Abstract
Purpose of Review
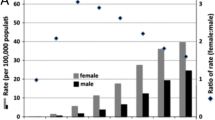
While females make up almost 60% of all brain and spinal cord tumors in adults, guidelines that address women’s issues in neuro-oncology are lacking. This review sheds light on two common women’s issues in neuro-oncology.
Recent Findings
Neuro-oncology providers are often faced with patient questions about fertility and pregnancy maintenance or prevention and typically respond with generic cancer chemotherapy recommendations, based on the paucity of evidence on the use of common neuro-oncology chemotherapies and pregnancy. While these remain important gap issues, there are several other poorly researched issues in the Neuro-Oncology of Women (N.O.W.) including recommendations around endogenous and iatrogenic hormone exposure and female sexuality in cancer.
Summary
As a significant percentage of cancers are hormone-dependent, it is important to understand how changes in hormone levels impact tumor biology over the course of a woman’s lifespan. Furthermore, greater attention should be given to the impact of tumors and tumor treatments on female sexuality. This article is intended to serve as an introduction to these two specific subjects within the vast expanse of N.O.W. subject matter.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance or • Of importance
Salakari M, Nurminen R, Sillanmäki L, Pylkkänen L, Suominen S. The importance of and satisfaction with sex life among breast cancer survivors in comparison with healthy female controls and women with mental depression or arterial hypertension: results from the Finnish nationwide HeSSup cohort study. Support Care Cancer. 2020;28(8):3847–54.
von Hippel C, Rosenberg SM, Austin SB, Sprunck-Harrild K, Ruddy KJ, Schapira L, et al. Identifying distinct trajectories of change in young breast cancer survivors’ sexual functioning. Psychooncology. 2019;28(5):1033–40.
Salonia A, Adaikan G, Buvat J, Carrier S, El-Meliegy A, Hatzimouratidis K, et al. Sexual rehabilitation after treatment for prostate cancer-part 2: recommendations from the Fourth International Consultation for Sexual Medicine (ICSM 2015). J Sex Med. 2017;14(3):297–315.
McInnis MK, Pukall CF. Sex after prostate cancer in gay and bisexual men: a review of the literature. Sex Med Rev. 2020;8(3):466–72.
Zhou WJ, Ma SC, Zhao M, Liu C, Guan XD, Bao ZS, et al. Risk factors and the prognosis of sexual dysfunction in male patients with pituitary adenomas: a multivariate analysis. Asian J Androl. 2018;20(1):43–9.
Bouloux PM, Grossman A. Hyperprolactinaemia and sexual function in the male. Br J Hosp Med. 1987;37(6):503–10.
Corona G, Mannucci E, Jannini EA, Lotti F, Ricca V, Monami M, et al. Hypoprolactinemia: a new clinical syndrome in patients with sexual dysfunction. J Sex Med. 2009;6(5):1457–66.
Donovan KA, Gonzalez BD, Nelson AM, Fishman MN, Zachariah B, Jacobsen PB. Effect of androgen deprivation therapy on sexual function and bother in men with prostate cancer: a controlled comparison. Psychooncology. 2018;27(1):316–24.
Hordern A. Intimacy and sexuality after cancer: a critical review of the literature. Cancer Nurs. 2008;31(2):E9–17. https://doi.org/10.1097/01.NCC.0000305695.12873.d5.
••Ostrom QT, Patil N, Cioffi G, Waite K, Kruchko C, Barnholtz-Sloan JS. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2013–2017. Neuro Oncol. 2020;22(Supplement_1):iv1–96. https://doi.org/10.1093/neuonc/noaa200The CBTRUS is a premiere epidemiological resource for use in brain tumor. This most recent issue also includes information about brain tumor statistics in rural populations.
Kaaks R, et al. Serum sex steroids in premenopausal women and breast cancer risk within the European Prospective Investigation into Cancer and Nutrition (EPIC). J Natl Cancer Inst. 2005;97(10):755–65.
Zheng D, Williams C, Vold JA, et al. Regulation of sex hormone receptors in sexual dimorphism of human cancers. Cancer Lett. 2018;438:24–31.
Brinton LA, Key TJ, Kolonel LN, et al. Prediagnostic sex steroid hormones in relation to male breast cancer risk. J Clin Oncol. 2015;33(18):2041–50.
Ostrom QT, Gittleman H, Xu J, Kromer C, Wolinsky Y, Kruchko C, et al. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the united states in 2009–2013. Neuro Oncol. 2016;18(5):1–75.
Korhonen K, et al. Female predominance in meningiomas cannot be explained by differences in progesterone, estrogen, or androgen receptor expression. J Neurooncol. 2006;80(1):1–7.
Wolfsberger S, et al. Progesterone-receptor index in meningiomas: correlation with clinico-pathological parameters and review of the literature. Neurosurg Rev. 2004;27(4):238–45.
Ji Y, Rankin C, Grunberg S, Sherrod AE, Ahmadi J, Townsend JJ, et al. Double-blind phase III randomized trial of the antiprogestin agent mifepristone in the treatment of unresectable meningioma: SWOG S9005. J Clin Oncol. 2015;33(34):4093–8.
Rubinstein AB, Loren D, Geier A, Reichenthal E, Gadoth N. Hormone receptors in initially excised versus recurrent intracranial meningiomas. J Neurosurg. 1994;81:184–7.
Chamberlain MC, Barnholtz JS. Medical treatment of recurrent meningiomas. Exp Rev Neurothera. 2014;11(10):1425–32.
Carroll RS, Zhang J, Dashner K, Sar M, Wilson EM, Black PM. Androgen receptor expression in meningiomas. J Neurosurg. 1995;82:453–60.
Qi Z, Shao C, Huang Y, Huo G, Zhou Y, Wang Z. Reproductive and exogenous hormone factors in relation to risk of meningioma in women: a meta-analysis. PLoS ONE. 2013;8(12):e83261.
Blitshteyn S, Cook JE, Jaeckle KA. Is there an association between meningioma and hormone replacement therapy? J Clin Oncol. 2008;26(2):279–82.
••Shu X, et al. Association of hormone replacement therapy with increased risk of meningioma in women: A hospital-based multicenter study with propensity score matching. Asia Pac J Clin Oncol. 2019;15(5):e147–53 This paper deals with an actual patient population and is valuable to the clinical practitioners in this way. This is also important to help drive guideline development for this very common intracranial tumor in women.
Deli T, et al. Hormone replacement therapy in cancer survivors - review of the literature. Pathol Oncol Res. 2020;26(1):63–78.
Mccormick A, Peterson E. Cancer in pregnancy. Obstet Gynecol Clin North Am. 2018;45(2):187–200.
Yust-Katz S, de Groot JF, Liu D, Wu J, Yuan Y, Anderson MD, et al. Pregnancy and glial brain tumors. Neurooncology. 2014;16(9):1289–94.
van Westrhenen A, Senders JT, Martin E, DiRisio AC, Broekman MLD. Clinical challenges of glioma and pregnancy: a systematic review. J Neurooncol. 2018;139(1):1–11.
Peeters S, Pagès M, Gauchotte G, et al. Interactions between glioma and pregnancy: insight from a 52-case multicenter series. J Neurosurg. 2017;128:1–11.
Pallud J, Duffau H, Razak RA, et al. Influence of pregnancy in the behavior of diffuse gliomas: clinical cases of a French glioma study group. J Neurol. 2009;256(12):2014–20.
Pallud J, Mandonnet E, Deroulers C, et al. Pregnancy increases the growth rates of World Health Organization Grade II Gliomas. Ann Neurol. 2010;67(3):398–404.
Huang W, Molitch ME. Pituitary tumors in pregnancy. Endocrinol Metab Clin North Am. 2019;48(3):569–81.
Molitch ME. Diagnosis and treatment of pituitary adenomas: a review. JAMA. 2017;317(5):516–24.
••Glezer A, Bronstein MD. Prolactinomas in pregnancy: considerations before conception and during pregnancy. Pituitary. 2020;23(1):65–9 This is an important read as this offers up-to-date information for the clinical providers about considerations in the treatment and surveillance of these patients. This paper covers a very common hormone-dependent intracranial tumor which commonly impact reproductive and sexuality in women.
Anastasiadi Z, et al. Breast cancer in young women: an overview. Updates Surg. 2017;69(3):313–7.
ESHRE Capri Workshop Group. Hormones and breast cancer. Hum Reprod Update. 2004;10(4):281–93.
Yao H, et al. Triple-negative breast cancer: is there a treatment on the horizon? Oncotarget. 2017;8(1):1913–24.
Al-Mahmood S, et al. Metastatic and triple-negative breast cancer: challenges and treatment options. Drug Deliv Transl Res. 2018;8(5):1483–507.
Phipps AI, Chlebowski RT, Prentice R, McTiernan A, Wactawski-Wende J, Kuller LH, et al. Reproductive history and oral contraceptive use in relation to risk of triple-negative breast cancer. J Natl Cancer Inst. 2011;103(6):470.
Figura NB, et al. Breast leptomeningeal disease: a review of current practices and updates on management. Breast Cancer Res Treatment. 2019;177(2):277–94.
•Gatson NT. CADD-53. Glioblastoma tumorgenesis – lessons from reproductive immunology. Neurooncology. 2018;20(suppl_6):vi283 This work discusses the biological and immunological overlap between fetal development and glioma development. This is important to provide an overview of the potential research future directions as well as helping with understanding of tumor progression during pregnancy.
Gatson NN, Pillianayagam C, Rao G, Chi TL, Prabhu S, Weathers S. SURG-12. Laser interstitial thermal therapy (LITT) to manage progressed glioblastoma during pregnancy. Neurooncology. 2016;18(suppl_6):vi193.
Gao F, Cui Y, Jiang H, Dali S, Wang Y, Jiang Z, et al. Circulating tumor cell is a common property of brain glioma and promotes the monitoring system. Oncotarget. 2016;7(44):71330–40.
Khosrotehrani K, Johnson KL, Cha DH, Salomon RN, Bianchi DW. Transfer of fetal cells with multilineage potential to maternal tissue. JAMA. 2004;292(1):75–80.
Chan WN, Gurnot C, Montine TJ, Sonnen JA, Guthrie KA, Nelson JL. Male microchimerism in the human female brain. PLoS ONE. 2012;7(9):e45592.
••Carlson RH. Study: women with brain tumors have 10 times rate of divorce as men with brain tumors. Oncol Times. 2001;23(8):63 While this paper was published 20 years ago, it remains the most reliable study that evaluates the social and psychological impact of brain tumors as it pertains to women. This further emphasizes the fact that little research has been focused on this issue in the neuro-oncology of women.
Heinrichs KD, MacKnee C, Auton-Cuff F, Domene JF. Factors affecting sexual-self esteem among young adult women in long-term heterosexual relationships. Can J Hum Sex. 2009;18(4):183–99.
Calogero RM, Thompson JK. Potential implications of the objectification of women’s bodies for women’s sexual satisfaction. Body Image. 2009;6(2):145–8.
•Bragstad S, Flatebo M, Natvig GK, Eide GE, Skeie GO, Behbahani M, et al. Predictors of quality of life and survival following Gamma Knife surgery for lung cancer brain metastases: a prospective study. J Neurosurg. 2018;129(1):71–83 Comparative study in lung cancer of the importance of quality of life on survival.
Brown PD, Ballman KV, Rummans TA, Maurer MJ, Sloan JA, Boeve BF, et al. Prospective study of quality of life in adults with newly diagnosed high-grade gliomas. J Neurooncol. 2006;76(3):283–91.
Finocchiaro CY, et al. Hidden reality: sexual sphere in brain tumor patients. Psychol Health Med. 2017;22(3):370–80.
Surbeck W, Herbet G, Duffau H. Sexuality after surgery for diffuse low-grade glioma. Neuro Oncol. 2015;17(4):574–9.
Lundberg PO, Hulter B. Sexual dysfunction in patients with hypothalamo-pituitary disorders. Exp Clin Endocrinol. 1991;98(2):81–8.
Zaidi HA, Cote DJ, Castlen JP, Burke WT, Liu YH, Smith TR, et al. Time course of resolution of hyperprolactinemia after transsphenoidal surgery among patients presenting with pituitary stalk compression. World Neurosurg. 2017;97:2–7.
Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503–12. https://doi.org/10.1517/14740338.2016.1151493.
Darzy KH, Shalet SM. Hypopituitarism following radiotherapy revisited. Endocrine Dev. 2009;15:1–24.
Darzy KH, Shalet SM. Hypopituitarism after cranial irradiation. J Endocrinol Invest. 2005;28(5 Suppl):78–87.
Ritvonen E, Karppinen A, Sintonen H, Vehkavaara S, Kivipelto L, Roine RP, et al. Normal long-term health-related quality of life can be achieved in patients with functional pituitary adenomas having surgery as primary treatment. Clin Endocrinol (Oxf). 2015;82(3):412–21.
Acquati S, Pizzocaro A, Tomei G, Giovanelli M, Libe R, Faglia G, et al. A comparative evaluation of effectiveness of medical and surgical therapy in patients with macroprolactinoma. J Neurosurg Sci. 2001;45(2):65–9.
Lucchiari C, Botturi A, Pravettoni G. The impact of decision models on self-perceived quality of life: a study on brain cancer patients. Ecancermedicalscience. 2010;4:187.
Sugitani Y, Sugitani N, Ono S. Quantitative preferences for lung cancer treatment from the patients’ perspective: a systematic review. Patient. 2020;13(5):521–36. https://doi.org/10.1007/s40271-020-00434-7.
Pinzone JJ, Katznelson L, Danila DC, Pauler DK, Miller CS, Klibanski A. Primary medical therapy of micro- and macroprolactinomas in men. J Clin Endocrinol Metab. 2000;85(9):3053–7.
••Boccia ML, Anyanda EI, Fonkem E. A preliminary report on quality of life and sexual function in brain tumor patients. J Sex Med. 2021;18(4):737–42. Detailed study of impact of brain tumor diagnosis and treatment on sexual function and relationship of sexual function to quality of life in these patients.
Boccia M, Anyanda EI, Fonkem E. A preliminary report on quality of life and sexual function in brain cancer patients. Neurooncology. 2020;22(S2):ii179.
Shindel AW, Parish SJ. Sexuality education in North American medical schools current status and future directions. J Sex Med. 2013;10(1):3–17 quiz 8.
Minniti G, Esposito V, Piccirilli M, Fratticci A, Santoro A, Jaffrain-Rea ML. Diagnosis and management of pituitary tumours in the elderly: a review based on personal experience and evidence of literature. Eur J Endocrinol. 2005;153(6):723–35.
Simon JA, Thorp J, Millheiser L. Flibanserin for premenopausal hypoactive sexual desire disorder: pooled analysis of clinical trials. J Womens Health. 2019;28(6):769–77.
••Mayer D, Lynch SE. Bremelanotide: new drug approved for treating hypoactive sexual desire disorder. Ann Pharmacother. 2020;54(7):684–90 Important new option for treatment of hypoactive sexual desire disorder in women that may be useful for treating patients with this diagnosis.
Institute of Medicine (US) Committee on the ethical and legal issues relating to the inclusion of women in clinical studies. In: Mastroianni AC, Faden R, Federman D editors. Women and health research: ethical and legal issues of including women in clinical studies: volume 2: workshop and commissioned papers. Washington (DC): National Academies Press (US); 1999. Women's participation in clinical research: from protectionism to access. Available from: https://www.ncbi.nlm.nih.gov/books/NBK236577. Accessed 20 Jan 2021.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Na Tosha N. Gatson has served as an advisory board consultant for Novocure. Maria L. Boccia declares that she has no conflict of interest. Kerianne R. Taylor declares that she has no conflict of interest. Jada K.O. Mack declares that she has no conflict of interest. Ekokobe Fonkem has served as an advisory board consultant for Novocure.
Human and Animal Rights
All reported studies/experiments with human or animal subjects performed by the authors have been previously published and complied with all applicable ethical standards as per the institutional and international research committee standards.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical collection on Neuro-oncology
Rights and permissions
About this article
Cite this article
Gatson, N.T.N., Boccia, M.L., Taylor, K.R. et al. Hormone-Dependent Tumors and Sexuality in the Neuro-Oncology of Women (N.O.W.): Women’s Brain Tumors, Gaps in Sexuality Considerations, and a Need for Evidence-Based Guidelines. Curr Oncol Rep 23, 127 (2021). https://doi.org/10.1007/s11912-021-01115-w
Accepted:
Published:
DOI: https://doi.org/10.1007/s11912-021-01115-w