Abstract
Purpose of Review
For millennia, there has been interest in the use of cannabis for the treatment of epilepsy. However, it is only recently that appropriately powered controlled studies have been completed. In this review, we present an update on the research investigating the use of cannabidiol (CBD), a non-psychoactive component of cannabis, in the treatment of epilepsy.
Recent Findings
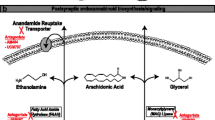
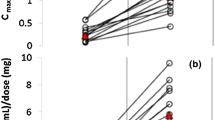
While the anticonvulsant mechanism of action of CBD has not been entirely elucidated, we discuss the most recent data available including its low affinity for the endocannabinoid receptors and possible indirect modulation of these receptors via blocking the breakdown of anandamide. Additional targets include activation of the transient receptor potential of vanilloid type-1 (TRPV1), antagonist action at GPR55, targeting of abnormal sodium channels, blocking of T-type calcium channels, modulation of adenosine receptors, modulation of voltage-dependent anion selective channel protein (VDAC1), and modulation of tumor necrosis factor alpha release. We also discuss the most recent studies on various artisanal CBD products conducted in patients with epilepsy in the USA and internationally. While a high percentage of patients in these studies reported improvement in seizures, these studies were either retrospective or conducted via survey. Dosage/preparation of CBD was either unknown or not controlled in the majority of these studies. Finally, we present data from both open-label expanded access programs (EAPs) and randomized placebo-controlled trials (RCTs) of a highly purified oral preparation of CBD, which was recently approved by the FDA in the treatment of epilepsy. In the EAPs, there was a significant improvement in seizure frequency seen in a large number of patients with various types of treatment-refractory epilepsy. The RCTs have shown significant seizure reduction compared to placebo in patients with Dravet syndrome and Lennox-Gastaut syndrome. Finally, we describe the available data on adverse effects and drug-drug interactions with highly purified CBD. While this product is overall well tolerated, the most common side effects are diarrhea and sedation, with sedation being much more common in patients taking concomitant clobazam. There was also an increased incidence of aspartate aminotransferase and alanine aminotransferase elevations while taking CBD, with many of the patients with these abnormalities also taking concomitant valproate. CBD has a clear interaction with clobazam, significantly increasing the levels of its active metabolite N-desmethylclobazam in several studies; this is felt to be due to CBD’s inhibition of CYP2C19. EAP data demonstrate other possible interactions with rufinamide, zonisamide, topiramate, and eslicarbazepine. Additionally, there is one case report demonstrating need for warfarin dose adjustment with concomitant CBD.
Summary
Understanding of CBD’s efficacy and safety in the treatment of TRE has expanded significantly in the last few years. Future controlled studies of various ratios of CBD and THC are needed as there could be further therapeutic potential of these compounds for patients with epilepsy.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Maa E, Figi P. The case for medical marijuana in epilepsy. Epilepsia. 2014;55:783–6.
Szaflarski JP, Bebin EM. Cannabis, cannabidiol, and epilepsy--from receptors to clinical response. Epilepsy Behav. 2014;41:277–82.
Gaston TE, Friedman D. Pharmacology of cannabinoids in the treatment of epilepsy. Epilepsy Behav. 2017;70:313–8.
Pertwee RG, Cascio MG. Known pharmacological actions of delta-9-tetrahydrocannabinol and of four other chemical constituents of cannabis that activate cannabinoid receptors. In: Pertwee RG, editor. Handbook of cannabis. Oxford: Oxford University Press; 2014. p. 115.
Pertwee RG. The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: delta9-tetrahydrocannabinol, cannabidiol and delta9-tetrahydrocannabivarin. Br J Pharmacol. 2008;153:199–215.
Wallace MJ, Blair RE, Falenski KW, Martin BR, DeLorenzo RJ. The endogenous cannabinoid system regulates seizure frequency and duration in a model of temporal lobe epilepsy. J Pharmacol Exp Ther. 2003;307:129–37.
Wallace MJ, Martin BR, DeLorenzo RJ. Evidence for a physiological role of endocannabinoids in the modulation of seizure threshold and severity. Eur J Pharmacol. 2002;452:295–301.
Wallace MJ, Wiley JL, Martin BR, DeLorenzo RJ. Assessment of the role of CB1 receptors in cannabinoid anticonvulsant effects. Eur J Pharmacol. 2001;428:51–7.
McPartland JM, Duncan M, Di Marzo V, Pertwee RG. Are cannabidiol and Delta(9) -tetrahydrocannabivarin negative modulators of the endocannabinoid system? A systematic review. Br J Pharmacol. 2015;172:737–53.
Perucca E. Cannabinoids in the treatment of epilepsy: hard evidence at last? J Epilepsy Res. 2017;7:61–76.
Thomas A, Baillie GL, Phillips AM, Razdan RK, Ross RA, Pertwee RG. Cannabidiol displays unexpectedly high potency as an antagonist of CB1 and CB2 receptor agonists in vitro. Br J Pharmacol. 2007;150:613–23.
Bisogno T, Hanus L, De Petrocellis L, et al. Molecular targets for cannabidiol and its synthetic analogues: effect on vanilloid VR1 receptors and on the cellular uptake and enzymatic hydrolysis of anandamide. Br J Pharmacol. 2001;134:845–52.
• Cleeren E, Casteels C, Goffin K, et al. Positron emission tomography imaging of cerebral glucose metabolism and type 1 cannabinoid receptor availability during temporal lobe epileptogenesis in the amygdala kindling model in rhesus monkeys. Epilepsia. 2018;59:959–70. This study documents the relationship between epileptogenesis and endocannabinoid system through all stages of epilepsy development.
Szaflarski J. The highs and lows of the endocannabinoid system – Another piece to the epilepsy puzzle? Epilepsy Curr. 2018;in print.
Vilela LR, Lima IV, Kunsch EB, Pinto HPP, de Miranda AS, Vieira ÉLM, et al. Anticonvulsant effect of cannabidiol in the pentylenetetrazole model: pharmacological mechanisms, electroencephalographic profile, and brain cytokine levels. Epilepsy Behav. 2017;75:29–35.
Naziroglu M. TRPV1 channel: a potential drug target for treating epilepsy. Curr Neuropharmacol. 2015;13:239–47.
Ryberg E, Larsson N, Sjogren S, et al. The orphan receptor GPR55 is a novel cannabinoid receptor. Br J Pharmacol. 2007;152:1092–101.
Hurst K, Badgley C, Ellsworth T, Bell S, Friend L, Prince B, et al. A putative lysophosphatidylinositol receptor GPR55 modulates hippocampal synaptic plasticity. Hippocampus. 2017;27:985–98.
Kaplan JS, Stella N, Catterall WA, Westenbroek RE. Cannabidiol attenuates seizures and social deficits in a mouse model of Dravet syndrome. Proc Natl Acad Sci U S A. 2017;114:11229–34.
Patel RR, Barbosa C, Brustovetsky T, Brustovetsky N, Cummins TR. Aberrant epilepsy-associated mutant Nav1.6 sodium channel activity can be targeted with cannabidiol. Brain J Neurol. 2016;139:2164–81.
Ross HR, Napier I, Connor M. Inhibition of recombinant human T-type calcium channels by Delta9-tetrahydrocannabinol and cannabidiol. J Biol Chem. 2008;283:16124–34.
Devinsky O, Cilio MR, Cross H, Fernandez-Ruiz J, French J, Hill C, et al. Cannabidiol: pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders. Epilepsia. 2014;55:791–802.
During MJ, Spencer DD. Adenosine: a potential mediator of seizure arrest and postictal refractoriness. Ann Neurol. 1992;32:618–24.
Rimmerman N, Ben-Hail D, Porat Z, Juknat A, Kozela E, Daniels MP, et al. Direct modulation of the outer mitochondrial membrane channel, voltage-dependent anion channel 1 (VDAC1) by cannabidiol: a novel mechanism for cannabinoid-induced cell death. Cell Death Dis. 2013;4:e949.
Kozela E, Juknat A, Vogel Z. Modulation of astrocyte activity by cannabidiol, a nonpsychoactive cannabinoid. Int J Mol Sci. 2017;18
Pelz MC, Schoolcraft KD, Larson C, Spring MG, Lopez HH. Assessing the role of serotonergic receptors in cannabidiol’s anticonvulsant efficacy. Epilepsy Behav. 2017;73:111–8.
• Sulak D, Saneto R, Goldstein B. The current status of artisanal cannabis for the treatment of epilepsy in the United States. Epilepsy Behav. 2017;70:328–33. While open label, this study discusses various aspects of treating epilepsy with artisanal cannabis products in various settings.
Press CA, Knupp KG, Chapman KE. Parental reporting of response to oral cannabis extracts for treatment of refractory epilepsy. Epilepsy Behav. 2015;45:49–52.
Hussain SA, Zhou R, Jacobson C, Weng J, Cheng E, Lay J, et al. Perceived efficacy of cannabidiol-enriched cannabis extracts for treatment of pediatric epilepsy: a potential role for infantile spasms and Lennox-Gastaut syndrome. Epilepsy Behav. 2015;47:138–41.
Tzadok M, Uliel-Siboni S, Linder I, Kramer U, Epstein O, Menascu S, et al. CBD-enriched medical cannabis for intractable pediatric epilepsy: the current Israeli experience. Seizure. 2016;35:41–4.
Hausman-Kedem M, Menascu S, Kramer U. Efficacy of CBD-enriched medical cannabis for treatment of refractory epilepsy in children and adolescents - an observational, longitudinal study. Brain and Development. 2018;40:544–51.
Suraev AS, Todd L, Bowen MT, Allsop DJ, McGregor IS, Ireland C, et al. An Australian nationwide survey on medicinal cannabis use for epilepsy: history of antiepileptic drug treatment predicts medicinal cannabis use. Epilepsy Behav. 2017;70:334–40.
Suraev A, Lintzeris N, Stuart J, Kevin RC, Blackburn R, Richards E, et al. Composition and use of cannabis extracts for childhood epilepsy in the Australian community. Sci Rep. 2018;8:10154.
Devinsky O, Marsh E, Friedman D, Thiele E, Laux L, Sullivan J, et al. Cannabidiol in patients with treatment-resistant epilepsy: an open-label interventional trial. Lancet Neurol. 2016;15:270–8.
• Szaflarski JP, Bebin EM, Comi AM, et al. Long-term safety and treatment effects of cannabidiol in children and adults with treatment-resistant epilepsies: Expanded access program results. Epilepsia 2018;59:1540–1548. This open-label study documents the sustained response to cannabidiol over the duration of exposure.
Szaflarski JP, Bebin E, Cutter GR, et al. Cannabidiol improves seizure frequency and severity and reduces adverse events in an open-label prospective study. Epilepsy Behav. 2018; Accepted; In Press.
Hess EJ, Moody KA, Geffrey AL, Pollack SF, Skirvin LA, Bruno PL, et al. Cannabidiol as a new treatment for drug-resistant epilepsy in tuberous sclerosis complex. Epilepsia. 2016;57:1617–24.
Devinsky O, Verducci C, Thiele EA, Laux LC, Patel AD, Filloux F, et al. Open-label use of Highly* purified CBD (Epidiolex®) in patients with CDKL5 deficiency disorder and Aicardi, Dup15q, and Doose syndromes. Epilepsy Behav. 2018;86:131–7.
Gofshteyn JS, Wilfong A, Devinsky O, Bluvstein J, Charuta J, Ciliberto MA, et al. Cannabidiol as a potential treatment for febrile infection-related epilepsy syndrome (FIRES) in the acute and chronic phases. J Child Neurol. 2017;32:35–40.
McCoy B, Wang L, Zak M, et al. A prospective open-label trial of CBD/THC cannabis oil in Dravet syndrome. Ann Clin Transl Neurol. in print.
Gloss D, Vickrey B. Cannabinoids for epilepsy. Cochrane Database Syst Rev. 2014;3:CD009270.
Devinsky O, Patel AD, Thiele EA, Wong MH, Appleton R, Harden CL, et al. Randomized, dose-ranging safety trial of cannabidiol in Dravet syndrome. Neurology. 2018;90:e1204–11.
• Devinsky O, Patel AD, Cross JH, et al. Effect of cannabidiol on drop seizures in the Lennox-Gastaut syndrome. N Engl J Med. 2018;378:1888–97. RCT of cannabidiol for the treatment of seizures associated with Lennox-Gastaut syndrome
Espay AJ, Norris MM, Eliassen JC, Dwivedi A, Smith MS, Banks C, et al. Placebo effect of medication cost in Parkinson disease: a randomized double-blind study. Neurology. 2015;84:794–802.
Vandrey R, Raber JC, Raber ME, Douglass B, Miller C, Bonn-Miller MO. Cannabinoid dose and label accuracy in edible medical cannabis products. Jama. 2015;313:2491–3.
• Devinsky O, Cross JH, Laux L, et al. Trial of cannabidiol for drug-resistant seizures in the Dravet syndrome. N Engl J Med. 2017;376:2011–20. RCT of cannabidiol for the treatment of seizures associated with Dravet syndrome.
• Thiele EA, Marsh ED, French JA, et al. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome (GWPCARE4): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2018;391:1085–96. RCT of cannabidiol for the treatment of seizures associated with Lennox-Gastaut syndrome.
Geffrey AL, Pollack SF, Bruno PL, Thiele EA. Drug-drug interaction between clobazam and cannabidiol in children with refractory epilepsy. Epilepsia. 2015;56:1246–51.
Jiang R, Yamaori S, Okamoto Y, Yamamoto I, Watanabe K. Cannabidiol is a potent inhibitor of the catalytic activity of cytochrome P450 2C19. Drug Metab Pharmacokinet. 2013;28:332–8.
• Gaston TE, Bebin EM, Cutter GR, Liu Y, Szaflarski JP. Interactions between cannabidiol and commonly used antiepileptic drugs. Epilepsia. 2017;58:1586–92. Detailed analysis of interactions of cannabidiol with common anti-seizure drugs.
Grayson L, Vines B, Nichol K, Szaflarski JP, Program UC. An interaction between warfarin and cannabidiol, a case report. Epilepsy Behav Case Rep. 2018;9:10–1.
Gaston TE, Liu Y, Cutter GR, Bebin E, Grayson LE, Szaflarski JP. Effect of pharmaceutical formulation of cannabidiol (CBD) on seizure frequency and severity does not appear to be dependent on drug-drug interactions with other anti-epileptic drugs. American Academy of Neurology Annual Meeting; 2018 25 April 2018; Los Angeles, CA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr. Szaflarski reports personal fees from GW Pharmaceuticals; grants and personal fees from Serina Therapeutics, Inc., during the conduct of the study; personal fees from SK Life Sciences; personal fees from LivaNova Inc.; personal fees from Lundbeck; personal fees from NeuroPace Inc.; personal fees from Upsher-Smith Laboratories, Inc.; grants and personal fees from SAGE Pharmaceuticals; grants from UCB Pharma; grants from Biogen; and grants from Eisai Inc., outside the submitted work.
Dr. Gaston reports personal fees from GW Pharmaceuticals, during the conduct of the study.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Epilepsy
Rights and permissions
About this article
Cite this article
Gaston, T.E., Szaflarski, J.P. Cannabis for the Treatment of Epilepsy: an Update. Curr Neurol Neurosci Rep 18, 73 (2018). https://doi.org/10.1007/s11910-018-0882-y
Published:
DOI: https://doi.org/10.1007/s11910-018-0882-y