Abstract
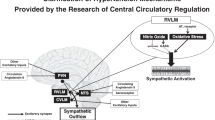
There is evidence of sympathetic overdrive in a significant proportion of patients with essential hypertension and an animal model of the condition, the spontaneously hypertensive rat (SHR). The reasons for this remain elusive. However, there is also evidence of narrowing of the arteries supplying the brainstem in the SHR and hypertensive humans. In this review, we discuss the possible role of brainstem hypoperfusion in driving increased sympathetic activity and hypertension.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Mann SJ. Neurogenic essential hypertension revisited: the case for increased clinical and research attention. Am J Hypertens. 2003;16(10):881–8.
Plump A. Accelerating the pulse of cardiovascular R&D. Nat Rev Drug Discov. 2010;9(11):823–4.
Sundlof G, Wallin BG. Human muscle nerve sympathetic activity at rest. Relationship to blood pressure and age. J Physiol. 1978;274:621–37.
Grassi G, Colombo M, Seravalle G, et al. Dissociation between muscle and skin sympathetic nerve activity in essential hypertension, obesity, and congestive heart failure. Hypertension. 1998;31(1):64–7.
Grassi G. Sympathetic and baroreflex function in hypertension: implications for current and new drugs. Curr Pharm Des. 2004;10(29):3579–89.
Smith PA, Graham LN, Mackintosh AF, et al. Relationship between central sympathetic activity and stages of human hypertension. Am J Hypertens. 2004;17(3):217–22.
Wu JS, Lu FH, Yang YC, et al. Epidemiological study on the effect of pre-hypertension and family history of hypertension on cardiac autonomic function. J Am Coll Cardiol. 2008;51(19):1896–901.
Maver J, Strucl M, Accetto R. Autonomic nervous system activity in normotensive subjects with a family history of hypertension. Clin Auton Res. 2004;14(6):369–75.
Converse Jr RL, Jacobsen TN, Toto RD. Sympathetic overactivity in patients with chronic renal failure. N Engl J Med. 1992;327(27):1912–8.
Leimbach Jr WN, Wallin BG, Victor RG, et al. Direct evidence from intraneural recordings for increased central sympathetic outflow in patients with heart failure. Circulation. 1986;73(5):913–9.
Barretto AC, Santos AC, Munhoz R, et al. Increased muscle sympathetic nerve activity predicts mortality in heart failure patients. Int J Cardiol. 2009;135(3):302–7.
Zoccali C, Mallamaci F, Parlongo S, et al. Plasma norepinephrine predicts survival and incident cardiovascular events in patients with end-stage renal disease. Circulation. 2002;105(11):1354–9.
Okamoto K, Aoki K. Development of a strain of spontaneously hypertensive rats. Jpn Circ J. 1963;27:282–93.
Pinto YM, Paul M, Ganten D. Lessons from rat models of hypertension: from Goldblatt to genetic engineering. Cardiovasc Res. 1998;39(1):77–88.
Lundin S, Ricksten SE, Thoren P. Renal sympathetic activity in spontaneously hypertensive rats and normotensive controls, as studied by three different methods. Acta Physiol Scand. 1984;120(2):265–72.
•• Simms AE, Paton JF, Pickering AE, et al. Amplified respiratory-sympathetic coupling in the spontaneously hypertensive rat: does it contribute to hypertension? J Physiol. 2009;587(Pt 3):597–610. This study showed enhanced coupling of breathing with SNA generation in SHR versus normotensive rats, and that breathing-related SNA is a major contributor to total peripheral resistance in SHR.
Cabassi A, Vinci S, Calzolari M, et al. Regional sympathetic activity in pre-hypertensive phase of spontaneously hypertensive rats. Life Sci. 1998;62(12):1111–8.
Harrap SB, Van der Merwe WM, Griffin SA, et al. Brief angiotensin converting enzyme inhibitor treatment in young spontaneously hypertensive rats reduces blood pressure long-term. Hypertension. 1990;16(6):603–14.
Dickinson CJ, Thomson AD. A post mortem study of the main cerebral arteries with special reference to their possible role in blood pressure regulation. Clin Sci. 1960;19:513–38.
• Paton JF, Dickinson CJ, Mitchell G. Harvey Cushing and the regulation of blood pressure in giraffe, rat and man: introducing ‘Cushing’s mechanism’. Exp Physiol. 2009;94(1):11–7. This comprehensive review discusses the important distinction between Cushing’s response and Cushing’s mechanism.
• Dickinson CJ, Paton JF: Cerebrovascular Hypertension: Book Guild Publising Ltd; 2012. This book contains a wealth of information supporting the hypothesis of Cushing’s mechanism driving hypertension and is a rich bibliographical resource.
Jannetta PJ, Gendell HM. Clinical observations on etiology of essential hypertension. Surg Forum. 1979;30:431–2.
Thuerl C, Rump LC, Otto M, et al. Neurovascular contact of the brain stem in hypertensive and normotensive subjects: MR findings and clinical significance. AJNR Am J Neuroradiol. 2001;22(3):476–80.
Zizka J, Ceral J, Elias P, et al. Vascular compression of rostral medulla oblongata: prospective MR imaging study in hypertensive and normotensive subjects. Radiology. 2004;230(1):65–9.
Cushing H. Concerning a definitive regulatory mechanism of the vaso-motor centre which controls blood pressure during cerebral compression. Bull Johns Hopk Hosp. 1901;12:290–2.
Kety SS, Skenkin HA, Schmidt CF. The effects of increased intracranial pressure on cerebral circulatory functions in man. J Clin Invest. 1948;27(4):493–9.
Kalmar AF, Van Aken J, Caemaert J, et al. Value of Cushing reflex as warning sign for brain ischaemia during neuroendoscopy. Br J Anaesth. 2005;94(6):791–9.
Dampney RA, Kumada M, Reis DJ. Central neural mechanisms of the cerebral ischemic response. Characterization, effect of brainstem and cranial nerve transections, and simulation by electrical stimulation of restricted regions of medulla oblongata in rabbit. Circ Res. 1979;45(1):48–62.
Braga VA, Paton JF, Machado BH. Ischaemia-induced sympathoexcitation in spinalyzed rats. Neurosci Lett. 2007;415(1):73–6.
Reis DJ. The brain and hypertension: reflections on 35 years of inquiry into the neurobiology of the circulation. Circulation. 1984;70(5 Pt 2):III31–45.
Phillips AM, Jardine DL, Parkin PJ, et al. Brain stem stroke causing baroreflex failure and paroxysmal hypertension. Stroke; a journal of cerebral circulation. 2000;31(8):1997–2001.
•• Waki H, Bhuiyan ME, Gouraud SS, et al. Acute reductions in blood flow restricted to the dorsomedial medulla induce a pressor response in rats. J Hypertens. 2011;29(8):1536–45. This is a novel experimental animal model that provides strong supporting evidence that focally induced brainstem ischemia triggers a strong sympathoexcitatory pressor response in rats.
Paton JF, Guild S, Malpas S et al: Is cerebral blood flow a determinant of a long term arterial pressure set-point in the rat? Proc Physiol Soc 23 2011.
Lee RM. Morphology of cerebral arteries. Pharmacol Ther. 1995;66(1):149–73.
Duvernoy HM. Human Brainstem Vessels. Berlin: Springer-Verlag; 1978.
Mayhan WG, Faraci FM, Heistad DD. Disruption of the blood–brain barrier in cerebrum and brain stem during acute hypertension. Am J Physiol. 1986;251(6 Pt 2):H1171–1175.
Hamel E. Perivascular nerves and the regulation of cerebrovascular tone. J Appl Physiol. 2006;100(3):1059–64.
Brian Jr JE. Carbon dioxide and the cerebral circulation. Anesthesiology. 1998;88(5):1365–86.
•• Leoni RF, Paiva FF, Henning EC, et al. Magnetic resonance imaging quantification of regional cerebral blood flow and cerebrovascular reactivity to carbon dioxide in normotensive and hypertensive rats. NeuroImage. 2011;58(1):75–81. Using state-of-the-art imaging techniques, this paper demonstrates an inability of the PHSHR cerebral vasculature to vasodilate in response to hypercapnia. This is in keeping with the idea of a limited reserve in cerebral blood flow in SHR and potentially increasing the risk of cerebral and brainstem hypoperfusion.
Paulson OB, Strandgaard S, Edvinsson L. Cerebral autoregulation. Cerebrovasc Brain Metab Rev. 1990;2(2):161–92.
Strandgaard S. Autoregulation of cerebral circulation in hypertension. Acta Neurol Scand Suppl. 1978;66:1–82.
Fujishima M, Sadoshima S, Ogata J, et al. Autoregulation of cerebral blood flow in young and aged spontaneously hypertensive rats (SHR). Gerontology. 1984;30(1):30–6.
Paton JF, Waki H, Abdala AP, et al. Vascular-brain signaling in hypertension: role of angiotensin II and nitric oxide. Current hypertension reports. 2007;9(3):242–7.
• Cates MJ, Steed PW, Abdala AP, et al. Elevated vertebrobasilar artery resistance in neonatal spontaneously hypertensive rats. J Appl Physiol. 2011;111(1):149–56. This article provides evidence that increased vertebrobasilar resistance precedes hypertension in the SHR and that the juvenile SHR brainstem is more sensitive to reduced blood flow, in terms of a sympathoexcitatory response, prior to the onset of hypertension.
Eccleston-Joyner CA, Gray SD. Arterial hypertrophy in the fetal and neonatal spontaneously hypertensive rat. Hypertension. 1988;12(5):513–8.
Lee RM. Vascular changes at the prehypertensive phase in the mesenteric arteries from spontaneously hypertensive rats. Blood Vessels. 1985;22(3):105–26.
Smeda JS, Lee RM, Forrest JB. Prenatal and postnatal hydralazine treatment does not prevent renal vessel wall thickening in SHR despite the absence of hypertension. Circ Res. 1988;63(3):534–42.
Lee RM, Triggle CR, Cheung DW, et al. Structural and functional consequence of neonatal sympathectomy on the blood vessels of spontaneously hypertensive rats. Hypertension. 1987;10(3):328–38.
Ibayashi S, Ogata J, Sadoshima S, et al. The effect of long-term antihypertensive treatment on medial hypertrophy of cerebral arteries in spontaneously hypertensive rats. Stroke; a journal of cerebral circulation. 1986;17(3):515–9.
Harrap SB. Angiotensin converting enzyme inhibitors, regional vascular hemodynamics, and the development and prevention of experimental genetic hypertension. Am J Hypertens. 1991;4(3 Pt 2):212S–6S.
Jablonskis LT, Rogers PF, Lungershausen YK, et al. Chronic central administration of enalaprilat lowers blood pressure in stroke-prone spontaneously hypertensive rats. J Auton Nerv Syst. 1992;39(2):119–26.
Ito S, Komatsu K, Tsukamoto K, et al. Ventrolateral medulla AT1 receptors support blood pressure in hypertensive rats. Hypertension. 2002;40(4):552–9.
•• Chang HH, Lee YC, Chen MF et al: Sympathetic activation increases basilar arterial blood flow in normotensive but not hypertensive rats. Am J Physiol Heart Circ Physiol 2011. This is a very interesting paper showing that sympathetic activation increases brainstem blood flow in normotensive rats, but that this process is impaired in SHRs. If our hypothesis is correct, then this process in normotensives may act as an important negative feedback loop to prevent excessive sympathoexcitation. An impairment of this mechanism in SHRs is also in keeping with the hypothesis that brainstem perfusion can only be maintained at the expense of systemic hypertension.
Pickering TG, Harshfield GA, Kleinert HD, et al. Blood pressure during normal daily activities, sleep, and exercise. Comparison of values in normal and hypertensive subjects. JAMA. 1982;247(7):992–6.
Yao H, Sadoshima S, Okada Y, et al. Hindbrain ischemia produced by bilateral vertebral artery occlusion and moderate hypotension in spontaneously hypertensive rats. Angiology. 1990;41(10):848–54.
Potts JT, Shaohua Y, Paton JF: Nucleus tractus solitarii (NTS) blood flow and oxygenation are compromised in spontaneously hypertensive rats (SHR). FASEB 2009, 23 (meeting abstract supplement):959.953.
•• Schierling W, Troidl K, Apfelbeck H, et al. Cerebral arteriogenesis is enhanced by pharmacological as well as fluid-shear-stress activation of the Trpv4 calcium channel. European journal of vascular and endovascular surgery: the official journal of the European Society for Vascular Surgery. 2011;41(5):589–96. This study begins to elucidate the mechanisms by which increased shear stress triggers cerebral arteriogenesis, a process likely to be of significant interest in future pharmacologic research.
• de Groot D, Pasterkamp G, Hoefer IE. Cardiovascular risk factors and collateral artery formation. Eur J Clin Invest. 2009;39(12):1036–47. This intruiging study links cardiovascular disease with an impaired ability to undergo arteriogenesis. It would be of particular interest to know whether impaired arteriogenesis within the brainstem is linked to elevated SNA and hypertension.
Pickering AE, Paton JF. A decerebrate, artificially-perfused in situ preparation of rat: utility for the study of autonomic and nociceptive processing. J Neurosci Methods. 2006;155(2):260–71.
Dickinson CJ. Cerebral oxidative metabolism in essential hypertension: a meta-analysis. J Hypertens. 1995;13(6):653–8.
Rodbard S, Stone W. Pressor mechanisms induced by intracranial compression. Circulation. 1955;12(5):883–90.
Osborn JW. Hypothesis: set-points and long-term control of arterial pressure. A theoretical argument for a long-term arterial pressure control system in the brain rather than the kidney. Clin Exp Pharmacol Physiol. 2005;32(5–6):384–93.
Powell FL, Kim BC, Johnson SR, et al. Oxygen sensing in the brain–invited article. Adv Exp Med Biol. 2009;648:369–76.
Neubauer JA, Sunderram J. Oxygen-sensing neurons in the central nervous system. J Appl Physiol. 2004;96(1):367–74.
Adapala RK, Talasila PK, Bratz IN, et al. PKCalpha mediates acetylcholine-induced activation of TRPV4-dependent calcium influx in endothelial cells. Am J Physiol Heart Circ Physiol. 2011;301(3):H757–765.
Burnstock G. Purinergic mechanosensory transduction and visceral pain. Mol Pain. 2009;5:69.
Chae CU, Lee RT, Rifai N, et al. Blood pressure and inflammation in apparently healthy men. Hypertension. 2001;38(3):399–403.
Engstrom G, Janzon L, Berglund G, et al. Blood pressure increase and incidence of hypertension in relation to inflammation-sensitive plasma proteins. Arterioscler Thromb Vasc Biol. 2002;22(12):2054–8.
Waki H, Gouraud SS, Maeda M, et al. Evidence of specific inflammatory condition in nucleus tractus solitarii of spontaneously hypertensive rats. Exp Physiol. 2010;95(5):595–600.
Waki H, Liu B, Miyake M, et al. Junctional adhesion molecule-1 is upregulated in spontaneously hypertensive rats: evidence for a prohypertensive role within the brain stem. Hypertension. 2007;49(6):1321–7.
Ostermann G, Fraemohs L, Baltus T, et al. Involvement of JAM-A in mononuclear cell recruitment on inflamed or atherosclerotic endothelium: inhibition by soluble JAM-A. Arterioscler Thromb Vasc Biol. 2005;25(4):729–35.
Yu Y, Zhang ZH, Wei SG, et al. Central gene transfer of interleukin-10 reduces hypothalamic inflammation and evidence of heart failure in rats after myocardial infarction. Circ Res. 2007;101(3):304–12.
Kang YM, He RL, Yang LM, et al. Brain tumour necrosis factor-alpha modulates neurotransmitters in hypothalamic paraventricular nucleus in heart failure. Cardiovasc Res. 2009;83(4):737–46.
Shi P, Diez-Freire C, Jun JY, et al. Brain microglial cytokines in neurogenic hypertension. Hypertension. 2010;56(2):297–303.
Li H, Gao Y, Qi Y, et al. Macrophage migration inhibitory factor in hypothalamic paraventricular nucleus neurons decreases blood pressure in spontaneously hypertensive rats. FASEB J. 2008;22(9):3175–85.
Fisher JP, Young CN, Fadel PJ. Central sympathetic overactivity: maladies and mechanisms. Auton Neurosci. 2009;148(1–2):5–15.
• Marvar PJ, Thabet SR, Guzik TJ, et al. Central and peripheral mechanisms of T-lymphocyte activation and vascular inflammation produced by angiotensin II-induced hypertension. Circ Res. 2010;107(2):263–70. This paper elegantly demonstrates an interaction between hypertension, central angiotensin II activity, and the immune system.
Kandalam U, Clark MA. Angiotensin II activates JAK2/STAT3 pathway and induces interleukin-6 production in cultured rat brainstem astrocytes. Regul Pept. 2010;159(1–3):110–6.
Manabe S, Okura T, Watanabe S, et al. Effects of angiotensin II receptor blockade with valsartan on pro-inflammatory cytokines in patients with essential hypertension. J Cardiovasc Pharmacol. 2005;46(6):735–9.
Coles B, Fielding CA, Rose-John S, et al. Classic interleukin-6 receptor signaling and interleukin-6 trans-signaling differentially control angiotensin II-dependent hypertension, cardiac signal transducer and activator of transcription-3 activation, and vascular hypertrophy in vivo. Am J Pathol. 2007;171(1):315–25.
Lu Y, Chen J, Yin X, et al. Angiotensin II receptor 1 involved in the central pressor response induced by interleukin-1 beta in the paraventricular nucleus. Neurol Res. 2009;31(4):420–4.
Iadecola C, Alexander M. Cerebral ischemia and inflammation. Curr Opin Neurol. 2001;14(1):89–94.
Soriano SG, Piva S. Central nervous system inflammation. Eur J Anaesthesiol Suppl. 2008;42:154–9.
Marks L, Carswell HV, Peters EE, et al. Characterization of the microglial response to cerebral ischemia in the stroke-prone spontaneously hypertensive rat. Hypertension. 2001;38(1):116–22.
Fisher JP, Paton JF: The sympathetic nervous system and blood pressure in humans: implications for hypertension. J Hum Hypertens 2012. doi: 10.1038/jhh.2011.66. [Epub ahead of print].
Grisk O, Rose HJ, Rettig R. The development of post-transplantation hypertension in recipients of an SHR kidney is independent of reinnervation of the graft. Pflugers Arch. 1999;438(4):502–7.
Grisk O, Kloting I, Exner J, et al. Long-term arterial pressure in spontaneously hypertensive rats is set by the kidney. J Hypertens. 2002;20(1):131–8.
DiBona GF, Kopp UC. Neural control of renal function. Physiol Rev. 1997;77(1):75–197.
Winternitz SR, Katholi RE, Oparil S. Role of the renal sympathetic nerves in the development and maintenance of hypertension in the spontaneously hypertensive rat. J Clin Invest. 1980;66(5):971–8.
Grisk O, Rose HJ, Lorenz G, et al. Sympathetic-renal interaction in chronic arterial pressure control. Am J Physiol Regul Integr Comp Physiol. 2002;283(2):R441–450.
Pilowsky PM, Goodchild AK. Baroreceptor reflex pathways and neurotransmitters: 10 years on. J Hypertens. 2002;20(9):1675–88.
Cowley Jr AW. Long-term control of arterial blood pressure. Physiol Rev. 1992;72(1):231–300.
Osborn JW, Jacob F, Guzman P. A neural set point for the long-term control of arterial pressure: beyond the arterial baroreceptor reflex. Am J Physiol Regul Integr Comp Physiol. 2005;288(4):R846–855.
Guyenet PG. The sympathetic control of blood pressure. Nat Rev Neurosci. 2006;7(5):335–46.
Schreihofer AM, Ito S, Sved AF. Brain stem control of arterial pressure in chronic arterial baroreceptor-denervated rats. Am J Physiol Regul Integr Comp Physiol. 2005;289(6):R1746–1755.
Cisternas JR, Valenti VE, Alves TB, et al. Cardiac baroreflex is already blunted in eight weeks old spontaneously hypertensive rats. Int Arch Med. 2010;3:2.
Acknowledgments
The authors are grateful to the British Heart Foundation and National Institutes of Health (HL033610) for their support of their research.
Dr. Paton was in receipt of a Royal Society Wolfson Research Merit Award.
Dr. Cates has received grant support from the Physiological Society.
Disclosure
No potential conflicts of interest relevant to this article were reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cates, M.J., Dickinson, C.J., Hart, E.C.J. et al. Neurogenic Hypertension and Elevated Vertebrobasilar Arterial Resistance: Is There a Causative Link?. Curr Hypertens Rep 14, 261–269 (2012). https://doi.org/10.1007/s11906-012-0267-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11906-012-0267-6