Abstract
Purpose of Review
Both anti-angiogenesis and immunotherapy are well-established therapeutic options in solid tumors. Here, we review the rationale as well as clinical evidence of combining these two approaches.
Recent Findings
There is strong rationale and substantial preclinical and clinical evidence that anti-angiogenesis plays a pivotal role in overcoming immunotherapy resistance. The combination of an anti-angiogenic agent and a checkpoint inhibitor offers a more robust treatment option in many clinical trials in a wide variety of solid tumor types.
Summary
Combination of anti-angiogenesis and immunotherapy has emerged as a standard of care in some tumor types and the indication is expected to expand to more tumor types in the years to come.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Welti J, Loges S, Dimmeler S, Carmeliet P. Recent molecular discoveries in angiogenesis and antiangiogenic therapies in cancer. J Clin Invest. 2013;123(8):3190–200.
Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004;350:2335–42.
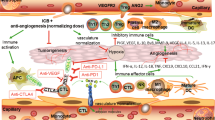
Hegde PS, Wallin JJ, Mancao C. Predictive markers of anti-VEGF and emerging role of angiogenesis inhibitors as immunotherapeutics. Semin Cancer Biol. 2018;52(Pt 2):117–24.
Jain RK. Antiangiogenesis strategies revisited: from starving tumors to alleviating hypoxia. Cancer Cell. 2014;26:605–22.
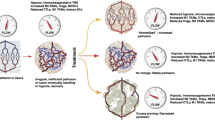
Huang YH, Goel S, Duda DG, Fukumura D, Jain RK. Vascular normalization as an emerging strategy to enhance cancer immunotherapy. Cancer Res. 2013;73:2943–8.
Melder RJ, Koenig GC, Witwer BP, Safabakhsh N, Munn LL, Jain RK. During angiogenesis, vascular endothelial growth factor and basic fibroblast growth factor regulate natural killer cell adhesion to tumor endothelium. Nat Med. 1996;2:992–7.
Hendry SA, et al. The role of the tumor vasculature in the host immune response: implications for therapeutic strategies targeting the tumor microenvironment. Front Immunol. 2016;7:621.
Motz GT, Santoro SP, Wang LP, Garrabrant T, Lastra RR, Hagemann IS, et al. Tumor endothelium FasL establishes a selective immune barrier promoting tolerance in tumors. Nat Med. 2014;20:607–15.
Gabrilovich DI, et al. Production of vascular endothelial growth factor by human tumors inhibits the functional maturation of dendritic cells. Nat Med. 1996;2:1267.
Maenhout SK, Thielemans K, Aerts JL. Location, location, location: functional and phenotypic heterogeneity between tumor-infiltrating and non-infiltrating myeloid-derived suppressor cells. Oncoimmunology. 2014;3:e956579.
Fukumura D, et al. Enhancing cancer immunotherapy using antiangiogenics: opportunities and challenges
Chen Y, Ramjiawan RR, Reiberger T, Ng MR, Hato T, Huang Y, et al. CXCR4 inhibition in tumor microenvironment facilitates anti-programmed death receptor-1 immunotherapy in sorafenib-treated hepatocellular carcinoma in mice. Hepatology. 2015;61(5):1591–602.
Liu XD, Hoang A, Zhou L, Kalra S, Yetil A, Sun M, et al. Resistance to antiangiogenic therapy is associated with an immunosuppressive tumor microenvironment in metastatic renal cell carcinoma. Cancer Immunol Res. 2015;3(9):1017–29.
Huang Y, Yuan J, Righi E, Kamoun WS, Ancukiewicz M, Nezivar J, et al. Vascular normalizing doses of antiangiogenic treatment reprogram the immunosuppressive tumor microenvironment and enhance immunotherapy. Proc Natl Acad Sci U S A. 2012;109(43):17561–6.
Li B, Lalani AS, Harding TC, Luan B, Koprivnikar K, Huan Tu G, et al. Vascular endothelial growth factor blockade reduces intratumoral regulatory T cells and enhances the efficacy of a GM-CSF-secreting cancer immunotherapy. Clin Cancer Res. 2006;12(22):6808–16.
Hodi FS, Lawrence D, Lezcano C, Wu X, Zhou J, Sasada T, et al. Bevacizumab plus ipilimumab in patients with metastatic melanoma. Cancer Immunol Res. 2014;2(7):632–42.
Wallin JJ, Bendell JC, Funke R, Sznol M, Korski K, Jones S, et al. Atezolizumab in combination with bevacizumab enhances antigen-specific T-cell migration in metastatic renal cell carcinoma. Nat Commun. 2016;7:12624.
Ramjiawan RR, Griffioen AW, Duda DG. Anti-angiogenesis for cancer revisited: is there a role for combinations with immunotherapy?
Nathan P, Chao D, Brock C, et al. The place of VEGF inhibition in the current management of renal cell carcinoma. Br J Cancer. 2006;94(9):1217–20.
Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Oudard S, et al. Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J Clin Oncol. 2009;27:3584–90.
Sternberg CN, Davis ID, Mardiak J, Szczylik C, Lee E, Wagstaff J, et al. Pazopanib in locally advanced or metastatic renal cell carcinoma: results of a randomized phase III trial. J Clin Oncol. 2010;28:1061–8.
Rini BI, Melichar B, Ueda T, Grünwald V, Fishman MN, Arranz JA, et al. Axitinib with or without dose titration for first-line metastatic renal-cell carcinoma: a randomized double-blind phase 2 trial. Lancet Oncol. 2013;14:1233–42.
Choueri TK, Halabi S, Sanford BL, et al. Cabozantinib versus sunitinib as initial targeted therapy for patients with metastatic renal cell carcinoma of poor or intermediate risk: the Alliance A031203 CABOSUN trial. J Clin Oncol. 2017;35:591–7.
Motzer RJ, Hutson TE, Glen H, Michaelson MD, Molina A, Eisen T, et al. Lenvatinib, everolimus, and the combination in patients with metastatic renal cell carcinoma: a randomized, phase 2, open-label, multicenter trial. Lancet Oncol. 2015;16:1473–82.
Heng DY, Xie W, Regan MM, et al. External validation and comparison with other models of the International Metastatic Renal-Cell Carcinoma Database Consortium prognostic model: a population-based study. Lancet Oncol. 2013;14:141–8.
Motzer RJ, Tannir NM, McDermott DF, Arén Frontera O, Melichar B, Choueiri TK, et al. Nivolumab plus ipilimumab versus sunitinib in advanced renal-cell carcinoma. N Engl J Med. 2018;378:1277–90.
McDermott DF, Lee JL, Szczylik C, et al. Pembrolizumab monotherapy as first-line therapy in advanced clear cell renal cell carcinoma: results from cohort A of KEYNOTE-427. J Clin Oncol. 2018;36S:ASCO # 4500.
Rini BI, Plimack ER, Stus V, Gafanov R, Hawkins R, Nosov D, et al. Pembrolizumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380:1116 This open-label phase 3 clinical trial compared pembrolizumab plus axitinib versus sunitinib alone as first-line therapy for advanced clear-cell RCC. The combination of pembrolizumab plus axitinib achieved a longer overall survival and progression-free survival specifically in patients with intermediate/poor-risk RCC.
Choueiri TK, Powles T, Burotto M, et al. Nivolumab + cabozantinib vs. sunitinib in first-line treatment for advanced renal cell carcinoma: first results from the randomized phase III ChekMate 9ER trial. Ann Oncol. 2020;31S4:S1142–215 This abstract details first results from the phase 3 CheckMate 9ER trial which evaluated the use of nivolumab with cabozantinib versus sunitinib. Patients treated with nivolumab and cabozantinib experienced a higher overall response rate and longer median duration of response when compared to the sunitinib group.
Motzer, R et al. Lenvatinib plus Pembrolizumab or Everolimus for Advanced Renal Cell Carcinoma. N Engl J Med 2021; 384:1289–300. This trial compared pembrolizumab plus lenvatinib to sunitinib monotherapy in untreated advanced renal cell carcinoma. The combination demonstrated a longer progression free survival, improved overall survival and a complete remission rate.
Morse MA, Sun W, Kim R, He AR, et al. The role of angiogenesis in hepatocellular carcinoma. Clin Cancer Res. 2019;259(3):912–20.
Choi SB, Han HJ, Kim WB, Song TJ, Choi SY. VEGF overexpression predicts poor survival in hepatocellular carcinoma. Open Med. 2017;12:430–9.
Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359(4):378–90.
Kudo M, Finn RS, Qin S, Han KH, Ikeda K, Piscaglia F, et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomized phase 3 non-inferiority trial. Lancet. 2018;391(10126):1163–73.
Abou-Alfa GK, Meyer T, Cheng AL, El-Koueiry AB, et al. Cabozantinib in patients with advanced and progressing hepatocellular carcinoma. N Engl J Med. 2018;379(1):54–63.
Bruix J, Qin S, Merle P, Granito A. et al; RESORCE Investigators. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;389(10064):56–66.
Zhu AX, Kang Y-K, Yen C-J, Finn RS, Galle PR, Llovet JM, et al. Ramucirumab after sorafenib in patients with increased α-fetoprotein concentrations (REACH-2): a randomized, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20(2):282–96.
Siegel AB, Cohen E, Ocean A, Lehrer D, et al. Phase II trial evaluating the clinical and biologic effects of bevacizumab in unresectable hepatocellular carcinoma. J Clin Oncol. 2008;26(18):2992–8.
Hsu CH, Yang TS, Hsu C, Toh HC, Epstein RJ, Hsiao LT, et al. Efficacy and tolerability of bevacizumab plus capecitabine as first-line therapy in patients with advanced hepatocellular carcinoma. Br J Cancer. 2010;102(6):981–6.
Hou J, Zhang H, Sun B, Karin M, et al. The immunobiology of hepatocellular carcinoma in humans and mice: basic concepts and therapeutic implications. J Hepatol. 2020;72(1):167–82.
Hato T, Zhu AX, Duda DG. Rationally combining anti-VEGF therapy with checkpoint inhibitors in hepatocellular carcinoma. Immunotherapy. 2016;8(3):299–313.
Endig J, Buitrago-Molina LE, Marhenke S, Reisinger F, Saborowski A, Schütt J, et al. Dual role of the adaptive immune system in liver injury and hepatocellular carcinoma development. Cancer Cell. 2016;30:308–23. https://doi.org/10.1016/j.ccell.2016.06.009.
Yau T, Park JW, Finn RS, Cheng A-L, et al. CheckMate 459: a randomized, multi-center phase III study of nivolumab (NIVO) vs sorafenib (SOR) as first-line (1L) treatment in patients (pts) with advanced hepatocellular carcinoma(aHCC). 2019;30(5):874–5.
Yau T, Kang Y-K, Kim T-Y, El-Khoueiry AB, et al. Efficacy and safety of nivolumab plus ipilimumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib. The CheckMate 040 randomized clinical trial. JAMA Oncol. 2020;6(11):e204564.
Zhu AX, Finn RS, Edeline J, Cattan S, Ogasawara S, Palmer D, et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): A non-randomized, open-label phase 2 trial. Lancet Oncol. 2018;19:940–52.
Finn RS, Qin S, Ikeda M, Galle PR, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med. 2020;382:1894–905 The open-label, phase 3 IMbrave150 trial studied atezolizumab with bevacizumab versus single-agent sorafenib in hepatocellular carcinoma patients with Child-Pugh A hepatic function. Overall survival and median progression-free survival were improved with atezolizumab plus bevacizumab, leading to the FDA approval of this combination in HCC.
Zhu AX, Finn RS, Ikeda M, Sung MW, Baron AD, Kudo M, et al. A phase Ib study of lenvatinib (LEN) plus pembrolizumab (PEMBRO) in unresectable hepatocellular carcinoma (uHCC). J Clin Oncol. 2020;38(suppl):4519.
Llovet JM, Kudo M, Chen A-L, Finn RS, et al. Lenvatinib (len) plus pembrolizumab (pembro) for the first-line treatment of patients (pts) with advanced hepatocellular carcinoma (HCC): Phase 3 LEAP-002 study. J Clin Oncol. 2019;37(15 suppl).
Marabelle A, Le DT, Ascierto PA, et al. Efficacy of pembrolizumab in patients with noncolorectal high microsatellite instability/mismatch repair-deficient cancer: results from the Phase II KEYNOTE-158 study. J Clin Oncol. 2020;38:1–10.
Soumerai TE, Donoghue MTA, Bandlamudi C, Srinivasan P, Chang MT, Zamarin D, et al. Clinical utility of prospective molecular characterization in advanced endometrial cancer. Clin Cancer Res. 2018;24:5939–47.
Ott PA, Bang YJ, Berton-Rigaud D, Elez E, Pishvaian MJ, Rugo HS, et al. Safety and antitumor activity of pembrolizumab in advanced programmed death ligand 1-positive endometrial cancer: results from the KEYNOTE-028 study. J Clin Oncol. 2017;35:2535–41.
Konstantinopoulos PA, Luo W, Liu JF, Gulhan DC, Krasner C, Ishizuka JJ, et al. Phase II study of avelumab in patients with mismatch repair deficient and mismatch repair proficient recurrent/persistent endometrial cancer. J Clin Oncol. 2019;37:2786–94.
Kimura T, Kato Y, Ozawa Y, Kodama K, Ito J, Ichikawa K, et al. Immunomodulatory activity of lenvatinib contributes to antitumor activity in the Hepa1-6 hepatocellular carcinoma model. Cancer Sci. 2018;109:3993–4002.
Vergote I, Teneriello M, Powell MA, et al. A phase II trial of lenvatinib in patients with advanced or recurrent endometrial cancer: angiopoietin-2 as a predictive marker for clinical outcomes. J Clin Oncol. 2013;31(15 suppl; abstr 5520).
Makker V, Taylor MH, Aghajanian C, et al. Lenvatinib plus pembrolizumab in patients with advanced endometrial cancer. J Clin Oncol. 2020;38(26):2981–92 In this open-label, single-arm, phase 2 trial, patients with advanced endometrial cancer were given lenvatinib with pembrolizumab. This combination achieved an objective response in 39.6% of patients at week 24 and median overall survival was 16.7 months. Results from the phase 3 trial looking at lenvatinib with pembrolizumab in this patient population are pending.
Sandler A, Gray R, Perry MC, Brahmer J, Schiller JH, Dowlati A, et al. Paclitaxel–carboplatin alone or with bevacizumab for non–small-cell lung cancer. N Engl J Med. 2006;355:2542–50.
Socinski MA, Jotte RM, Cappuzzo F, et al. Atezolizumab for first line treatment of metastatic nonsquamous NSCLC. N Engl J Med. 2018;378(24):2288–301 This trial assessed the clinical benefit of adding atezolizumab to chemotherapy with or without bevacizumab in patients with nonsquamous NSCLC. The patients who received immunotherapy with chemotherapy plus bevacizumab experienced the most benefit, with prolonged progression-free survival and overall survival as well as higher response rates.
Clarke K, Smith K, Gullick WJ, Harris AL. Mutant epidermal growth factor receptor enhances induction of vascular endothelial growth factor by hypoxia and insulin-like growth factor-1 via a PI3 kinase dependent pathway. Br J Cancer. 2001;84:1322–9.
Reck M, Mok TSK, Nishio M, Jotte RM, Cappuzzo F, Orlandi F, et al. Atezolizumab plus bevacizumab and chemotherapy in non-small-cell lung cancer (IMpower150): key subgroup analyses of patients with EGFR mutations or baseline liver metastases in a randomized, open-label phase 3 trial. Lancet Respir Med. 2019;7(5):387–401.
Herbst RS, Arkenau HT. Santana-Davila R, et al Ramucirumab plus pembrolizumab in patients with previously treated advanced non-small-cell lung cancer, gastro-esophageal cancer, or urothelial carcinomas (JVDF): a multicohort, non-randomized, open-label, phase 1a/b trial. Lancet Oncol. 2019;20(8):1109–23.
Bang YJ, Golan T, Dahan L, Fu S, Moreno V, Park K, et al. Ramucirumab and durvalumab for previously treated, advanced non-small-cell lung cancer, gastric/gastro-oesophageal junction adenocarcinoma, or hepatocellular carcinoma: an open-label, phase Ia/b study (JVDJ). Eur J Cancer. 2020;137:272–84.
Srivastava A, Ralhan R, Kaur J. Angiogenesis in cutaneous melanoma: pathogenesis and clinical implications. Microsc Res Tech. 2003;60(2):208–24.
Graells J, Vinyals A, Figueras A, Llorens A, Moreno A, Marcoval J, et al. Overproduction of VEGF concomitantly expressed with its receptors promotes growth and survival of melanoma cells through MAPK and PI3K signaling. J Invest Dermatol. 2004;123(6):1151–61.
Ugurel S, Rappl G, Tilgen W, Reinhold U. Increased serum concentration of angiogenic factors in malignant melanoma patients correlates with tumor progression and survival. J Clin Oncol. 2001;19(2):577–83.
Ellis LM, Fidler IJ. Angiogenesis and metastasis. Eur J Cancer. 1996;32A(14):2451–60. https://doi.org/10.1016/s0959-8049(96)00389-9.
Shrimali RK, Yu Z, Theoret MR, Chinnasamy D, Restifo NP, Rosenberg SA. Antiangiogenic agents can increase lymphocyte infiltration into tumor and enhance the effectiveness of adoptive immunotherapy of cancer. Cancer Res. 2010;70(15):6171–80.
Perez DG, Suman VJ, Fitch TR, Amatruda T 3rd, Morton RF, Jilani SZ, et al. Phase 2 trial of carboplatin, weekly paclitaxel, and biweekly bevacizumab in patients with unresectable stage IV melanoma: a North Central Cancer Treatment Group study, N047A. Cancer. 2009;115(1):119–27.
Ferrucci PF, Minchella I, Mosconi M, Gandini S, Verrecchia F, Cocorocchio E, et al. Dacarbazine in combination with bevacizumab for the treatment of unresectable/metastatic melanoma: a phase II study. Melanoma Res. 2015;25(3):239–45.
Piperno-Neumann S, Diallo A, Etienne-Grimaldi MC, Bidard FC, Rodrigues M, Plancher C, et al. Phase II trial of bevacizumab in combination with temozolomide as first-line treatment in patients with metastatic uveal melanoma. Oncologist. 2016;21(3):281–2.
Grignol VP, Olencki T, Relekar K, Taylor C, Kibler A, Kefauver C, et al. A phase 2 trial of bevacizumab and high-dose interferon alpha 2B in metastatic melanoma. J Immunother. 2011;34(6):509–15.
Yuan J, Zhou J, Dong Z, Tandon S, Kuk D, Panageas KS, et al. Pretreatment serum VEGF is associated with clinical response and overall survival in advanced melanoma patients treated with ipilimumab. Cancer Immunol Res. 2014;2(2):127–32.
Ott PA, Hodi FS, Buchbinder EI. Inhibition of immune checkpoints and vascular endothelial growth factor as combination therapy for metastatic melanoma: an overview of rationale, preclinical evidence, and initial clinical data. Front Oncol. 2015;5:202.
Taylor MH, Lee CH, Makker V, Rasco D, Dutcus CE, Wu J, et al. Phase IB/II trial of lenvatinib plus pembrolizumab in patients with advanced renal cell carcinoma, endometrial cancer, and other selected advanced solid tumors. J Clin Oncol. 2020;38(11):1154–63.
Sheng X, Yan X, Chi Z, Si L, Cui C, Tang B, et al. Axitinib in combination with toripalimab, a humanized immunoglobulin G4 monoclonal antibody against programmed cell death-1, in patients with metastatic mucosal melanoma: an open-label phase IB trial. J Clin Oncol. 2019;37:2987–99.
Carter T, Shaw H, Cohn-Brown D, Chester K, Mulholland P. Ipilimumab and bevacizumab in glioblastoma. Clin Oncol. 2016;28:622–6.
Paoluzzi L, Cacavio A, Ghesani M, Karambelkar A, Rapkiewicz A, Weber J, et al. Response to anti-PD1 therapy with nivolumab in metastatic sarcomas. Clin Sarcoma Res. 2016;6:24.
Wilky BA, Trucco MM, Subhawong TK, Florou V, Park W, Kwon D, et al. Axitinib plus pembrolizumab in patients with advanced sarcomas including alveolar soft-part sarcoma: a single-centre, single-arm, phase 2 trial. Lancet Oncol. 2019;20(6):837–48.
Song L, Pan D, Zhou R. Combination nivolumab and bevacizumab for metastatic myxofibrosarcoma: a case report and review of the literature. Mol Clin Oncol. 2020;13(5):54.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Alex Chambers declares that she has no conflict of interest. Madappa Kundranda declares that he has no conflict of interest. Santosh Rao declares that he has no conflict of interest. Fade Mahmoud declares that he has no conflict of interest. Jiaxin Niu has received speaker’s honorarium from OncLive, and has received compensation from Boehringer Ingelheim, Merck, and AstraZeneca for participation on advisory boards.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Lung Cancer
Rights and permissions
About this article
Cite this article
Chambers, A., Kundranda, M., Rao, S. et al. Anti-angiogenesis Revisited: Combination with Immunotherapy in Solid Tumors. Curr Oncol Rep 23, 100 (2021). https://doi.org/10.1007/s11912-021-01099-7
Accepted:
Published:
DOI: https://doi.org/10.1007/s11912-021-01099-7