Abstract
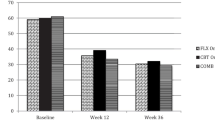
Adolescents with major depressive disorder (MDD), their families and clinicians experience significant challenges when weighing the potential risks versus benefits of available choices in the treatment of MDD. Although MDD is highly prevalent in adolescents and is associated with marked suffering, impairment and risk of suicide, the scientific data regarding the safety and efficacy of treatments for pediatric depression are limited. Controlled clinical trials have provided support for the use of psychotherapy and fluoxetine for the treatment of pediatric depression, but until recently no information on the comparative efficacy of these recommended interventions alone or in combination was available. The Treatment for Adolescents with Depression Study provides a very important therapeutic advance in the field by convincingly showing that combination treatment with cognitive behavioral therapy and fluoxetine has the best benefit to risk ratio for adolescents with moderate to severe depression, and is superior to monotherapy. Moreover, the study results confirm that fluoxetine alone is effective in the treatment of depressed adolescents.
Similar content being viewed by others
References and Recommended Reading
Birmaher B, Brent DA, Benson RS: Summary of the practice parameters for the assessment and treatment of children and adolescents with depressive disorders. J Am Acad Child Adolesc Psychiatry 1998, 37:1234–1238.
Food and Drug Administration: FDA Public Health Advisory, 2004. http://www.fda.gov/cder/drug/antidepressants/AntidepressanstPHA.htm.
Costello E, Pine DS, Hammen C, et al.: Development and natural history of mood disorders. Biol Psychiatry 2002, 52:529–542.
Gould MS, King R, Greenwald S, et al.: Psychopathology associated with suicidal ideation and attempts among children and adolescents. J Am Acad Child Adolesc Psychiatry 1998, 37:915–923.
Shaffer D, Gould MS, Fisher P, et al.: Psychiatric diagnosis in child and adolescent suicide. Arch Gen Psychiatry 1996, 53:339–348.
Brent DA, Holder D, Kolko D, et al.: A clinical psychotherapy trial for adolescent depression comparing cognitive, family, and supportive therapy. Arch Gen Psychiatry 1997, 54:877–885.
Emslie GJ, Rush AJ, Weinberg WA, et al.: A double-blind, randomized, placebo-controlled trial of fluoxetine in children and adolescents with depression. Arch Gen Psychiatry 1997, 54:1031–1037.
Lewinsohn PM, Clarke GN: Psychosocial treatments for adolescent depression. Clin Psychol Rev 1999, 19:329–342.
Harrington R, Whittaker J, Shoebridge P, Campbell F: Systematic review of efficacy of cognitive behaviour therapies in childhood and adolescent depressive disorder. BMJ 1998, 316:1559–1563.
Reinecke MA, Ryan NE, DuBois DL: Cognitive-behavioral therapy of depression and depressive symptoms during adolescence: a review and meta-analysis. J Am Acad Child Adolesc Psychiatry 1998, 37:26–34.
Emslie GJ, Heiligenstein JH, Wagner KD, et al.: Fluoxetine for acute treatment of depression in children and adolescents: a placebo-controlled, randomized clinical trial. J Am Acad Child Adolesc Psychiatry 2002, 41:1205–1215.
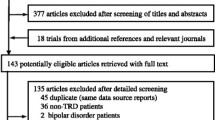
TADS: Treatment for Adolescents With Depression Study (TADS): rationale, design, and methods. J Am Acad Child Adolesc Psychiatry 2003, 42:531–542. This article describes the design of the trial, the rationale for the design choices made, and the methods used to carry out the trial.
Kaufman J, Birmaher B, Brent D, et al.: Schedule for Affective Disorders and Schizophrenia for School-Age Children-Present and Lifetime Version (K-SADS-PL): initial reliability and validity data. J Am Acad Child Adolesc Psychiatry 1997, 36:980–988.
Guy W: ECDEU Assessment Manual for Psychopharmacology. Second Edition. Vol DHEW Publication No. (ABM) 76-388 Washington, DC: U.S. Government Printing Office; 1976.
Poznanski E, Mokros H: Children’s Depression Rating Scale-Revised (CDRS-R). Los Angeles: WPS; 1995.
TADS: Fluoxetine, cognitive-behavioral therapy, and their combination for adolescents with depression: Treatment for Adolescents With Depression Study (TADS) randomized controlled trial. JAMA 2004, 292:807–820. This article presents the effectiveness results from the acute phase of TADS.
Fisher L, Dixon D, Herson J, et al.: Intention-to-treat in clinical trials. In Statistical Issues in Drug Research and Development. Peace KE. New York: Marcel Dekker; 1990.
Brown H, Prescott R: Applied Mixed Models in Medicine. New York: John Wiley and Sons; 1999.
Gueorguieva R, Krystal JH: Move over ANOVA: progress in analyzing repeated-measures data and its reflection in papers published in the Archives of General Psychiatry. Arch Gen Psychiatry 2004, 61:310–317.
Weinfert K: Repeated measures analyses: ANOVA, MANOVA, HLM. In Reading and Understanding More Multivariate Statistics. Edited by Grimm L, Yarnold P. Washington, DC: American Psychological Press; 2000:317–362.
TADS: The Treatment for Adolescents With Depression Study (TADS): demographic and clinical characteristics. In: J Am Acad Child Adolesc Psychiatry 2005, 44:28–40. This article describes the demographic and clinical characteristics of the sample and addresses external validity.
March J, Kratochvil C, Clarke G, et al.: AACAP 2002 research forum: placebo and alternatives to placebo in randomized controlled trials in pediatric psychopharmacology. J Am Acad Child Adolesc Psychiatry 2004, 43:1046–1056.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pathak, S., Kratochvil, C.J., Rogers, G.M. et al. Comparative efficacy of cognitive behavioral therapy, fluoxetine, and their combination in depressed adolescents: Initial lessons from the treatment for adolescents with depression study. Curr Psychiatry Rep 7, 429–434 (2005). https://doi.org/10.1007/s11920-005-0063-y
Issue Date:
DOI: https://doi.org/10.1007/s11920-005-0063-y