Abstract
Purpose of Review
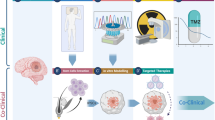
Recurrent glioblastoma (rGBM) has no standard treatment. Despite a better molecular knowledge, few therapies have brought changes in clinical practice so far. Here we will review the current data evaluating the re-radiation, re-resection, bevacizumab, and cytotoxic chemotherapy agents in this setting. We will also discuss the advances of immunotherapy and the possible benefit of this treatment for patients with rGBM.
Recent Findings
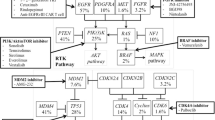
Next-generation sequencing is increasingly utilized in the clinical practice of neuro-oncologists, bringing gene mutations as targets for therapies. As in other solid tumors, immunotherapy has been also extensively studied in rGBM, with interesting results in phase I and II trials. The most promising therapies in the horizon are combinations including immune checkpoint inhibitors, virotherapy, vaccines, and monoclonal antibodies.
Summary
Although re-radiation, re-resection, bevacizumab, and chemotherapy are still the most widely used therapies for treating rGBM, the clinical benefit from these treatments is still not well established. Preliminary results of studies with immune checkpoint inhibitors were disappointing, but virotherapy emerges as more promising immunotherapy in rGBM, especially in combination with other strategies. In addition to the gain in overall survival, the improvement in the quality of life of these patients is also expected.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
• Wick W, Gorlia T, Bendszus M, Taphoorn M, Sahm F, Harting I, et al. Lomustine and bevacizumab in progressive glioblastoma. N Engl J Med. 2017;377(20):1954–63. https://doi.org/10.1056/NEJMoa1707358. The first published phase 3 trial with BEV in rGBM and did not show any gain in OS or in quality of life. However, FDA kept the appoval of BEV for rGBM.
•• Eckel-Passow JE, Lachance DH, Molinaro AM, Walsh KM, Decker PA, Sicotte H, et al. Glioma groups based on 1p/19q, IDH, and TERT promoter mutations in tumors. N Engl J Med. 2015;372(26):2499–508. https://doi.org/10.1056/NEJMoa1407279. Published in New England Journal of Medicine , it was one of the most downloaded article in 2015. Some of these mutations were incorporated to WHO tumor classification.
Brzozowska A, Toruń A, Mazurkiewicz M. The impact of surgery on the efficacy of adjuvant therapy in glioblastoma multiforme. Adv Clin Exp Med. 2015;24(2):279–87. https://doi.org/10.17219/acem/40456.
Sastry RA, Shankar GM, Gerstner ER, Curry WT. The impact of surgery on survival after progression of glioblastoma: a retrospective cohort analysis of a contemporary patient population. J Clin Neurosci. 2018;53:41–7. https://doi.org/10.1016/j.jocn.2018.04.004.
• Ringel F, Pape H, Sabel M, Krex D, Bock HC, Misch M, et al. Clinical benefit from resection of recurrent glioblastomas: results of a multicenter study including 503 patients with recurrent glioblastomas undergoing surgical resection. Neuro Oncol. 2016;18(1):96–104. https://doi.org/10.1093/neuonc/nov145. Positive results regarding re-resection in rGBM, with a robust numer of patients.
Hou LC, Veeravagu A, Hsu AR, Tse VC. Recurrent glioblastoma multiforme: a review of natural history and management options. Neurosurg Focus. 2006;20(4):E5.
•• Lu VM, Jue TR, McDonald KL, Rovin RA. The survival effect of repeat surgery at glioblastoma recurrence and its trend: a systematic review and meta-analysis. World Neurosurg. 2018;115:453–459.e3. https://doi.org/10.1016/j.wneu.2018.04.016. The newest and one of the largest systematic reviews about the benefit of re-resection in rGBM.
Pala A, Schmitz AL, Knoll A, Schneider M, Hlavac M, König R, et al. Is MGMT promoter methylation to be considered in the decision making for recurrent surgery in glioblastoma patients? Clin Neurol Neurosurg. 2018;167:6–10. https://doi.org/10.1016/j.clineuro.2018.02.003.
Wallner KE, Galicich JH, Krol G, Arbit E, Malkin MG. Patterns of failure following treatment for glioblastoma multiforme and anaplastic astrocytoma. Int J Radiat Oncol Biol Phys. 1989;16(6):1405–9.
Combs SE, Thilmann C, Edler L, Jr D, Schulz-Ertner D. Efficacy of fractionated stereotactic reirradiation in recurrent gliomas: long-term results in 172 patients treated in a single institution. J Clin Oncol. 2005;23(34):8863–9.
Reynaud T, Bertaut A, Farah W, Thibouw D, Crehange G, Truc G, et al. Hypofractionated stereotactic radiotherapy as a salvage therapy for recurrent high-grade gliomas: single-center experience. Technol Cancer Res Treat. 2018;1(17):1533033818806498. https://doi.org/10.1177/1533033818806498.
Fogh SE, Andrews DW, Glass J, Curran W, Glass C, Champ C, et al. Hypofractionated stereotactic radiation therapy: an effective therapy for recurrent high-grade gliomas. J Clin Oncol. 2010;28(18):3048–53. https://doi.org/10.1200/JCO.2009.25.6941.
Laing RW, Warrington AP, Graham J, Britton J, Hines F, Brada M. Efficacy and toxicity of fractionated stereotactic radiotherapy in the treatment of recurrent gliomas (phase I/II study). Radiother Oncol. 1993;27(1):22–9.
Hudes RS, Corn BW, Werner-Wasik M, Andrews D, Rosenstock J, Thoron L, et al. A phase I dose escalation study of hypofractionated stereotactic radiotherapy as salvage therapy for persistent or recurrent malignant glioma. Int J Radiat Oncol Biol Phys. 1999;43(2):293–8.
• Taunk NK, Moraes FY, Escorcia FE, Mendez LC, Beal K, Marta GN. External beam re-irradiation, combination chemoradiotherapy, and particle therapy for the treatment of recurrent glioblastoma. Expert Rev Anticancer Ther. 2016;16(3):347–58. https://doi.org/10.1586/14737140.2016.1143364. One prospective study that showed benefit of re-radiation and decrease of complication using bevacizumab.
Franceschi E, Omuro AM, Lassman AB, Demopoulos A, Nolan C, Abrey LE. Salvage temozolomide for prior temozolomide responders. Cancer. 2005;104(11):2473–6.
Minniti G, Armosini V, Salvati M, Lanzetta G, Caporello P, Mei M, et al. Fractionated stereotactic reirradiation and concurrent temozolomide in patients with recurrent glioblastoma. J Neuro-Oncol. 2011;103(3):683–91. https://doi.org/10.1007/s11060-010-0446-8.
Palmer JD, Bhamidipati D, Song A, Eldredge-Hindy HB, Siglin J, Dan TD, et al. Bevacizumab and re-irradiation for recurrent high grade gliomas: does sequence matter? J Neuro-Oncol. 2018;140(3):623–8. https://doi.org/10.1007/s11060-018-2989-z.
Barney C, Shukla G, Bhamidipati D, Palmer JD. Re-irradiation for recurrent glioblastoma multiforme. Chin Clin Oncol. 2017;6(4):36. https://doi.org/10.21037/cco.2017.06.18.
Topfer L-A, Farrah K. Alternating electric fields (“tumour-treating fields”) for the treatment of glioblastoma. CADTH Issues in Emerging Health Technologies: Canadian Agency for Drugs and Technologies in Health; 2018.
• Stupp R, Wong ET, Kanner AA, Steinberg D, Engelhard H, Heidecke V, et al. NovoTTF-100A versus physician’s choice chemotherapy in recurrent glioblastoma: a randomised phase III trial of a novel treatment modality. Eur J Cancer. 2012;48(14):2192–202. https://doi.org/10.1016/j.ejca.2012.04.011. Despite not showing gain in OS (primary end point) for rGBM, TTF emerges as a treatment option and was approved by FDA in this context.
Mrugala MM, Engelhard HH, Tran DD, Kew Y, Cavaliere R, Villano JL, et al. Clinical practice experience with NovoTTF-100A™ system for glioblastoma: the Patient Registry Dataset (PRiDe). Semin Oncol. 2014;41(Suppl 6):S4–S13. https://doi.org/10.1053/j.seminoncol.2014.09.010.
Stupp R, Mason WP, Van Den Bent MJ, Weller M, Fisher B, Taphoorn MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352(10):987–96.
Kim S-H, Yoo H, Chang JH, Kim C-Y, Chung DS, Kim SH, et al. Procarbazine and CCNU chemotherapy for recurrent glioblastoma with MGMT promoter methylation. J Korean Med Sci. 2018;33(24):e167. https://doi.org/10.3346/jkms.2018.33.e167.
• Jungk C, Chatziaslanidou D, Ahmadi R, Capper D, Bermejo JL, Exner J, et al. Chemotherapy with BCNU in recurrent glioma: analysis of clinical outcome and side effects in chemotherapy-naïve patients. BMC Cancer. 2016(16):81. https://doi.org/10.1186/s12885-016-2131-6. A study showing the real benefit and activity of Lomustine in rGBM after Stupp protocol being the standard treatment for newly diagnosed GBM.
• Weller M, Tabatabai G, Kästner B, Felsberg J, Steinbach JP, Wick A, et al. MGMT promoter methylation is a strong prognostic biomarker for benefit from dose-intensified temozolomide rechallenge in progressive glioblastoma: the DIRECTOR trial. Clin Cancer Res. 2015;21(9):2057–64. https://doi.org/10.1158/1078-0432.CCR-14-2737 The trial helped to identified patients that could have benefit or temozolomide re-exposure.
Franceschi E, Lamberti G, Visani M, Paccapelo A, Mura A, Tallini G, et al. Temozolomide rechallenge in recurrent glioblastoma: when is it useful? Future Oncol. 2018;14(11):1063–9. https://doi.org/10.2217/fon-2017-0681.
• Johnson DR, Omuro AM, Ravelo A, Sommer N, Guerin A, Ionescu-Ittu R, et al. Overall survival in patients with glioblastoma before and after bevacizumab approval. Curr Med Res Opin. 2018;34(5):813–20. https://doi.org/10.1080/03007995.2017.1392294. After the phase 3 not showing gain in OS, these study data support the continued use of bevacizumab in clinical practice.
Hoang-Minh LB, Mitchell DA. Immunotherapy for brain tumors. Curr Treat Options in Oncol. 2018;19(11):60. https://doi.org/10.1007/s11864-018-0576-3.
Nduom EK, Wei J, Yaghi NK, Huang N, Kong L-Y, Gabrusiewicz K, et al. PD-L1 expression and prognostic impact in glioblastoma. Neuro-Oncology. 2016;18(2):195–205. https://doi.org/10.1093/neuonc/nov172.
•• Reardon D, Omuro A, Brandes A, Rieger J, Wick A, Sepulveda J, et al. OS10.3 randomized phase 3 study evaluating the efficacy and safety of nivolumab vs bevacizumab in patients with recurrent glioblastoma: CheckMate 143. Neuro Oncol. 2017;19(suppl_3):iii21–ii. First randomized data about the activity of immune checkpoint inhibitors in GBM.
•• Reardon DA, Sampson JH, Sahebjam S, Lim M, Baehring JM, Vlahovic G, et al. Safety and activity of nivolumab (nivo) monotherapy and nivo in combination with ipilimumab (ipi) in recurrent glioblastoma (GBM): updated results from checkmate-143. American Society of Clinical Oncology Annual Meeting; Chicago 2016. First randomized data about the safety of immune checkpoint inhibitors in GBM.
Cloughesy TF, Mochizuki AY, Orpilla JR, Hugo W, Lee AH, Davidson TB, et al. Neoadjuvant anti-PD-1 immunotherapy promotes a survival benefit with intratumoral and systemic immune responses in recurrent glioblastoma. Nat Med. 2019;25(3):477–86. https://doi.org/10.1038/s41591-018-0337-7.
• Groot Jd. Window-of-opportunity clinical trial of pembrolizumab in recurrent glioblastoma patients. Society of Neuro-Oncology Meeting; New Orleans 2018. Trying to indentify why immune checkpoint inhibitors fail in brain tumors.
• Reardon DA, Schuster J, Tran DD, Fink KL, Nabors LB, Li G, et al. ReACT: overall survival from a randomized phase II study of rindopepimut (CDX-110) plus bevacizumab in relapsed glioblastoma. American Society of Clinical Oncology Annual Meeting; Chicago 2015. Study that showed clinical benefit of vaccine in rGBM.
•• Weller M, Butowski N, Tran DD, Recht LD, Lim M, Hirte H, et al. Rindopepimut with temozolomide for patients with newly diagnosed, EGFRvIII-expressing glioblastoma (ACT IV): a randomised, double-blind, international phase 3 trial. Lancet Oncol. 2017;18(10):1373–85. https://doi.org/10.1016/S1470-2045(17)30517-X. A phase 3 study, with robust data, showing no activity of vaccine anti EGFRvIII in GBM.
Foreman PM, Friedman GK, Cassady KA, Markert JM. Oncolytic virotherapy for the treatment of malignant glioma. Neurotherapeutics. 2017;14(2):333–44. https://doi.org/10.1007/s13311-017-0516-0.
Parker JN, Bauer DF, Cody JJ, Markert JM. Oncolytic viral therapy of malignant glioma. Neurotherapeutics. 2009;6(3):558–69. https://doi.org/10.1016/j.nurt.2009.04.011.
Bahreyni A, Ghorbani E, Fuji H, Ryzhikov M, Khazaei M, Erfani M, et al. Therapeutic potency of oncolytic virotherapy–induced cancer stem cells targeting in brain tumors, current status, and perspectives. J Cell Biochem. 2019;120(3):2766–73. https://doi.org/10.1002/jcb.27661.
•• Lang FF, Conrad C, Gomez-Manzano C, Yung WA, Sawaya R, Weinberg JS, et al. Phase I study of DNX-2401 (Delta-24-RGD) oncolytic adenovirus: replication and immunotherapeutic effects in recurrent malignant glioma. J Clin Oncol. 2018;36(14):1419–27. https://doi.org/10.1200/JCO.2017.75.8219. Positive trial with viraltherapy in rGBM.
•• Zadeh G. Interim results of a phase II multicenter study of the conditionally replicative oncolytic adenovirus DNX-2401 with pembrolizumab (Keytruda) for recurrent glioblastoma; CAPTIVE study (KEYNOTE-192). Society of Neuro-Oncology Meeting; New Orleans 2018. Positive trial with viraltherapy in rGBM and safe combination with immune checkpoint inhibitor.
Cloughesy TF, Landolfi J, Hogan DJ, Bloomfield S, Carter B, Chen CC, et al. Phase 1 trial of vocimagene amiretrorepvec and 5-fluorocytosine for recurrent high-grade glioma. Sci Transl Med. 2016;8(341):341ra75. https://doi.org/10.1126/scitranslmed.aad9784.
Vredenburgh JJ, Desjardins A, Herndon JE, Marcello J, Reardon DA, Quinn JA, et al. Bevacizumab plus irinotecan in recurrent glioblastoma multiforme. J Clin Oncol. 2007;25(30):4722–9.
Blumenthal DT, Dvir A, Lossos A, Tzuk-Shina T, Lior T, Limon D, et al. Clinical utility and treatment outcome of comprehensive genomic profiling in high grade glioma patients. J Neuro-Oncol. 2016;130(1):211–9.
Van Den Bent MJ, French P, Eoli M, Sepúlveda JM, Walenkamp AME, Frenel J-S, et al. Updated results of the INTELLANCE 2/EORTC trial 1410 randomized phase II study on Depatux–M alone, Depatux-M in combination with temozolomide (TMZ) and either TMZ or lomustine (LOM) in recurrent EGFR amplified glioblastoma (NCT02343406). American Society of Clinical Oncology Annual Meeting; Chicago 2018. It was the first phase II trial with patients with rGBM demonstrating OS gain blocking EGFR pathway.
Kaley TJ, Touat M, Subbiah V, Hollebecque A, Rodon J, Lockhart AC, et al. BRAF inhibition in BRAFV600-mutant gliomas: results from the VE-BASKET Study. J Clin Oncol. 2018:JCO2018789990. https://doi.org/10.1200/JCO.2018.78.9990.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publishers Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Neuro-oncology
Rights and permissions
About this article
Cite this article
Chaul-Barbosa, C., Marques, D.F. How We Treat Recurrent Glioblastoma Today and Current Evidence. Curr Oncol Rep 21, 94 (2019). https://doi.org/10.1007/s11912-019-0834-y
Published:
DOI: https://doi.org/10.1007/s11912-019-0834-y