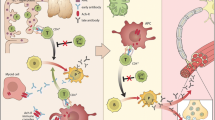
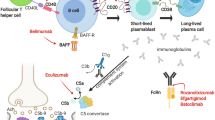
Abstract
Myasthenia gravis (MG) is an antibody-mediated, neuromuscular transmission disorder, which ranges in clinical manifestations of ocular myasthenia that may be visually disabling to myasthenic crisis with patients suffering life-threatening respiratory insufficiency. MG also has pathophysiologic subgroups based on presence or absence of acetylcholine receptor or muscle-specific kinase antibodies and presence of thymoma. Cholinesterase inhibitors partially improve weakness, but the vast majority of patients require therapies that moderate the autoimmune attack. Mortality of MG has been reduced over the last century, but adverse effects of treatment compromise patient care and rigorous evidence to guide the clinician are lacking.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Vincent A: Immunology of disorders of neuromuscular transmission. Acta Neurol Scand Suppl 2006, 183:1–7.
• Leite MI, Jacob S, Viegas S, et al.: IgG1 antibodies to acetylcholine receptors in ‘seronegative’ myasthenia gravis. Brain 2008, 131:1940–1952. This article indicates that even patients who do not have antibodies directed against the AChR or MuSK likely have low levels of serum antibodies that bind the AChR. Some of these seronegative patients still may have antibodies directed at other proteins.
Bae JS, Go SM, Kim BJ: Clinical predictors of steroid-induced exacerbation in myasthenia gravis. J Clin Neurosci 2006, 13:1006–1010.
Palace J, Newsom-Davis J, Lecky B: A randomized double-blind trial of prednisolone alone or with azathioprine in myasthenia gravis. Myasthenia Gravis Study Group. Neurology 1998, 50:1778–1783.
Howard FM Jr, Duane DD, Lambert EH, Daube JR: Alternate-day prednisone: preliminary report of a double-blind controlled study. Ann N Y Acad Sci 1976, 274:596–607.
A randomised clinical trial comparing prednisone and azathioprine in myasthenia gravis. Results of the second interim analysis. Myasthenia Gravis Clinical Study Group [no authors listed]. J Neurol Neurosurg Psychiatry 1993, 56:1157–1163.
Juel VC, Massey JM: Autoimmune myasthenia gravis: recommendations for treatment and immunologic modulation. Curr Treat Options Neurol 2005, 7:3–14.
Bromberg MB, Wald JJ, Forshew DA, et al.: Randomized trial of azathioprine or prednisone for initial immunosuppressive treatment of myasthenia gravis. J Neurol Sci 1997, 150:59–62.
Tindall RS, Phillips JT, Rollins JA, et al.: A clinical therapeutic trial of cyclosporine in myasthenia gravis. Ann N Y Acad Sci 1993, 681:539–551.
Tindall RS, Rollins JA, Phillips JT, et al.: Preliminary results of a double-blind, randomized, placebo-controlled trial of cyclosporine in myasthenia gravis. N Engl J Med 1987, 316:719–724.
Ciafaloni E, Nikhar NK, Massey JM, Sanders DB: Retrospective analysis of the use of cyclosporine in myasthenia gravis. Neurology 2000, 55:448–450.
Allison AC, Eugui EM: Mycophenolate mofetil and its mechanisms of action. Immunopharmacology 2000, 47:85–118.
Ciafaloni E, Massey JM, Tucker-Lipscomb B, Sanders DB: Mycophenolate mofetil for myasthenia gravis: an open-label pilot study. Neurology 2001, 56:97–99.
Meriggioli MN, Rowin J, Richman JG, Leurgans S: Mycophenolate mofetil for myasthenia gravis: a double-blind, placebo-controlled pilot study. Ann N Y Acad Sci 2003, 998:494–499.
• Sanders DB, Hart IK, Mantegazza R, et al.: An international, phase III, randomized trial of mycophenolate mofetil in myasthenia gravis. Neurology 2008, 71:400–406. This study was a well-performed, randomized, placebo-controlled trial that showed no benefit of mycophenolate and prednisone compared with prednisone alone. The study likely did not have long enough periods of study.
• Muscle Study Group: A trial of mycophenolate mofetil with prednisone as initial immunotherapy in myasthenia gravis. Neurology 2008, 71:394–399. This study was a well-performed, randomized, placebo-controlled trial that showed no benefit of mycophenolate and prednisone compared with prednisone alone. The study likely did not have long enough periods of study.
Furukawa Y, Yoshikawa H, Iwasa K, Yamada M: Clinical efficacy and cytokine network-modulating effects of tacrolimus in myasthenia gravis. J Neuroimmunol 2008, 195:108–115.
Ponseti JM, Gamez J, Azem J, et al.: Post-thymectomy combined treatment of prednisone and tacrolimus versus prednisone alone for consolidation of complete stable remission in patients with myasthenia gravis: a non-randomized, non-controlled study. Curr Med Res Opin 2007, 23:1269–1278.
Ponseti JM, Azem J, Fort JM, et al.: Long-term results of tacrolimus in cyclosporine- and prednisone-dependent myasthenia gravis. Neurology 2005, 64:1641–1643.
Nagane Y, Utsugisawa K, Obara D, et al.: Efficacy of low-dose FK506 in the treatment of Myasthenia gravis—a randomized pilot study. Eur Neurol 2005, 53:146–150.
Lewis RA, Lisak RP: “Rebooting” the immune system with cyclophosphamide: taking risks for a “cure”? Ann Neurol 2003, 53:7–9.
Drachman DB, Brodsky RA: High-dose therapy for autoimmune neurologic diseases. Curr Opin Oncol 2005, 17:83–88.
Lin PT, Martin BA, Weinacker AB, So YT: High-dose cyclophosphamide in refractory myasthenia gravis with MuSK antibodies. Muscle Nerve 2006, 33:433–435.
De Feo LG, Schottlender J, Martelli NA, Molfino NA: Use of intravenous pulsed cyclophosphamide in severe, generalized myasthenia gravis. Muscle Nerve 2002, 26:31–36.
Baek WS, Bashey A, Sheean GL: Complete remission induced by rituximab in refractory, seronegative, muscle-specific, kinase-positive myasthenia gravis. J Neurol Neurosurg Psychiatry 2007, 78:771.
Hain B, Jordan K, Deschauer M, Zierz S: Successful treatment of MuSK antibody-positive myasthenia gravis with rituximab. Muscle Nerve 2006, 33:575–580.
Gajdos P, Chevret S, Clair B, et al.: Clinical trial of plasma exchange and high-dose intravenous immunoglobulin in myasthenia gravis. Myasthenia Gravis Clinical Study Group. Ann Neurol 1997, 41:789–796.
Ronager J, Ravnborg M, Hermansen I, Vorstrup S: Immunoglobulin treatment versus plasma exchange in patients with chronic moderate to severe myasthenia gravis. Artif Organs 2001, 25:967–973.
Bedlack RS, Sanders DB: Steroid treatment for myasthenia gravis: steroids have an important role. Muscle Nerve 2002, 25:117–121.
Gajdos P, Tranchant C, Clair B, et al.: Treatment of myasthenia gravis exacerbation with intravenous immunoglobulin: a randomized double-blind clinical trial. Arch Neurol 2005, 62:1689–1693.
• Zinman L, Ng E, Bril V: IV immunoglobulin in patients with myasthenia gravis: a randomized controlled trial. Neurology 2007, 68:837–841. The study is among the few controlled trials in MG. It demonstrates the efficacy of IVIG in the treatment of exacerbations of MG.
Qureshi AI, Choudhry MA, Akbar MS, et al.: Plasma exchange versus intravenous immunoglobulin treatment in myasthenic crisis. Neurology 1999, 52:629–632.
Gajdos P, Simon N, de Rohan-Chabot P, et al.: [Long-term effects of plasma exchange in myasthenia. Results of a randomized study] [in French]. Presse Med 1983, 12:939–942.
Gajdos P, Chevret S, Toyka K: Plasma exchange for myasthenia gravis. Cochrane Database Syst Rev 2002, (4):CD002275.
Gronseth GS, Barohn RJ: Practice parameter: thymectomy for autoimmune myasthenia gravis (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2000, 55:7–15.
• Newsom-Davis J, Cutter G, Wolfe GI, et al.: Status of the thymectomy trial for nonthymomatous myasthenia gravis patients receiving prednisone. Ann N Y Acad Sci 2008, 1132:344–347. The article describes the first randomized, single-blinded, controlled trial of thymectomy for patients with MG. The study is about two-thirds completed at the time of publication of this review.
Magee MJ, Mack MJ: Surgical approaches to the thymus in patients with myasthenia gravis. Thorac Surg Clin 2009, 19:83–89.
Manlulu A, Lee TW, Wan I, et al.: Video-assisted thoracic surgery thymectomy for nonthymomatous myasthenia gravis. Chest 2005, 128:3454–3460.
Benatar M, Kaminski HJ: Evidence report: the medical treatment of ocular myasthenia (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2007, 68:2144–2149.
Sommer N, Sigg B, Melms A, et al.: Ocular myasthenia gravis: response to long-term immunosuppressive treatment. J Neurol Neurosurg Psychiatry 1997, 62:156–162.
Kupersmith MJ, Ying G: Ocular motor dysfunction and ptosis in ocular myasthenia gravis: effects of treatment. Br J Ophthalmol 2005, 89:1330–1334.
Mee J, Paine M, Byrne E, et al.: Immunotherapy of ocular myasthenia gravis reduces conversion to generalized myasthenia gravis. J Neuroophthalmol 2003, 23:251–255.
McGrogan A, Sneddon S, de Vries CS: The incidence of myasthenia gravis: a systematic literature review. Neuroepidemiology 2010, 34:171–183.
Andrews PI: Autoimmune myasthenia gravis in childhood. Semin Neurol 2004, 24:101–110.
• Evoli A: Acquired myasthenia gravis in childhood. Curr Opin Neurol 2010, 23:536–540. This is an outstanding review of the challenges faced in caring for pediatric patients with MG. The author is an outstanding clinical scientist with a wealth of clinical experience.
Hoch W, McConville J, Helms S, et al.: Auto-antibodies to the receptor tyrosine kinase MuSK in patients with myasthenia gravis without acetylcholine receptor antibodies. Nat Med 2001, 7:365–368.
Pasnoor M, Wolfe GI, Nations S, et al.: Clinical findings in MuSK-antibody positive myasthenia gravis: a U.S. experience. Muscle Nerve 2010, 41:370–374.
Evoli A, Bianchi MR, Riso R, et al.: Response to therapy in myasthenia gravis with anti-MuSK antibodies. Ann N Y Acad Sci 2008, 1132:76–83.
Guptill JT, Sanders DB: Update on muscle-specific tyrosine kinase antibody positive myasthenia gravis. Curr Opin Neurol 2010, 23:530–535.
Conti-Fine BM, Milani M, Kaminski HJ: Myasthenia gravis: past, present, and future. J Clin Invest 2006, 116:2843–2854.
Acknowledgment
H.J. Kaminski receives support from grants from the National Institutes of Health R01 EY14837 and R01 NS42685.
Disclosures
Conflicts of interest: V. Kumar: none; H.J. Kaminski has performed consultations for GlaxoSmithKline, Bayhill Therapeutics, Cytokinetics, and Varleigh Limited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, V., Kaminski, H.J. Treatment of Myasthenia Gravis. Curr Neurol Neurosci Rep 11, 89–96 (2011). https://doi.org/10.1007/s11910-010-0151-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11910-010-0151-1