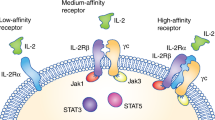
Abstract
Purpose of Review
In this review, we discuss modern cytokine delivery systems in oncologic care, focusing on modalities being developed in the clinical trials or currently in use. These include pegylation, immune-cytokine drug conjugates, cytokine-expressing plasmid nanoparticles, nonviral cytokine nanoparticles, viral systems, and AcTakines.
Recent Findings
Cytokine therapy has the potential to contribute to cancer treatment options by modulating the immune system towards an improved antitumor response and has shown promise both independently and in combination with other immunotherapy agents. Despite promising preliminary studies, systemic toxicities and challenges with administration have limited the impact of unmodified cytokine therapy. In the last decade, novel delivery systems have been developed to address these challenges and facilitate cytokine-based oncologic treatments.
Summary
Novel delivery systems provide potential solutions to decrease dose-limiting side effects, facilitate administration, and increase the therapeutic activity of cytokine treatments in oncology care. The expanding clinical and translational research in these systems provides an opportunity to augment the armamentarium of immune oncology and may represent the next frontier of cytokine-based immuno-oncology.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71(1):7–33.
The Nobel Prize in Physiology or Medicine [press release]. The Nobel Assembly at Karolinska Institutet; 2018.
Buchbinder E, Hodi FS. Cytotoxic T lymphocyte antigen-4 and immune checkpoint blockade. J Clin Invest. 2015;125(9):3377–83.
Chen L, Han X. Anti–PD-1/PD-L1 therapy of human cancer: past, present, and future. J Clin Invest. 2015;125(9):3384–91.
Sadelain M. CAR therapy: the CD19 paradigm. J Clin Invest. 2015;125(9):3392–400.
Dinarello CA. Historical insights into cytokines. Eur J Immunol. 2007;37(S1):S34–45.
Kammula US, White DE, Rosenberg SA. Trends in the safety of high dose bolus interleukin-2 administration in patients with metastatic cancer. Cancer. 1998;83(4):797–805.
Lee S, Margolin K. Cytokines in cancer immunotherapy. Cancers. 2011;3(4).
Lazarus HM, et al. Sargramostim (rhu GM-CSF) as cancer therapy (systematic review) and an immunomodulator. A drug before its time? Front Immunol. 2021. 12(3196).
Abuchowski A, Van Es T, Palczuk N, Davis F. Alteration of immunological properties of bovine serum albumin by covalent attachment of polyethylene glycol. J Biol Chem. 1977;252(11):3578–81.
Molineux G. The design and development of pegfilgrastim (PEG-rmetHuG-CSF, Neulasta®). Curr Pharm Des. 2004;10(11):1235–44.
• Mahler LJ, DiBlasi R, Perez A, Gaspard J, McCauley D. On-body injector: an administration device for pegfilgrastim. Clin J Oncol Nurs. 2017;21(1):121–2 Pegylation of cytokines allows delayed administration with decreased toxicity. The on-body injection device represents an innovative technology that facilitates convenient pegfilgrastim administration after chemotherapy administration.
Eggermont AM, Suciu S, Santinami M, Testori A, Kruit WH, Marsden J, et al. Adjuvant therapy with pegylated interferon alfa-2b versus observation alone in resected stage III melanoma: final results of EORTC 18991, a randomised phase III trial. Lancet. 2008;372(9633):117–26.
Bex A, Mallo H, Kerst M, Haanen J, Horenblas S. Gast GCd. A phase-II study of pegylated interferon alfa-2b for patients with metastatic renal cell carcinoma and removal of the primary tumor. Cancer Immunol Immunother. 2005;54(7):713–9.
• How J, Hobbs G. Use of interferon alfa in the treatment of myeloproliferative neoplasms: perspectives and review of the literature. Cancers. 2020;12(7):1954 This review outlines the evolution in interferon-alfa pegylation facilitating its use in treatment for myeloproliferative neoplasms.
Charych D, Khalili S, Dixit V, Kirk P, Chang T, Langowski J, et al. Modeling the receptor pharmacology, pharmacokinetics, and pharmacodynamics of NKTR-214, a kinetically-controlled interleukin-2 (IL2) receptor agonist for cancer immunotherapy. PLoS One. 2017;12(7):e0179431.
•• Naing A, Papadopoulos KP, Autio KA, Ott PA, Patel MR, Wong DJ, et al. Safety, antitumor activity, and immune activation of pegylated recombinant human interleukin-10 (AM0010) in patients with advanced solid tumors. J Clin Oncol. 2016;34(29):3562 Phase Ib clinical trial demonstrating the potential of cytokine therapy (pegilodecakin, a pegylated IL-10) in combination with immune checkpoint blockade (pembrolizumab or nivolumab, anti-PD-1 agents.).
Naing A, Wong DJ, Infante JR, Korn WM, Aljumaily R, Papadopoulos KP, et al. Pegilodecakin combined with pembrolizumab or nivolumab for patients with advanced solid tumours (IVY): a multicentre, multicohort, open-label, phase 1b trial. Lancet Oncol. 2019;20(11):1544–55.
Mishra P, Nayak B, Dey RK. PEGylation in anti-cancer therapy: an overview. Asian J Pharm Sci. 2016;11(3):337–48.
• Klein C, Waldhauer I, Nicolini VG, Freimoser-Grundschober A, Nayak T, Vugts DJ, et al. Cergutuzumab amunaleukin (CEA-IL2v), a CEA-targeted IL-2 variant-based immunocytokine for combination cancer immunotherapy: overcoming limitations of aldesleukin and conventional IL-2-based immunocytokines. Oncoimmunology. 2017;6(3):e1277306 This preclinical trial used immunocytokine delivery to preferentially target certain immune cell populations and tumor tissues. Combination with immune checkpoint blockade exhibited synergy in preclinical models.
Bilusic M, Heery CR, Collins JM, Donahue RN, Palena C, Madan RA, et al. Phase I trial of HuMax-IL8 (BMS-986253), an anti-IL-8 monoclonal antibody, in patients with metastatic or unresectable solid tumors. J ImmunoTher Cancer. 2019;7(1):240.
Neumann E, Rosenheck K. Permeability changes induced by electric impulses in vesicular membranes. J Membr Biol. 1972;10(3):279–90.
Titomirov AV, Sukharev S, Kistanova E. In vivo electroporation and stable transformation of skin cells of newborn mice by plasmid DNA. Biochim Biophys Acta. 1991;1088(1):131–4.
Berraondo P, Sanmamed MF, Ochoa MC, Etxeberria I, Aznar MA, Pérez-Gracia JL, et al. Cytokines in clinical cancer immunotherapy. Br J Cancer. 2019;120(1):6–15.
Kelsall BL, Stüber E, Neurath M, Strober W. Interleukin-12 production by dendritic cells. The role of CD40-CD40L interactions in Th1 T-cell responses. Ann N Y Acad Sci. 1996;795:116–26.
Ibe S, Qin Z, Schüler T, Preiss S, Blankenstein T. Tumor rejection by disturbing tumor stroma cell interactions. J Exp Med. 2001;194(11):1549–59.
Leonard JP, Sherman ML, Fisher GL, Buchanan LJ, Larsen G, Atkins MB, et al. Effects of single-dose interleukin-12 exposure on interleukin-12-associated toxicity and interferon-gamma production. Blood. 1997;90(7):2541–8.
Lucas ML, Heller L, Coppola D, Heller R. IL-12 plasmid delivery by in vivo electroporation for the successful treatment of established subcutaneous B16.F10 melanoma. Mol Ther. 2002;5(6):668–75.
• Algazi A, Bhatia S, Agarwala S, Molina M, Lewis K, Faries M, et al. Intratumoral delivery of tavokinogene telseplasmid yields systemic immune responses in metastatic melanoma patients. Ann Oncol. 2020;31(4):532–40 This plasmid encoding IL-12 was well tolerated in patients with advanced melanoma and provoked objective response in a significant number of patients. However, immune tolerance limited its impact.
Weide B, Neri D, Elia G. Intralesional treatment of metastatic melanoma: a review of therapeutic options. Cancer Immunol Immunother. 2017;66(5):647–56.
Fewell JG, Matar M, Slobodkin G, Han S-O, Rice J, Hovanes B, et al. Synthesis and application of a non-viral gene delivery system for immunogene therapy of cancer. J Control Release. 2005;109(1-3):288–98.
•• Thaker PH, Brady WE, Lankes HA, Odunsi K, Bradley WH, Moore KN, et al. A phase I trial of intraperitoneal GEN-1, an IL-12 plasmid formulated with PEG-PEI-cholesterol lipopolymer, administered with pegylated liposomal doxorubicin in patients with recurrent or persistent epithelial ovarian, fallopian tube or primary peritoneal cancers: an NRG Oncology/Gynecologic Oncology Group study. Gynecol Oncol. 2017;147(2):283–90 Phase I clinical trial combining plasmid encoding IL-12 with neoadjuvant chemotherapy, demonstrated high tolerability with no dose limiting toxicities and positive findings in the tumor micro-environment.
Wright AA, Bohlke K, Armstrong DK, Bookman MA, Cliby WA, Coleman RL, et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: Society of Gynecologic Oncology and American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2016;34(28):3460–73.
Vergote I, Tropé CG, Amant F, Kristensen GB, Ehlen T, Johnson N, et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N Engl J Med. 2010;363(10):943–53.
Thaker PH, Bradley WH, Leath CA, Gunderson Jackson C, Borys N, Anwer K, et al. GEN-1 in combination with neoadjuvant chemotherapy for patients with advanced epithelial ovarian cancer: a phase I dose-escalation study. Clin Cancer Res. 2021:clincanres.0360.2021.
Zhai YZ, Zhou Y, Ma L, Feng GH. Effects of cell-mediated immunity induced by intramuscular chitosan-pJME/ GM-CSF nano-DNA vaccine in BAlb/c mice. Bing Du Xue Bao. 2014;30(4):423–8.
Xu Q, Guo L, Gu X, Zhang B, Hu X, Zhang J, et al. Prevention of colorectal cancer liver metastasis by exploiting liver immunity via chitosan-TPP/nanoparticles formulated with IL-12. Biomaterials. 2012;33(15):3909–18.
Sabel MS, Su G, Griffith KA, Chang AE. Intratumoral delivery of encapsulated IL-12, IL-18 and TNF-alpha in a model of metastatic breast cancer. Breast Cancer Res Treat. 2010;122(2):325–36.
Suzuki R, Namai E, Oda Y, Nishiie N, Otake S, Koshima R, et al. Cancer gene therapy by IL-12 gene delivery using liposomal bubbles and tumoral ultrasound exposure. J Control Release. 2010;142(2):245–50.
Brzóska K, Grądzka I, Kruszewski M. Impact of silver, gold, and iron oxide nanoparticles on cellular response to tumor necrosis factor. Toxicol Appl Pharmacol. 2018;356:140–50.
Hara K, Shirasuna K, Usui F, Karasawa T, Mizushina Y, Kimura H, et al. Interferon-tau attenuates uptake of nanoparticles and secretion of interleukin-1β in macrophages. PLoS ONE. 2014;9(12):e113974.
Hu B, Du H-J, Yan G-P, Zhuo R-X, Wu Y, Fan C-L. Magnetic polycarbonate microspheres for tumor-targeted delivery of tumor necrosis factor. Drug Deliv. 2014;21(3):204–12.
Qiu Y, Su M, Liu L, Tang Y, Pan Y, Sun J. Clinical application of cytokines in cancer immunotherapy. Drug Des Devel Ther. 2021;15:2269–87.
Cifani N, Chronopoulou L, Pompili B, Di Martino A, Bordi F, Sennato S, et al. Improved stability and efficacy of chitosan/pDNA complexes for gene delivery. Biotechnol Lett. 2015;37(3):557–65.
Yan C, Jie L, Yongqi W, Weiming X, Juqun X, Yanbing D, et al. Delivery of human NKG2D-IL-15 fusion gene by chitosan nanoparticles to enhance antitumor immunity. Biochem Biophys Res Commun. 2015;463(3):336–43.
Tan L, Han S, Ding S, Xiao W, Ding Y, Qian L, et al. Chitosan nanoparticle-based delivery of fused NKG2D-IL-21 gene suppresses colon cancer growth in mice. Int J Nanomedicine. 2017;12:3095–107.
Park J, Wrzesinski SH, Stern E, Look M, Criscione J, Ragheb R, et al. Combination delivery of TGF-β inhibitor and IL-2 by nanoscale liposomal polymeric gels enhances tumour immunotherapy. Nat Mater. 2012;11(10):895–905.
Libutti SK, Paciotti GF, Myer L, Haynes R, Gannon W, Walker M, et al. Results of a completed phase I clinical trial of CYT-6091: a pegylated colloidal gold-TNF nanomedicine. J Clin Oncol. 2009;27(15_suppl):3586.
Powell AC, Paciotti GF, Libutti SK. Colloidal gold: a novel nanoparticle for targeted cancer therapeutics. In: Grobmyer SR, Moudgil BM, editors. Cancer nanotechnology: methods and protocols. Totowa: Humana Press; 2010. p. 375–84.
• Anselmo AC, Mitragotri S. Nanoparticles in the clinic: an update. Bioeng Transl Med. 2019;4(3):e10143-e Review of clinically relevant nanoparticle technology and future directions.
Pol JG, Workenhe ST, Konda P, Gujar S, Kroemer G. Cytokines in oncolytic virotherapy. Cytokine Growth Factor Rev. 2020;56:4–27.
Cruz FM, Colbert JD, Merino E, Kriegsman BA, Rock KL. The biology and underlying mechanisms of cross-presentation of exogenous antigens on MHC-I molecules. Annu Rev Immunol. 2017;35(1):149–76.
Pol JG, Rességuier J, Lichty BD. Oncolytic viruses: a step into cancer immunotherapy. Virus Adapt Treat. 2011;4(1):1–21.
Kaufman HL, Amatruda T, Reid T, Gonzalez R, Glaspy J, Whitman E, et al. Systemic versus local responses in melanoma patients treated with talimogene laherparepvec from a multi-institutional phase II study. J ImmunoTher Cancer. 2016;4(1).
Havunen R, Santos JM, Sorsa S, Rantapero T, Lumen D, Siurala M, et al. Abscopal effect in non-injected tumors achieved with cytokine-armed oncolytic adenovirus. Mol Ther Oncol. 2018;11:109–21.
Kuryk L, Møller A-SW, Jaderberg M. Abscopal effect when combining oncolytic adenovirus and checkpoint inhibitor in a humanized NOG mouse model of melanoma. J Med Virol. 2019;91(9):1702–6.
•• Friedman GK, Johnston JM, Bag AK, Bernstock JD, Li R, Aban I, et al. Oncolytic HSV-1 G207 immunovirotherapy for pediatric high-grade gliomas. N Engl J Med. 2021;384(17):1613–22 Modified HSV-1 oncolytic virus showed changes in the tumor micro-environment via increase tumor infiltrating lymphocytes and improved patient overall survival.
•• Gonzalez-Pastor R, Goedegebuure PS, Curiel DT. Understanding and addressing barriers to successful adenovirus-based virotherapy for ovarian cancer. Cancer Gene Ther. 2021;28(5):375–89 Review of the adenovirus oncolytic virus system, including its potential role in combination with immune checkpoint inhibition or CAR-T therapy.
Eckert EC, Nace RA, Tonne JM, Evgin L, Vile RG, Russell SJ. Generation of a tumor-specific chemokine gradient using oncolytic vesicular stomatitis virus encoding CXCL9. Mol Ther Oncol. 2020;16:63–74.
Liu Z, Ravindranathan R, Li J, Kalinski P, Guo ZS, Bartlett DL. CXCL11-armed oncolytic poxvirus elicits potent antitumor immunity and shows enhanced therapeutic efficacy. OncoImmunology. 2016;5(3):e1091554.
Francis L, Sheng Guo Z, Liu Z, Ravindranathan R, Urban JA, Sathaiah M, et al. Modulation of chemokines in the tumor microenvironment enhances oncolytic virotherapy for colorectal cancer. Oncotarget. 2016;7(16).
Pelosci A. FDA grants Fast Track Designation to ONCOS-102 for treatment of advanced melanoma: Cancer Network; 2021 [Available from: https://www.cancernetwork.com/view/fda-grants-fast-track-designation-to-oncos-102-for-treatment-of-advanced-melanoma.
Greig SL. Talimogene laherparepvec: first global approval. Drugs. 2016;76(1):147–54.
Raman SS, Hecht JR, Chan E. Talimogene laherparepvec: review of its mechanism of action and clinical efficacy and safety. Immunotherapy. 2019;11(8):705–23.
• Arend RC, Beer HM, Cohen YC, Berlin S, Birrer MJ, Campos SM, et al. Ofranergene obadenovec (VB-111) in platinum-resistant ovarian cancer; favorable response rates in a phase I/II study are associated with an immunotherapeutic effect. Gynecol Oncol. 2020;157(3):578–84 Phase I/II trial combining chemotherapy and transgene-delivering adenovirus to disrupt the tumor micro-environment in recurrent ovarian cancer.
Pujade-Lauraine E, Hilpert F, Weber B, Reuss A, Poveda A, Kristensen G, et al. Bevacizumab combined with chemotherapy for platinum-resistant recurrent ovarian cancer: the AURELIA open-label randomized phase III trial. J Clin Oncol. 2014;32(13):1302–8.
Huyghe L, Van Parys A, Cauwels A, Van Lint S, De Munter S, Bultinck J, et al. Safe eradication of large established tumors using neovasculature-targeted tumor necrosis factor-based therapies. EMBO Mol Med. 2020;12(2):e11223.
Cauwels A, Tavernier J. Tolerizing strategies for the treatment of autoimmune diseases: from ex vivo to in vivo strategies. Front Immunol. 2020;11(674).
Funding
CAL was supported in part by the UG1 CA23330 and P50 CA098252.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Jhalak Dholakia declares that she has no conflict of interest. Alexander C. Cohen declares that he has no conflict of interest. Charles A. Leath III declares that he has no conflict of interest. Elizabeth T. Evans declares that she has no conflict of interest. Ronald D. Alvarez declares that he has no conflict of interest. Premal H. Thaker has received research funding (paid to her institution) from Merck and GlaxoSmithKline; has received compensation for service as a consultant from Celsion; has received speaker’s honoraria from GlaxoSmithKline and AstraZeneca; has participated on Data Safety Monitoring or advisory boards for Iovance Biotherapeutics, Novocure, Celsion, Agenus, Eisai, GlaxoSmithKline, and Seagen; and owns stock/stock options in Celsion.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Immuno-oncology
Rights and permissions
About this article
Cite this article
Dholakia, J., Cohen, A.C., Leath, C.A. et al. Development of Delivery Systems for Local Administration of Cytokines/Cytokine Gene-Directed Therapeutics: Modern Oncologic Implications. Curr Oncol Rep 24, 389–397 (2022). https://doi.org/10.1007/s11912-022-01221-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11912-022-01221-3