Abstract
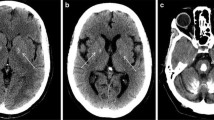
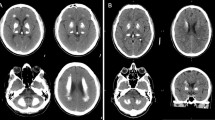
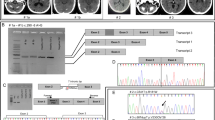
Bilateral accumulation of calcium in the brain, most commonly in the basal ganglia, but also in the cerebellum, thalamus, and brainstem can be inherited in an autosomal dominant fashion and is then referred to as primary familial brain calcifications (PFBC). Clinical manifestations include a spectrum of movement disorders and neuropsychiatric abnormalities. In the past 2 years, 3 genes have been identified to cause PFBC, (ie, SLC20A2, PDGFRB, and PDGFB). SCL20A2 encodes the Type III sodium-dependent inorganic phosphate (Pi) transporter 2 (PiT2) and, when mutated, uptake of Pi is severely impaired likely causing buildup of calcium phosphate. The second identified cause of PFBC is mutations in PDGFRB, which codes for platelet-derived growth factor receptor β (PDGF-Rβ). Interestingly, the third PFBC gene is PDGFB that encodes the ligand of PDGF-Rβ, which is secreted during angiogenesis to recruit pericytes, thereby implying impairment of the blood-brain barrier as a disease mechanism of PFBC.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Baba Y, Broderick DF, Uitti RJ, Hutton ML, Wszolek ZK. Heredofamilial brain calcinosis syndrome. Mayo Clin Proc. 2005;80(5):641–51.
Sobrido MJ, Coppola G, Oliveira J, Hopfer S, Geschwind DH. Primary Familial Brain Calcification. GeneReviews (Internet). Seattle: University of Washington; 2004. p. 1993–2014. This publication is of importance given that it is currently the most comprehensive review of all aspects of PFBC.
Forstl H, Krumm B, Eden S, Kohlmeyer K. Neurological disorders in 166 patients with basal ganglia calcification: a statistical evaluation. J Neurol. 1992;239(1):36–8.
Yamada M, Asano T, Okamoto K, Hayashi Y, Kanematsu M, Hoshi H, et al. High frequency of calcification in basal ganglia on brain computed tomography images in Japanese older adults. Geriatr Gerontol Int. 2013;13(3):706–10.
Fahr T. Idiopathische Verkalkung der Hirngefässe. Zbl Allg Pathol Pathol Anat. 1930;50:129–30.
Klein C, Vieregge P. Fahr's disease—far from a disease. Mov Disord. 1998;13(3):620–1.
Delacour A. Ossification des capillaires du cerveau. Ann Med Psychol. 1850;2:458–61.
Geschwind DH, Loginov M, Stern JM. Identification of a locus on chromosome 14q for idiopathic basal ganglia calcification (Fahr disease). Am J Hum Genet. 1999;65(3):764–72.
Volpato CB, De Grandi A, Buffone E, Facheris M, Gebert U, Schifferle G, et al. 2q37 as a susceptibility locus for idiopathic basal ganglia calcification (IBGC) in a large South Tyrolean family. J Mol Neurosci. 2009;39(3):346–53.
Dai X, Gao Y, Xu Z, Cui X, Liu J, Li Y, et al. Identification of a novel genetic locus on chromosome 8p21.1-q11.23 for idiopathic basal ganglia calcification. Am J Med Genet B Neuropsychiatr Genet. 2010;153B(7):1305–10.
Nicolas G, Pottier C, Maltete D, Coutant S, Rovelet-Lecrux A, Legallic S, et al. Mutation of the PDGFRB gene as a cause of idiopathic basal ganglia calcification. Neurology. 2013;80(2):181–7. This publication is of major importance as it reports for the first time mutations in PDGFRB as a cause of PFBC.
Keller A, Westenberger A, Sobrido MJ, Garcia-Murias M, Domingo A, Sears RL, et al. Mutations in the gene encoding PDGF-B cause brain calcifications in humans and mice. Nat Genet. 2013;45(9):1077–82. This publication is of major importance given that it reports for the first time mutations in PDGFB as a cause of PFBC.
Marras C, Lohmann K, Lang A, Klein C. Fixing the broken system of genetic locus symbols: Parkinson disease and dystonia as examples. Neurology. 2012;78(13):1016–24.
Hsu SC, Sears RL, Lemos RR, Quintans B, Huang A, Spiteri E, et al. Mutations in SLC20A2 are a major cause of familial idiopathic basal ganglia calcification. Neurogenetics. 2013;14(1):11–22. This publication is of importance since it is the most extensive screening study of the SLC20A2 gene in patients with a positive family history published to date. It also reports that mutations in SLC20A2 are the cause of PFBC in the family originaly thought to be linked to the IBGC1 locus.
Wang C, Li Y, Shi L, Ren J, Patti M, Wang T, et al. Mutations in SLC20A2 link familial idiopathic basal ganglia calcification with phosphate homeostasis. Nat Genet. 2012;44(3):254–6. This publication is of major importance given that it reports for the first time mutations in SLC20A2 as a cause of PFBC.
Yamada M, Tanaka M, Takagi M, Kobayashi S, Taguchi Y, Takashima S, et al. Evaluation of SLC20A2 mutations that cause idiopathic basal ganglia calcification in Japan. Neurology. 2014;82(8):705–12. This publication is of importance because it is the most extensive screening study of the SLC20A2 gene in the Japanese population of familial and sporadic PFBC cases published to date.
Bottger P, Pedersen L. Mapping of the minimal inorganic phosphate transporting unit of human PiT2 suggests a structure universal to PiT-related proteins from all kingdoms of life. BMC Biochem. 2011;12:21.
Baker M, Strongosky AJ, Sanchez-Contreras MY, Yang S, Ferguson W, Calne DB, et al. SLC20A2 and THAP1 deletion in familial basal ganglia calcification with dystonia. Neurogenetics. 2014;15(1):23–30. This publication is of importance given that it reports the first deletion that involves the entire SLC20A2 gene, as well as the entire THAP1 gene.
Jensen N, Schroder HD, Hejbol EK, Fuchtbauer EM, de Oliveira JR, Pedersen L. Loss of function of Slc20a2 associated with familial idiopathic Basal Ganglia calcification in humans causes brain calcifications in mice. J Mol Neurosci. 2013;51(3):994–9.
Kobayashi S, Yamadori I, Miki H, Ohmori M. Idiopathic nonarteriosclerotic cerebral calcification (Fahr's disease): an electron microscopic study. Acta Neuropathol. 1987;73(1):62–6.
Nicolas G, Pottier C, Charbonnier C, Guyant-Marechal L, Le Ber I, Pariente J, et al. Phenotypic spectrum of probable and genetically-confirmed idiopathic basal ganglia calcification. Brain. 2013;136(Pt 11):3395–407. This publication is of importance given that it provides the first comprehensive overview of clinical features of patients affected by definite (caused by mutations in SLC20A2 and PDGFRB) and probable PFBC.
Sanchez-Contreras M, Baker MC, Finch NA, Nicholson A, Wojtas A, Wszolek ZK, et al. Genetic Screening and Functional Characterization of PDGFRB Mutations Associated With Basal Ganglia Calcification of Unknown Etiology. Hum Mutat. 2014. doi:10.1002/humu.22582. This publication is of importance as it reports the first study of pathogenic mechanisms of the mutations in the PDGF-Rβ gene causing PFBC.
Andrae J, Gallini R, Betsholtz C. Role of platelet-derived growth factors in physiology and medicine. Genes Dev. 2008;22(10):1276–312.
Martignetti JA, Tian L, Li D, Ramirez MC, Camacho-Vanegas O, Camacho SC, et al. Mutations in PDGFRB cause autosomal-dominant infantile myofibromatosis. Am J Hum Genet. 2013;92(6):1001–7.
Cheung YH, Gayden T, Campeau PM, LeDuc CA, Russo D, Nguyen VH, et al. A recurrent PDGFRB mutation causes familial infantile myofibromatosis. Am J Hum Genet. 2013;92(6):996–1000.
Nicolas G, Rovelet-Lecrux A, Pottier C, Martinaud O, Wallon D, Vernier L, et al. PDGFB partial deletion: a new, rare mechanism causing brain calcification with leukoencephalopathy. J Mol Neurosci. 2014;53(2):171–5.
Nicolas G, Jacquin A, Thauvin-Robinet C, Rovelet-Lecrux A, Rouaud O, Pottier C, et al. A de novo nonsense PDGFB mutation causing idiopathic basal ganglia calcification with laryngeal dystonia. Eur J Hum Genet. 2014. doi:10.1038/ejhg.2014.9
Armulik A, Genove G, Betsholtz C. Pericytes: developmental, physiological, and pathological perspectives, problems, and promises. Dev Cell. 2011;21(2):193–215.
Gaengel K, Genove G, Armulik A, Betsholtz C. Endothelial-mural cell signaling in vascular development and angiogenesis. Arterioscler Thromb Vasc Biol. 2009;29(5):630–8.
Armulik A, Genove G, Mae M, Nisancioglu MH, Wallgard E, Niaudet C, et al. Pericytes regulate the blood-brain barrier. Nature. 2010;468(7323):557–61.
Manyam BV, Walters AS, Narla KR. Bilateral striopallidodentate calcinosis: clinical characteristics of patients seen in a registry. Mov Disord. 2001;16(2):258–64.
Larsen TA, Dunn HG, Jan JE, Calne DB. Dystonia and calcification of the basal ganglia. Neurology. 1985;35(4):533–7.
Zhang Y, Guo X, Wu A. Association between a novel mutation in SLC20A2 and familial idiopathic basal ganglia calcification. PLoS One. 2013;8(2):e57060.
Brodaty H, Mitchell P, Luscombe G, Kwok JJ, Badenhop RF, McKenzie R, et al. Familial idiopathic basal ganglia calcification (Fahr's disease) without neurological, cognitive and psychiatric symptoms is not linked to the IBGC1 locus on chromosome 14q. Hum Genet. 2002;110(1):8–14.
Oliveira JR, Spiteri E, Sobrido MJ, Hopfer S, Klepper J, Voit T, et al. Genetic heterogeneity in familial idiopathic basal ganglia calcification (Fahr disease). Neurology. 2004;63(11):2165–7.
Chen WJ, Yao XP, Zhang QJ, Ni W, He J, Li HF, et al. Novel SLC20A2 mutations identified in southern Chinese patients with idiopathic basal ganglia calcification. Gene. 2013;529(1):159–62.
Kasuga K, Konno T, Saito K, Ishihara A, Nishizawa M, Ikeuchi T. A Japanese family with idiopathic basal ganglia calcification with novel SLC20A2 mutation presenting with late-onset hallucination and delusion. J Neurol. 2014;261(1):242–4.
Zhu M, Zhu X, Wan H, Hong D. Familial IBGC caused by SLC20A2 mutation presenting as paroxysmal kinesigenic dyskinesia. Parkinsonism Relat Disord. 2014;20(3):353–4.
Lemos RR, Oliveira MF, Oliveira JR. Reporting a new mutation at the SLC20A2 gene in familial idiopathic basal ganglia calcification. Eur J Neurol. 2013;20(3):e43–4.
Schottlaender L. Interesting clinical features associated with mutations in the SLC20A2 gene. Eur J Neurol. 2013;19:40.
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
Ana Westenberger has received an intramural grant from the Medical Genetics Priority Program from the University of Lübeck, Germany and a research grant from the Fritz Thyssen Foundation. Christine Klein has received an intramural grant from the Medical Genetics Priority Program from the University of Lübeck, Germany. Dr. Klein also is a board member of the Bachmann Strauss Dystonia and Parkinson’s Disease Foundation; SAB of VIB University of Leuven. She also has received consultancy fees as a medical advisor to Centogene. Dr. Klein has also received a Career development award from the Hermann and Lilly Schilling foundation, DFG, BMBF, EU, and honoraria for speaking at the Annual Meeting of the American Academy of Neurology.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Genetics
Rights and permissions
About this article
Cite this article
Westenberger, A., Klein, C. The Genetics of Primary Familial Brain Calcifications. Curr Neurol Neurosci Rep 14, 490 (2014). https://doi.org/10.1007/s11910-014-0490-4
Published:
DOI: https://doi.org/10.1007/s11910-014-0490-4