Abstract
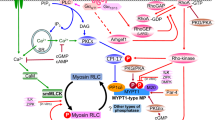
A variety of contractile agonists trigger activation of the small GTPase RhoA. An important target of activated RhoA in smooth muscle is Rho-associated kinase (ROK), one of the downstream targets that is the myosin binding subunit (MYPT1) of myosin light chain phosphatase (MLCP). Phosphorylation of MYPT1 at T695 by activated ROK results in a decrease in phosphatase activity of MLCP and an increase in myosin light chain (LC20) phosphorylation catalyzed by Ca2+/calmodulin-dependent myosin light chain kinase and/or a distinct Ca2+-independent kinase. LC20 phosphorylation in turn triggers cross-bridge cycling and force development. ROK also phosphorylates the cytosolic protein CPI-17 (at T38), which thereby becomes a potent inhibitor of MLCP. The RhoA/ROK pathway has been implicated in the tonic phase of force maintenance in response to various agonists, with no evident role in the phasic response, suggesting this pathway as a potential target for antihypertensive therapy. Indeed, ROK inhibitors restore normal blood pressure in several rat hypertensive models.
Similar content being viewed by others
References and Recommended Reading
Mita M, Walsh MP: a1-Adrenoceptor-mediated phosphorylation of myosin in rat tail arterial smooth muscle. Biochem J 1997, 327:669–674.
Buus CL, Aalkjær C, Nilsson H, et al.: Mechanisms of Ca2+ sensitization of force production by noradrenaline in rat mesenteric small arteries. J Physiol 1998, 510:577–590.
Swärd K, Dreja K, Susnjar M, et al.: Inhibition of Rho-associated kinase blocks agonist-induced Ca2+ sensitization of myosin phosphorylation and force in guinea-pig ileum. J Physiol 2000, 522:33–49.
Nobe K, Paul RJ: Distinct pathways of Ca2+ sensitization in porcine coronary artery. Effects of Rho-related kinase and protein kinase C inhibition on force and intracellular Ca2+. Circ Res 2001, 88:1283–1290.
Somlyo AP, Somlyo AV: Signal transduction by G-proteins, Rho-kinase and protein phosphatase to smooth muscle and non-muscle myosin II. J Physiol 2000, 522:177–185.
Kamm KE, Stull JT: Dedicated myosin light chain kinases with diverse cellular functions. J Biol Chem 2001, 276:4527–4530.
Wilson DP, Sutherland C, Walsh MP: Ca2+ activation of smooth muscle contraction. Evidence for the involvement of calmodulin that is bound to the Triton-insoluble fraction even in the absence of Ca2+. J Biol Chem 2002, 277:2186–2192. This paper challenged current dogma concerning the mechanism of activation of smooth muscle contraction by Ca2+ by demonstrating that a specific pool of calmodulin, permanently anchored to the myofilaments, is involved in Ca2+ activation of contraction.
Johnson JD, Snyder C, Walsh MP, Flynn M: Effects of myosin light chain kinase and peptides on Ca2+ exchange with the N- and C-terminal Ca2+ binding sites of calmodulin. J Biol Chem 1996, 271:761–767.
Feng J, Ito M, Ichikawa K, et al.: Inhibitory phosphorylation site for Rho-associated kinase on smooth muscle myosin phosphatase. J Biol Chem 1999, 274:37385–37390.
Nishimura J, Kolber M, van Breemen C: Norepinephrine and GTP-γ-S increase myofilament Ca2+ sensitivity in α-toxin permeabilized arterial smooth muscle. Biochem Biophys Res Commun 1988, 157:677–683.
Gong MC, Iizuka K, Nixon G, et al.: Role of guanine nucleotide-binding proteins — ras-family or trimeric proteins or both — in Ca2+ sensitization of smooth muscle. Proc Natl Acad Sci U S A 1996, 93:1340–1345.
Otto B, Steusloff A, Just I, et al.: Role of Rho proteins in carbachol-induced contractions in intact and permeabilized guinea-pig intestinal smooth muscle. J Physiol 1996, 496:317–329.
Miyazaki K, Yano T, Schmidt DJ, et al.: Rho-dependent agonistinduced spatio-temporal change in myosin phosphorylation in smooth muscle cells. J Biol Chem 2002, 277:725–734. This elegant study utilizing GFP-tagged RhoA to follow translocation in living cells led to the intriguing conclusion that MLCK and RhoA/ROK pathways change myosin phosphorylation differently, both spatially and temporally.
Hirata K, Kikuchi A, Sasaki T, et al.: Involvement of rho p21 in the GTP-enhanced calcium ion sensitivity of smooth muscle contraction. J Biol Chem 1992, 267:8719–8722.
Fujihara H, Walker LA, Gong MC, et al.: Inhibition of RhoA translocation and calcium sensitization by in vivo ADP-ribosylation with the chimeric toxin DC3B. Mol Biol Cell 1997, 8:2437–2447.
Sauzeau V, Le Mellionnec E, Bertoglio J, et al.: Human urotensin II-induced contraction and arterial smooth muscle cell proliferation are mediated by RhoA and Rho-kinase. Circ Res 2001, 88:1102–1104.
Lucius C, Arner A, Steusloff A, et al.: Clostridium difficile toxin B inhibits carbachol-induced force and myosin light chain phosphorylation in guinea-pig smooth muscle: role of Rho proteins. J Physiol 1998, 506:83–93.
Gong MC, Gorenne I, Read P, et al.: Regulation by GDI of RhoA/Rho-kinase-induced Ca2+ sensitization of smooth muscle myosin II. Am J Physiol 2001, 281:C257-C269.
Loirand G, Cario-Toumaniantz C, Chardin P, Pacaud P: The Rho-related protein Rnd1 inhibits Ca2+ sensitization of rat smooth muscle. J Physiol 1999, 516:825–834.
Gong MC, Fujihara H, Walker LA, et al.: Down-regulation of Gprotein-mediated Ca2+ sensitization in smooth muscle. Mol Biol Cell 1997, 8:279–286.
Uehata M, Ishizaki T, Satoh H, et al.: Calcium sensitization of smooth muscle mediated by a Rho-associated protein kinase in hypertension. Nature 1997, 389:990–994.
Nagumo H, Sasaki Y, Ono Y, et al.: Rho kinase inhibitor HA-1077 prevents Rho-mediated myosin phosphatase inhibition in smooth muscle cells. Am J Physiol 2000, 278:C57-C65.
Kureishi Y, Kobayashi S, Amano M, et al.: Rho-associated kinase directly induces smooth muscle contraction through myosin light chain phosphorylation. J Biol Chem 1997, 272:12257–12260.
Shirao S, Kashiwagi S, Sato M, et al.: Sphingosylphosphorylcholine is a novel messenger for Rho-kinase-mediated Ca2+ sensitization in the bovine cerebral artery. Unimportant role for protein kinase C. Circ Res 2002, 91:112–119. The demonstration that sphingosylphosphorylcholine induces Ca2+ sensitization of smooth muscle contraction via activation of ROK suggests a novel signaling function for this sphingolipid.
Araki S, Ito M, Kureishi Y, et al.: Arachidonic acid-induced Ca2+ sensitization of smooth muscle contraction through activation of Rho-kinase. Pflügers Archiv 2001, 441:596–603.
Hartshorne DJ, Ito M, Erdodi F: Myosin light chain phosphatase: subunit composition, interactions and regulation. J Muscle Res Cell Motil 1998, 19:325–341.
Gong MC, Fujihara H, Somlyo AV, Somlyo AP: Translocation of rhoA associated with Ca2+ sensitization of smooth muscle. J Biol Chem 1997, 272:10704–10709.
Taggart MJ, Lee YH, Morgan KG: Cellular redistribution of PKCá, rhoA and ROKá following smooth muscle agonist stimulation. Exp Cell Res 1999, 251:92–101.
Shin H-M, Je H-D, Gallant C, et al.: Differential association and localization of myosin phosphatase subunits during agonist-induced signal transduction in smooth muscle. Circ Res 2002, 90:546–553. This paper provides novel and unexpected insights into the translocation of myosin light chain phosphatase subunits in smooth muscle cells in response to agonist stimulation.
Li L, Eto M, Lee MR, et al.: Possible involvement of the novel CPI-17 protein in protein kinase C signal transduction of rabbit arterial smooth muscle. J Physiol 1998, 508:871–881.
Andrea JE, Walsh MP: Protein kinase C of smooth muscle. Hypertension 1992, 20:585–595.
Eto M, Kitazawa T, Yazawa M, et al.: Histamine-induced vasoconstriction involves phosphorylation of a specific inhibitor protein for myosin phosphatase by protein kinase C á and ä isoforms. J. Biol. Chem 2001, 276:29072–29078.
Koyama M, Ito M, Feng J, et al.: Phosphorylation of CPI-17, an inhibitory phosphoprotein of smooth muscle myosin phosphatase, by Rho-kinase. FEBS Lett 2000, 475:197–200.
Weber LP, Van Lierop JE, Walsh MP: Ca2+-independent phosphorylation of myosin in rat caudal artery and chicken gizzard myofilaments. J Physiol 1999, 516:805–824.
Deng JT, Van Lierop JE, Sutherland C, Walsh MP: Ca2+-independent smooth muscle contraction. A novel function for integrin-linked kinase. J Biol Chem 2001, 276:16365–16373. A Ca2+-independent kinase capable of phosphorylating myosin in the absence of Ca2+ was isolated from smooth muscle and identified as integrin-linked kinase.
Wu C, Dedhar S: Integrin-linked kinase (ILK) and its interactors: a new paradigm for the coupling of extracellular matrix to actin cytoskeleton and signaling complexes. J Cell Biol 2001, 155:505–510.
Murányi A, MacDonald JA, Deng JT, et al.: Phosphorylation of the myosin phosphatase target subunit by integrin-linked kinase. Biochem J 2002, 366:211–216.
Deng JT, Sutherland C, Brautigan DL, et al.: Phosphorylation of the myosin phosphatase inhibitors, CPI-17 and PHI-1, by integrin-linked kinase. Biochem J 2002, 367:517–524.
Shimokawa H, Seto M, Katsumata N, et al.: Rho-kinase-mediated pathway induces enhanced myosin light chain phosphorylations in a swine model of coronary artery spasm. Cardiovacsc Res 1999, 43:1029–1039.
Mukai Y, Shimokawa H, Matoba T, et al.: Involvement of Rhokinase in hypertensive vascular disease: a novel therapeutic target in hypertension. FASEB J 2001, 15:1062–1064. This study provided the first evidence that ROK upregulation plays a key role in the pathogenesis of hypertensive vascular disease.
Masumoto A, Hirookka Y, Shimokawa H, et al.: Possible involvement of Rho-kinase in the pathogenesis of hypertension in humans. Hypertension 2001, 38:1307–1310. This comparative study of the effects of fasudil on groups of hypertensive patients and age-matched normotensive subjects provided the first evidence that ROK may be involved in the etiology of hypertension in humans.
Sato M, Tani E, Fujikawa H, Kaibuchi K: Involvement of Rho-kinase-mediated phosphorylation of myosin light chain in enhancement of cerebral vasospasm. Circ Res 2000, 87:195–200.
Kandabashi T, Shimokawa H, Miyata K, et al.: Inhibition of myosin phosphatase by upregulated Rho-kinase plays a key role for coronary artery spasm in a porcine model with interleukin-1β. Circulation 2000, 101:1319–1323.
Shibata R, Kai H, Seki Y, et al.: Role of Rho-associated kinase in neointima formation after vascular injury. Circulation 2001, 103:284–289.
Chitaley K, Wingard CJ, Webb RC, et al.: Antagonism of Rhokinase stimulates rat penile erection via a nitric oxide-independent pathway. Nature Med 2001, 7:119–122.
Amano M, Fukata Y, Kaibuchi K: Regulation and functions of Rho-associated kinase. Exp Cell Res 2000, 261:44–51.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Swärd, K., Mita, M., Wilson, D.P. et al. The role of RhoA and Rho-associated kinase in vascular smooth muscle contraction. Current Science Inc 5, 66–72 (2003). https://doi.org/10.1007/s11906-003-0013-1
Issue Date:
DOI: https://doi.org/10.1007/s11906-003-0013-1