombal B, Karup C, Malmberg A, Persson BE. Disease control outcomes from analysis of pooled individual patient data from five comparative randomized clinical trials of Degarelix versus lutenizing hormone-releasing hormone agonists. Eur Urol. 2014;66:1101–8. Post hoc analysis of five prospective phase 3 or 3b randomized trials comparing the efficacy of GnRH agonists against an antagonist which showed improved overall survival and adverse events with degarelix
•• Albertsen P, Klotz L, Bertrand T, Grady J, Olesen T, Nilsson J. Cardiovascular morbidity associated with gonadotropin releasing hormone agonists and an antagonist. Eur Urol. 2014;65:565–73. Post hoc analysis of six prospective randomized trials comparing the efficacy of GnRH agonists against an antagonist which found that GnRH antagonists halved the number of cardiac events experienced by men with pre-existing cardiovascular disease compared to GnRH agonists
Huhtaniemi I, White R, McArdle C, Persson B. Will GnRH antagonists improve prostate cancer treatment? Trends Endocrinol Metab. 2009;20:43–50.
Varenhorst E, Wallentin L, Carlstrom K. The effects of orchidectomy, estrogens, and cyproterone acetate on plasma testosterone, LH, and FSH concentrations in patients with carcinoma of the prostate. Scand J Urol Nephrol. 1982;16:31–6.
Millar R, Lu Z, Pawson A, Flanagan C, Morgan K, Maudsley S. Gonadotropin-releasing hormone receptors. Endocr Rev. 2004 Apr;25(2):235–75.
Harris W, Mostaghel E, Nelson P, Mongomery B. Androgen deprivation therapy: progress in understanding mechanisms of resistance and optimizing androgen depletion. Nat Clin Pract Urol. 2009;6(2):76–85.
Calais da Silva F, Bono A, Whelan P, Brausi M, et al. Intermittent androgen deprivation for locally advanced and metastatic prostate cancer: results from a randomized phase 3 study of the South European Uroncological Group. Eur Urol. 2009;55(6):1269–77.
Keating N, O’Malley A, Freeland S, Smith M. Diabetes and cardiovascular disease during androgen deprivation therapy: observational study of veterans with prostate cancer. J Natl Cancer Inst. 2010;102(1):39–46.
FDA drug safety communication: update to ongoing safety review of GnRH agonists and notification to manufacturers of GnRH agonists to add new safety information to labeling regarding increased risk of diabetes and certain cardiovascular diseases. US Food and Drug Administration Web site. http://www.fda.gov/Drugs/DrugSafety/ucm229986.htm Accessed January 2017
Van Hemelrijck M, Garmo H, Holmberg L, et al. Absolute and relative risk of cardiovascular disease in men with prostate cancer: results from the population-based PCBaSe Sweden. J Clin Oncol. 2010;28:3448–56.
Martin-Merino E, Johansson S, Morris T, Garcia RL. Androgen deprivation therapy and the risk of coronary heart disease and heart failure in patients with prostate cancer: a nested-case-control study in UK primary care. Drug Saf. 2011;34(11):1061–77.
Azoulay L, Yin H, Bernayoun S, Renoux C, Boivin J, Suissa S. Androgen-deprivation therapy and the risk of stroke in patients with prostate cancer. Eur Urol. 2011;60(6):1244–50.
Hu J, Williams S, O’Malley A, Smith M, Nguyen P, Keating N. Androgen-deprivation therapy for nonmetastatic prostate cancer is associated with an increased risk of peripheral arterial disease and venous thromboembolism. Eur Urol. 2012;61(6):1119–28.
Jespersen C, Norgaard M, Borre M. Androgen-deprivation therapy in treatment of prostate cancer and risk of myocardial infarction and stroke: a nationwide Danish population-based cohort study. Eur Urol. 2014;65(4):704–9.
• O’Farrell S, Garmo H, Holmberg L, Adolfsson J, Stattin P, Van Hemelrijck M. Risk and timing of cardiovascular disease after androgen-deprivation therapy in men with prostate cancer. J Clin Oncol. 2015;33(11):1243–51. Multivariate analysis of a cohort of 41,362 men with prostate cancer on ADT compared to age-matched prostate cancer-free men which found a 21% increased risk of cardiovascular disease in men on GnRH agonists compared with the comparison cohort. This risk was highest in the first 6 months of therapy
Alibhai S, Duong-Hua M, Sutradhar R, Fleshner N, Warde P, et al. Impact of androgen deprivation therapy on cardiovascular disease and diabetes. J Clin Oncol. 2009;27:3452–8.
Gandaglia G, Sun M, Popa I, Schiffmann J, Abdollah F, et al. The impact of the androgen deprivation therapy on the risk of coronary heart disease in patients with non-metastatic prostate cancer: a population-based study. BJU Int. 2014;114(6b):E82–9.
•• Zhao J, Zhu S, Sun L, Meng F, Zhao L, Zhao Y, Tian H, Li P, Niu Y. Androgen deprivation therapy for prostate cancer is associated with cardiovascular morbidity and mortality: a meta-analysis of population-based observational studies. PLoS One. 2014;9(9):–e107516. Meta-analysis of six population-based observational studies comparing ADT versus control which found that GnRH therapy is associated with an increased risk of cardiovascular disease and cardiovascular mortality
•• Bosco C, Bosnyak Z, Malmberg A, Adolfsson J, Keating N, Van Hemelrijck M. Quantifying observational evidence for risk of fatal and nonfatal cardiovascular disease following androgen deprivation therapy for prostate cancer: a meta-analysis. Eur Urol. 2015;68:386–96. Meta-analysis of eight observational studies comparing ADT with control which found a relative risk of 1.38 for non-fatal cardiovascular disease in me non-GnRH agonist therapy compared to men not treated with ADT
Punnen S, Cooperberg M, Sadetsky N, Carroll P. Androgen deprivation therapy and cardiovascular risk. J Clin Oncol. 2011;29(26):3510–6.
Wilcox C, Jautto A, Steigler A, Denham J. Androgen deprivation therapy for prostate cancer does not increase cardiovascular mortality in the long term. Oncology. 2012;82(1):56–8.
Nguyen P, Je Y, Schutz F, Hoffman K, Hu J, Parekh A, Beckman J, Choueiri T. Association of androgen deprivation therapy with cardiovascular death in patients with prostate cancer: a meta-analysis of randomized trials. JAMA. 2011;306(21):2359–66.
Libby P. Molecular bases of the acute coronary syndromes. Circulation. 1995;91:2844–50.
Jacobson J, Nisula B, Steinberg A. Modulation of the expression of murine lupus by gonadotropin-releasing hormone analogs. Endocrinology. 1994;134:2516–23.
Chen H, Jueng E, Stephenson M, Leung P. Ribonucleic acids that are regulated by GnRH in vitro. Endocrinol Metab. 1999;84:743–50.
Tanriverdi F, Gonzalez-Martinez D, Hu Y, Kelestimur F, Bouloux P. GnRH-I and GnRH-II have differential modulatory effects on human peripheral blood mononuclear cell proliferation and interleukin-2 receptor gamma-chain mRNA expression in healthy males. Clin Exp Immunol. 2005;142:103–10.
Grasso G, Massai L, De Leo V, Muscettola M. The effect of LHRH and TRH on human interferon-gamma production in vivo and in vitro. Life Sci. 1998;62(22):2005–14.
Andersson J, Libby P, Hansson G. Adaptive immunity and atherosclerosis. Clin Immunol. 2010;134(1):33–46.
Dixit V, Yang H, Udhayakumar V, Sridaran R. Gonadotropin-releasing hormone alters the t helper cytokine balance in the pregnant rat. Biol Reprod. 2003;68:2215–21.
Smith M, Klotz L, van der Meulen E, Colli E, Tanko L. Gonadotropin-releasing hormone blockers and cardiovascular disease risk: analysis of prospective clinical trials of degarelix. J Urol. 2011;186:1935–842.
Tombal B, Miller K, Boccon-GIbod L, et al. Additional analysis of the secondary end point of biochemical recurrence rate in a phase 3 trial (CS21) comparing degarelix 80 mg versus leuprolide in prostate cancer patients segmented by baseline characteristics. Eur Urol. 2010;57:836–42.
Crawford E, Tombal B, Miller K, et al. A phase III extension trial with a 1-arm crossover from leuprolide to degarelix: comparison of gonadotropin-releasing hormone agonist and antagonist effect on prostate cancer. J Urol. 2011;186:889–97.
• Crawford E, Shore N, Moul J, Tombal B, et al. Long-term tolerability and efficacy of degarelix: 5-year results from a phase III extension trial with a 1-arm crossover from leuprolide to degarelix. Urology. 2014;83(5):1122–8. The 5-year results from the cross-over trial of CS21 where patients receiving leuprolide were crossed over to degarelix after one year which showed a durable PSA progression-free survival benefit for degarelix over leuprolide at 5 years
Hopmans S, Duivenvoorden W, Werstuck G, Klotz L, Pinthus J. GnRH antagonist associates with less adiposity and reduced characteristics of metabolic syndrome and atherosclerosis compared with orchiectomy and GnRH agonist in a preclinical mouse model. Urol Oncol. 2014;32(8):1126–34.
Radu A, Pichon C, Camparo P, Antoine M, et al. Expression of follicle-stimulating hormone receptor in tumor blood vessels. N Engl J Med. 2010;363(17):1621–30.
Robinson D, Sandblom G, Johansson R, Garmo H, Stattin P, Mommsen S, et al. Prediction of survival of metastatic prostate cancer based on early serial measurements of prostate specific antigen and alkaline phosphatase. J Urol. 2008;179:117–22.
Schrӧder F, Tombal B, Miller K, Boccon-Gibod L, Shore N, Crawford E, et al. Changes in alkaline phosphatase levels in patients with prostate cancer receiving degarelix or leuprolide: results from a 12-month, comparative, phase III study. BJU Int. 2009;106:182–7.
Axcrona K, Aaltomaa S, Da Silva C, et al. Androgen deprivation therapy for volume reduction, lower urinary tract symptom relief and quality of life improvement in patients with prostate cancer: degarelix vs goserelin plus bicalutamide. BJU Int. 2012;110:1721–8.
Mason M, Maldonado P, Steidle C, et al. Neoadjuvant androgen deprivation therapy for prostate volume reduction, lower urinary tract symptoms relief and quality of life improvement in men with intermediate–to high-risk prostate cancer: a randomized non-inferiority trial of degarelix versus goserelin plus bicalutamide. Clin Oncol. 2013;25:190–6.
Anderson J, Al-Ali G, Wirth M, et al. Degarelix versus goserelin (+antiandrogen flare protection) in the relief of lower urinary tract symptoms secondary to prostate cancer: results from a phase IIIb study (NCT 00831233). Urol Int. 2013;90:321–8.
Bono A, Salvadore M, Celato N. Gonadotropin-releasing hormone receptors in prostate tissue. Anal Quant Cytol Histol. 2002;24:221–7.
Rick F, Schally A, Block N, et al. LHRH antagonist cetorelix reduces prostate size and gene expression of proinflammtory cytokines and growth factors in a rat model of benign prostatic hyperplasia. Prostate. 2001;71:736–47.
Giuliano F, Behr-Roussel D, Oger S, et al. Ozarelix, an LHRH antagonist, exerts a direct relaxing effect on human prostate in vitro. J Urol. 2009;181:693.
Hatoum H, Crawford E, Nielsen S, et al. Cost-effectiveness analysis comparing degarelix with leuprolide in hormonal therapy for patients with locally advanced prostate cancer. Expert Rev Pharmacoecon Outcomes Res. 2013;13:261–70.
Lee D, Porter J, Gladwell D, Brereton N, Nielsen S. A cost-utility analysis of degarelix in the treatment of advanced hormone-dependent prostate cancer in the United Kingdom. J Med Econ. 2014;17(4):233–47.
Kelly D, Jones T. Testosterone: a metabolic hormone in health and disease. J Endocrinol. 2013;217:24–45.
Corona G, Rastrelli G, Monami M, et al. Hypogonadism as a risk factor for cardiovascular mortality in men: a meta-analytic study. Eur J Endocrinol. 2011;165:687–701.
Zareba P, Duivenvoorden W, Leong D, Pinthus J. Androgen deprivation therapy and cardiovascular disease: what is the linking mechanism? Ther Adv Urol. 2016;8(2):118–29.
Smith M. Androgen deprivation therapy and risk for diabetes and cardiovascular disease in prostate cancer survivors. Curr Urol Rep. 2008;9(3):197–202.
Smith M, Finkelstein J, McGovern F, et al. Changes in body composition during androgen deprivation therapy for prostate cancer. J Clin Endocrinol Metab. 2002;87:599–603.
Bourghardt J, Wilhelmson A, Alexandeson C, De Gendt K, Verhoeven G, Krettek A, et al. Androgen receptor-dependent and independent atheroprotection by testosterone in male mice. Endocrinology. 2010;151:5428–37.
Falk E. Mophologic feature of unstable atherothrombotic plaques underlying acute coronary syndromes. Am J Cardiol. 1989;3:114–20.
Libby P. Molecular and cellular mechanisms of the thrombotic complications of atherosclerosis. J Lipid Res. 2009;50(suppl):S352–237.
Hansson G, Hermansson A. The immune system in atherosclerosis. Nat Immunol. 2011;12:204–12.
Bhatia N, Santos M, Jones L, Beckman J, Penson D, Morgans A, Moslehi J. Cardiovascular effects of androgen deprivation therapy for the treatment of prostate cancer: ABCDE steps to reduce cardiovascular disease in patients with prostate cancer. Circulation. 2016;133:537–41.
Jacobs E, Newton CC, Stevens V, Campbell P, Freedland S, Gapstur S. Daily aspirin use and prostate cancer-specific mortality in a large cohort of men with nonmetastatic prostate cancer. J Clin Oncol. 2014;32:3716–22.
Conteduca V, Di Lorenzo G, Tartarone A, Aieta M. The cardiovascular risk of gonadotropin releasing hormone agonists in men with prostate cancer: an unresolved controversy. Crit Rev Ocol Hematol. 2013;8:2–51.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Alyssa K. Greiman declares no potential conflicts of interest. Thomas E. Keane reports consultancies for Ferring, Bayer, and Jannsen.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Prostate Cancer
Rights and permissions
About this article
Cite this article
Greiman, A.K., Keane, T.E. Approach to Androgen Deprivation in the Prostate Cancer Patient with Pre-existing Cardiovascular Disease. Curr Urol Rep 18, 41 (2017). https://doi.org/10.1007/s11934-017-0688-5
Published:
DOI: https://doi.org/10.1007/s11934-017-0688-5