Abstract
Purpose of Review
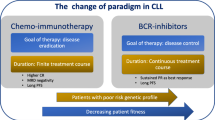
The treatment landscape of chronic lymphocytic leukemia has been rapidly evolving over the past few years. The prior standard of care, chemoimmunotherapy, is being replaced by targeted agents, and the utility of chemotherapy has come under question. In this review, we examine recent data comparing chemoimmunotherapy to targeted agents, how these data impact clinical management, and whether there are potential future roles for cytotoxic chemotherapy.
Recent Findings
Clinical trials have shown improved clinical outcomes with targeted agents compared to traditional chemoimmunotherapy. Based on these data, the current treatment paradigm primarily favors targeted agents over chemoimmunotherapy, with a few exceptions. However, targeted agents have notable limitations, and thus, there may be a future role of cytotoxic chemotherapy when administered in combination with targeted agents.
Summary
Although targeted agents have nearly replaced chemoimmunotherapy in the treatment of chronic lymphocytic leukemia, novel combinations utilizing chemotherapy are being developed that may lead to better outcomes.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69(1):7–34. https://doi.org/10.3322/caac.21551.
Institute NC. Surveillance Epidemiology and End Results Fact Sheets. In: Cancer: Chronic Lymphocytic Leukemia. seer.cancer.gov/statfacts/html/clyl.html. Accessed October 2nd 2019.
Chemotherapeutic options in chronic lymphocytic leukemia: a meta-analysis of the randomized trials. CLL Trialists' Collaborative Group. J Natl Cancer Inst. 1999;91(10):861–8. https://doi.org/10.1093/jnci/91.10.861.
Effects of chlorambucil and therapeutic decision in initial forms of chronic lymphocytic leukemia (stage A): results of a randomized clinical trial on 612 patients. The French Cooperative Group on Chronic Lymphocytic Leukemia. Blood. 1990;75(7):1414–21.
Hallek M, Cheson BD, Catovsky D, Caligaris-Cappio F, Dighiero G, Dohner H, et al. iwCLL guidelines for diagnosis, indications for treatment, response assessment, and supportive management of CLL. Blood. 2018;131(25):2745–60. https://doi.org/10.1182/blood-2017-09-806398.
Rai KR. Clinical staging of chronic lymphocytic leukemia. Blood. 1975;46(2):219–34.
Binet JL. A new prognostic classification of chronic lymphocytic leukemia derived from a multivariate survival analysis. Cancer. 1981;48(1):198–206.
Montserrat E, Sanchez-Bisono J, Vinolas N, Rozman C. Lymphocyte doubling time in chronic lymphocytic leukaemia: analysis of its prognostic significance. Br J Haematol. 1986;62(3):567–75. https://doi.org/10.1111/j.1365-2141.1986.tb02969.x.
Dohner H, Stilgenbauer S, Benner A, Leupolt E, Krober A, Bullinger L, et al. Genomic aberrations and survival in chronic lymphocytic leukemia. N Engl J Med. 2000;343(26):1910–6. https://doi.org/10.1056/nejm200012283432602.
Damle RN. Ig V gene mutation status and CD38 expression as novel prognostic indicators in chronic lymphocytic leukemia. Blood. 1999;94(6):1840–7.
Hamblin TJ. Unmutated Ig V(H) genes are associated with a more aggressive form of chronic lymphocytic leukemia. Blood. 1999;94(6):1848–54.
Kröber A. V(H) mutation status, CD38 expression level, genomic aberrations, and survival in chronic lymphocytic leukemia. Blood. 2002;100(4):1410–6.
Crespo M, Bosch F, Villamor N, Bellosillo B, Colomer D, Rozman M, et al. ZAP-70 expression as a surrogate for immunoglobulin-variable-region mutations in chronic lymphocytic leukemia. N Engl J Med. 2003;348(18):1764–75. https://doi.org/10.1056/NEJMoa023143.
Rassenti LZ, Jain S, Keating MJ, Wierda WG, Grever MR, Byrd JC, et al. Relative value of ZAP-70, CD38, and immunoglobulin mutation status in predicting aggressive disease in chronic lymphocytic leukemia. Blood. 2008;112(5):1923–30. https://doi.org/10.1182/blood-2007-05-092882.
The International CLL-IPI working group. An international prognostic index for patients with chronic lymphocytic leukaemia (CLL-IPI): a meta-analysis of individual patient data. Lancet Oncol. 2016;17(6):779–90.
Pflug N. Development of a comprehensive prognostic index for patients with chronic lymphocytic leukemia. Blood. 2014;124(1):49–62.
Hallek M, Shanafelt TD, Eichhorst B. Chronic lymphocytic leukaemia. Lancet. 2018;391(10129):1524–37. https://doi.org/10.1016/s0140-6736(18)30422-7.
Strati P, Keating MJ, O'Brien SM, Ferrajoli A, Burger J, Faderl S, et al. Outcomes of first-line treatment for chronic lymphocytic leukemia with 17p deletion. Haematologica. 2014;99(8):1350–5. https://doi.org/10.3324/haematol.2014.104661.
Fabbri G, Dalla-Favera R. The molecular pathogenesis of chronic lymphocytic leukaemia. Nat Rev Cancer. 2016;16(3):145–62. https://doi.org/10.1038/nrc.2016.8.
Keating MJ, O'Brien S, Albitar M, Lerner S, Plunkett W, Giles F, et al. Early results of a chemoimmunotherapy regimen of fludarabine, cyclophosphamide, and rituximab as initial therapy for chronic lymphocytic leukemia. J Clin Oncol. 2005;23(18):4079–88. https://doi.org/10.1200/jco.2005.12.051.
Tam CS, O'Brien S, Wierda W, Kantarjian H, Wen S, Do KA, et al. Long-term results of the fludarabine, cyclophosphamide, and rituximab regimen as initial therapy of chronic lymphocytic leukemia. Blood. 2008;112(4):975–80. https://doi.org/10.1182/blood-2008-02-140582.
Thompson PA, Tam CS, O'Brien SM, Wierda WG, Stingo F, Plunkett W, et al. Fludarabine, cyclophosphamide, and rituximab treatment achieves long-term disease-free survival in IGHV-mutated chronic lymphocytic leukemia. Blood. 2016;127(3):303–9. https://doi.org/10.1182/blood-2015-09-667675An important reference showing frontline FCR's ability to possibly induce cures in a subset of IgHV-mutated CLL patients.
Hallek M, Fischer K, Fingerle-Rowson G, Fink AM, Busch R, Mayer J, et al. Addition of rituximab to fludarabine and cyclophosphamide in patients with chronic lymphocytic leukaemia: a randomised, open-label, phase 3 trial. Lancet. 2010;376(9747):1164–74. https://doi.org/10.1016/s0140-6736(10)61381-5.
Eichhorst B, Fink AM, Bahlo J, Busch R, Kovacs G, Maurer C, et al. First-line chemoimmunotherapy with bendamustine and rituximab versus fludarabine, cyclophosphamide, and rituximab in patients with advanced chronic lymphocytic leukaemia (CLL10): an international, open-label, randomised, phase 3, non-inferiority trial. Lancet Oncol. 2016;17(7):928–42. https://doi.org/10.1016/s1470-2045(16)30051-1.
Fischer K, Bahlo J, Fink AM, Goede V, Herling CD, Cramer P, et al. Long-term remissions after FCR chemoimmunotherapy in previously untreated patients with CLL: updated results of the CLL8 trial. Blood. 2016;127(2):208–15. https://doi.org/10.1182/blood-2015-06-651125.
Rossi D, Terzi-di-Bergamo L, De Paoli L, Cerri M, Ghilardi G, Chiarenza A, et al. Molecular prediction of durable remission after first-line fludarabine-cyclophosphamide-rituximab in chronic lymphocytic leukemia. Blood. 2015;126(16):1921–4. https://doi.org/10.1182/blood-2015-05-647925.
Kovacs G, Robrecht S, Fink AM, Bahlo J, Cramer P, von Tresckow J, et al. Minimal residual disease assessment improves prediction of outcome in patients with chronic lymphocytic leukemia (CLL) who achieve partial response: comprehensive analysis of two Phase III studies of the German CLL Study Group. J Clin Oncol. 2016;34(31):3758–65. https://doi.org/10.1200/jco.2016.67.1305This study analyzed two large Phase III trials (CLL8 and CLL10) to show the strong independent correlation of attaining MRD negativity with better long-term clinical outcomes.
Bottcher S, Ritgen M, Fischer K, Stilgenbauer S, Busch RM, Fingerle-Rowson G, et al. Minimal residual disease quantification is an independent predictor of progression-free and overall survival in chronic lymphocytic leukemia: a multivariate analysis from the randomized GCLLSG CLL8 trial. J Clin Oncol. 2012;30(9):980–8. https://doi.org/10.1200/jco.2011.36.9348.
Strati P, Keating MJ, O'Brien SM, Burger J, Ferrajoli A, Jain N, et al. Eradication of bone marrow minimal residual disease may prompt early treatment discontinuation in CLL. Blood. 2014;123(24):3727–32. https://doi.org/10.1182/blood-2013-11-538116.
Lamanna N, Kalaycio M, Maslak P, Jurcic JG, Heaney M, Brentjens R, et al. Pentostatin, cyclophosphamide, and rituximab is an active, well-tolerated regimen for patients with previously treated chronic lymphocytic leukemia. J Clin Oncol. 2006;24(10):1575–81. https://doi.org/10.1200/jco.2005.04.3836.
Lamanna N, Jurcic JG, Noy A, Maslak P, Gencarelli AN, Panageas KS, et al. Sequential therapy with fludarabine, high-dose cyclophosphamide, and rituximab in previously untreated patients with chronic lymphocytic leukemia produces high-quality responses: molecular remissions predict for durable complete responses. J Clin Oncol. 2009;27(4):491–7. https://doi.org/10.1200/jco.2008.16.4459.
Fischer K, Cramer P, Busch R, Bottcher S, Bahlo J, Schubert J, et al. Bendamustine in combination with rituximab for previously untreated patients with chronic lymphocytic leukemia: a multicenter phase II trial of the German chronic lymphocytic leukemia study group. J Clin Oncol. 2012;30(26):3209–16. https://doi.org/10.1200/jco.2011.39.2688.
Fischer K, Cramer P, Busch R, Stilgenbauer S, Bahlo J, Schweighofer CD, et al. Bendamustine combined with rituximab in patients with relapsed and/or refractory chronic lymphocytic leukemia: a multicenter phase II trial of the German chronic lymphocytic leukemia study group. J Clin Oncol. 2011;29(26):3559–66. https://doi.org/10.1200/jco.2010.33.8061.
Goede V, Fischer K, Busch R, Engelke A, Eichhorst B, Wendtner CM, et al. Obinutuzumab plus chlorambucil in patients with CLL and coexisting conditions. N Engl J Med. 2014;370(12):1101–10. https://doi.org/10.1056/NEJMoa1313984.
Goede V, Fischer K, Engelke A, Schlag R, Lepretre S, Montero LF, et al. Obinutuzumab as frontline treatment of chronic lymphocytic leukemia: updated results of the CLL11 study. Leukemia. 2015;29(7):1602–4.
Hillmen P, Robak T, Janssens A, Babu KG, Kloczko J, Grosicki S, et al. Chlorambucil plus ofatumumab versus chlorambucil alone in previously untreated patients with chronic lymphocytic leukaemia (COMPLEMENT 1): a randomised, multicentre, open-label phase 3 trial. Lancet. 2015;385(9980):1873–83. https://doi.org/10.1016/s0140-6736(15)60027-7.
Burger JA, Tedeschi A, Barr PM, Robak T, Owen C, Ghia P, et al. Ibrutinib as initial therapy for patients with chronic lymphocytic leukemia. N Engl J Med. 2015;373(25):2425–37. https://doi.org/10.1056/NEJMoa1509388.
Barr PM, Robak T, Owen C, Tedeschi A, Bairey O, Bartlett NL, et al. Sustained efficacy and detailed clinical follow-up of first-line ibrutinib treatment in older patients with chronic lymphocytic leukemia: extended phase 3 results from RESONATE-2. Haematologica. 2018;103(9):1502–10. https://doi.org/10.3324/haematol.2018.192328.
Woyach JA, Ruppert AS, Heerema NA, Zhao W, Booth AM, Ding W, et al. Ibrutinib regimens versus chemoimmunotherapy in older patients with untreated CLL. N Engl J Med. 2018;379(26):2517–28. https://doi.org/10.1056/NEJMoa1812836This reference shows the results of the Alliance A041202 Phase III trial, which displayed the superiority of ibrutinib containing regimens over BR chemoimmunotherapy for the frontline treatment of older and/or frail patients with CLL.
Moreno C, Greil R, Demirkan F, Tedeschi A, Anz B, Larratt L, et al. Ibrutinib plus obinutuzumab versus chlorambucil plus obinutuzumab in first-line treatment of chronic lymphocytic leukaemia (iLLUMINATE): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2019;20(1):43–56. https://doi.org/10.1016/s1470-2045(18)30788-5.
Shanafelt TD, Wang XV, Kay NE, Hanson CA, O'Brien S, Barrientos J, et al. Ibrutinib-rituximab or chemoimmunotherapy for chronic lymphocytic leukemia. N Engl J Med. 2019;381(5):432–43. https://doi.org/10.1056/NEJMoa1817073This reference shows analysis from the ECOG-ACRIN E1912 Phase III trial, which compared ibrutinib + rituximab to FCR in the frontline setting for young fit CLL patients.
Fischer K, Al-Sawaf O, Bahlo J, Fink AM, Tandon M, Dixon M, et al. Venetoclax and obinutuzumab in Patients with CLL and coexisting conditions. N Engl J Med. 2019;380(23):2225–36. https://doi.org/10.1056/NEJMoa1815281This reference shows data from the Phase III CLL14 trial, which led to the first FDA approval of a venetoclax-containing, fixed-duration regimen (venetoclax + obinutuzumab) in the frontline treatment of CLL.
Byrd JC, Brown JR, O'Brien S, Barrientos JC, Kay NE, Reddy NM, et al. Ibrutinib versus ofatumumab in previously treated chronic lymphoid leukemia. N Engl J Med. 2014;371(3):213–23. https://doi.org/10.1056/NEJMoa1400376.
Byrd JC, Hillmen P, O'Brien S, Barrientos JC, Reddy NM, Coutre S, et al. Long-term follow-up of the RESONATE phase 3 trial of ibrutinib vs ofatumumab. Blood. 2019;133(19):2031–42. https://doi.org/10.1182/blood-2018-08-870238.
Munir T, Brown JR, O'Brien S, Barrientos JC, Barr PM, Reddy NM, et al. Final analysis from RESONATE: up to 6 years of follow-up on ibrutinib in patients with previously treated chronic lymphocytic leukemia or small lymphocytic lymphoma. Am J Hematol. 2019;94(12):1353–63. https://doi.org/10.1002/ajh.25638This reference shows the long-term efficacy of ibrutinib in the treatment of relapsed/refractory CLL.
Chanan-Khan A, Cramer P, Demirkan F, Fraser G, Silva RS, Grosicki S, et al. Ibrutinib combined with bendamustine and rituximab compared with placebo, bendamustine, and rituximab for previously treated chronic lymphocytic leukaemia or small lymphocytic lymphoma (HELIOS): a randomised, double-blind, phase 3 study. Lancet Oncol. 2016;17(2):200–11. https://doi.org/10.1016/s1470-2045(15)00465-9.
Fraser G, Cramer P, Demirkan F, Silva RS, Grosicki S, Pristupa A, et al. Updated results from the phase 3 HELIOS study of ibrutinib, bendamustine, and rituximab in relapsed chronic lymphocytic leukemia/small lymphocytic lymphoma. Leukemia. 2019;33(4):969–80. https://doi.org/10.1038/s41375-018-0276-9.
Furman RR, Sharman JP, Coutre SE, Cheson BD, Pagel JM, Hillmen P, et al. Idelalisib and rituximab in relapsed chronic lymphocytic leukemia. N Engl J Med. 2014;370(11):997–1007. https://doi.org/10.1056/NEJMoa1315226.
Zelenetz AD, Barrientos JC, Brown JR, Coiffier B, Delgado J, Egyed M, et al. Idelalisib or placebo in combination with bendamustine and rituximab in patients with relapsed or refractory chronic lymphocytic leukaemia: interim results from a phase 3, randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2017;18(3):297–311. https://doi.org/10.1016/s1470-2045(16)30671-4.
Jones JA, Robak T, Brown JR, Awan FT, Badoux X, Coutre S, et al. Efficacy and safety of idelalisib in combination with ofatumumab for previously treated chronic lymphocytic leukaemia: an open-label, randomised phase 3 trial. Lancet Haematol. 2017;4(3):e114–e26. https://doi.org/10.1016/s2352-3026(17)30019-4.
Flinn IW, Cherry MA, Maris MB, Matous JV, Berdeja JG, Patel M. Combination trial of Duvelisib (IPI-145) with rituximab or Bendamustine/rituximab in patients with non-Hodgkin lymphoma or chronic lymphocytic leukemia. Am J Hematol. 2019;94(12):1325–34. https://doi.org/10.1002/ajh.25634.
Seymour JF, Kipps TJ, Eichhorst B, Hillmen P, D'Rozario J, Assouline S, et al. Venetoclax-rituximab in relapsed or refractory chronic lymphocytic leukemia. N Engl J Med. 2018;378(12):1107–20. https://doi.org/10.1056/NEJMoa1713976.
Kater AP, Seymour JF, Hillmen P, Eichhorst B, Langerak AW, Owen C, et al. Fixed duration of venetoclax-rituximab in relapsed/refractory chronic lymphocytic leukemia eradicates minimal residual disease and prolongs survival: post-treatment follow-up of the MURANO Phase III Study. J Clin Oncol. 2019;37(4):269–77. https://doi.org/10.1200/jco.18.01580This reference shows updated follow-up of the Phase III MURANO trial, displaying the ability of venetoclax + rituximab to induce durable MRD-negative remissions after a 2-year fixed duration of treatment in relapsed/refractory CLL.
Burger JA, Barr PM, Robak T, Owen C, Ghia P, Tedeschi A, et al. Long-term efficacy and safety of first-line ibrutinib treatment for patients with CLL/SLL: 5 years of follow-up from the phase 3 RESONATE-2 study. Leukemia. 2019. https://doi.org/10.1038/s41375-019-0602-xThis reference shows the long-term efficacy of ibrutinib in the frontline treatment of CLL.
Buccheri V, Barreto WG, Fogliatto LM, Capra M, Marchiani M, Rocha V. Prognostic and therapeutic stratification in CLL: focus on 17p deletion and p53 mutation. Ann Hematol. 2018;97(12):2269–78. https://doi.org/10.1007/s00277-018-3503-6.
Zenz T, Krober A, Scherer K, Habe S, Buhler A, Benner A, et al. Monoallelic TP53 inactivation is associated with poor prognosis in chronic lymphocytic leukemia: results from a detailed genetic characterization with long-term follow-up. Blood. 2008;112(8):3322–9. https://doi.org/10.1182/blood-2008-04-154070.
Landau DA, Tausch E, Taylor-Weiner AN, Stewart C, Reiter JG, Bahlo J, et al. Mutations driving CLL and their evolution in progression and relapse. Nature. 2015;526(7574):525–30. https://doi.org/10.1038/nature15395.
Stilgenbauer S, Schnaiter A, Paschka P, Zenz T, Rossi M, Dohner K, et al. Gene mutations and treatment outcome in chronic lymphocytic leukemia: results from the CLL8 trial. Blood. 2014;123(21):3247–54. https://doi.org/10.1182/blood-2014-01-546150.
Zenz T, Eichhorst B, Busch R, Denzel T, Habe S, Winkler D, et al. TP53 mutation and survival in chronic lymphocytic leukemia. J Clin Oncol. 2010;28(29):4473–9. https://doi.org/10.1200/jco.2009.27.8762.
Woyach JA, Ruppert AS, Guinn D, Lehman A, Blachly JS, Lozanski A, et al. BTK(C481S)-mediated resistance to Ibrutinib in chronic lymphocytic leukemia. J Clin Oncol. 2017;35(13):1437–43. https://doi.org/10.1200/jco.2016.70.2282.
O'Brien S, Jones JA, Coutre SE, Mato AR, Hillmen P, Tam C, et al. Ibrutinib for patients with relapsed or refractory chronic lymphocytic leukaemia with 17p deletion (RESONATE-17): a phase 2, open-label, multicentre study. Lancet Oncol. 2016;17(10):1409–18. https://doi.org/10.1016/s1470-2045(16)30212-1.
Seymour JF, Ma S, Brander DM, Choi MY, Barrientos J, Davids MS, et al. Venetoclax plus rituximab in relapsed or refractory chronic lymphocytic leukaemia: a phase 1b study. Lancet Oncol. 2017;18(2):230–40. https://doi.org/10.1016/s1470-2045(17)30012-8.
O'Brien S, Furman RR, Coutre S, Flinn IW, Burger JA, Blum K, et al. Single-agent ibrutinib in treatment-naive and relapsed/refractory chronic lymphocytic leukemia: a 5-year experience. Blood. 2018;131(17):1910–9. https://doi.org/10.1182/blood-2017-10-810044.
Sharman JP, Coutre SE, Furman RR, Cheson BD, Pagel JM, Hillmen P, et al. Final results of a randomized, phase III study of rituximab with or without Idelalisib followed by open-label Idelalisib in patients with relapsed chronic lymphocytic leukemia. J Clin Oncol. 2019;37(16):1391–402. https://doi.org/10.1200/jco.18.01460.
Gill S, Carney D, Ritchie D, Wolf M, Westerman D, Prince HM, et al. The frequency, manifestations, and duration of prolonged cytopenias after first-line fludarabine combination chemotherapy. Ann Oncol. 2010;21(2):331–4. https://doi.org/10.1093/annonc/mdp297.
Benjamini O, Jain P, Trinh L, Qiao W, Strom SS, Lerner S, et al. Second cancers in patients with chronic lymphocytic leukemia who received frontline fludarabine, cyclophosphamide and rituximab therapy: distribution and clinical outcomes. Leuk Lymphoma. 2015;56(6):1643–50. https://doi.org/10.3109/10428194.2014.957203.
Cooper JP, Khajaviyan S, Smith SD, Maloney DG, Shustov AR, Warren EH, et al. Outcomes of patients with therapy-related MDS after chemoimmunotherapy for chronic lymphocytic leukemia compared with patients with de novo MDS: a single-institution experience. Clin Lymphoma Myeloma Leuk. 2019;19(6):390–5. https://doi.org/10.1016/j.clml.2019.03.003.
Flinn IW, Hillmen P, Montillo M, Nagy Z, Illes A, Etienne G, et al. The phase 3 DUO trial: duvelisib vs ofatumumab in relapsed and refractory CLL/SLL. Blood. 2018;132(23):2446–55. https://doi.org/10.1182/blood-2018-05-850461.
Mato AR, Nabhan C, Thompson MC, Lamanna N, Brander DM, Hill B, et al. Toxicities and outcomes of 616 ibrutinib-treated patients in the United States: a real-world analysis. Haematologica. 2018;103(5):874–9. https://doi.org/10.3324/haematol.2017.182907.
Jain P, Thompson PA, Keating M, Estrov Z, Ferrajoli A, Jain N, et al. Long-term outcomes for patients with chronic lymphocytic leukemia who discontinue ibrutinib. Cancer. 2017;123(12):2268–73. https://doi.org/10.1002/cncr.30596.
Paydas S. Management of adverse effects/toxicity of ibrutinib. Crit Rev Oncol Hematol. 2019;136:56–63. https://doi.org/10.1016/j.critrevonc.2019.02.001.
O'Brien SM, Lamanna N, Kipps TJ, Flinn I, Zelenetz AD, Burger JA, et al. A phase 2 study of idelalisib plus rituximab in treatment-naive older patients with chronic lymphocytic leukemia. Blood. 2015;126(25):2686–94. https://doi.org/10.1182/blood-2015-03-630947.
Cuneo A, Barosi G, Danesi R, Fagiuoli S, Ghia P, Marzano A, et al. Management of adverse events associated with idelalisib treatment in chronic lymphocytic leukemia and follicular lymphoma: a multidisciplinary position paper. Hematol Oncol. 2019;37(1):3–14. https://doi.org/10.1002/hon.2540.
Woyach JA, Furman RR, Liu TM, Ozer HG, Zapatka M, Ruppert AS, et al. Resistance mechanisms for the Bruton’s tyrosine kinase inhibitor ibrutinib. N Engl J Med. 2014;370(24):2286–94. https://doi.org/10.1056/NEJMoa1400029.
Liu TM, Woyach JA, Zhong Y, Lozanski A, Lozanski G, Dong S, et al. Hypermorphic mutation of phospholipase C, gamma2 acquired in ibrutinib-resistant CLL confers BTK independency upon B-cell receptor activation. Blood. 2015;126(1):61–8. https://doi.org/10.1182/blood-2015-02-626846.
Blombery P, Anderson MA, Gong JN, Thijssen R, Birkinshaw RW, Thompson ER, et al. Acquisition of the recurrent Gly101Val mutation in BCL2 confers resistance to venetoclax in patients with progressive chronic lymphocytic leukemia. Cancer Discov. 2019;9(3):342–53. https://doi.org/10.1158/2159-8290.cd-18-1119.
Barnes JI, Divi V, Begaye A, Wong R, Coutre S, Owens DK, et al. Cost-effectiveness of ibrutinib as first-line therapy for chronic lymphocytic leukemia in older adults without deletion 17p. Blood Adv. 2018;2(15):1946–56. https://doi.org/10.1182/bloodadvances.2017015461.
Hilal T, Betcher JA, Leis JF. Economic impact of oral therapies for chronic lymphocytic leukemia-the burden of novelty. Curr Hematol Malig Rep. 2018;13(4):237–43. https://doi.org/10.1007/s11899-018-0461-y.
Shanafelt TD, Borah BJ, Finnes HD, Chaffee KG, Ding W, Leis JF, et al. Impact of ibrutinib and idelalisib on the pharmaceutical cost of treating chronic lymphocytic leukemia at the individual and societal levels. J Oncol Pract. 2015;11(3):252–8. https://doi.org/10.1200/jop.2014.002469.
Jain N, Thompson PA, Burgery JA, Ferrajoli A, Takahashi K, Estrov ZE et al. Ibrutinib, Fludarabine, cyclophosphamide, and obinutuzumab (iFCG) for first-line treatment of IGHV-mutated CLL and without Del (17p)/mutated TP53. American Society of Hematology annual meeting; Dec 8 2019; Orlando, Florida: American Society of Hematology.
Davids MS, Brander DM, Kim HT, Tyekucheva S, Bsat J, Savell A, et al. Ibrutinib plus fludarabine, cyclophosphamide, and rituximab as initial treatment for younger patients with chronic lymphocytic leukaemia: a single-arm, multicentre, phase 2 trial. Lancet Haematol. 2019;6(8):e419–e28. https://doi.org/10.1016/s2352-3026(19)30104-8This Phase II study shows the ability of a combination of FCR + ibrutinib to induce high response rates and MRD negativity rates in a relatively unselected, young, treatment-naïve CLL population.
Cramer P, von Tresckow J, Bahlo J, Robrecht S, Langerbeins P, Al-Sawaf O, et al. Bendamustine followed by obinutuzumab and venetoclax in chronic lymphocytic leukaemia (CLL2-BAG): primary endpoint analysis of a multicentre, open-label, phase 2 trial. Lancet Oncol. 2018;19(9):1215–28. https://doi.org/10.1016/s1470-2045(18)30414-5This Phase II study shows the possible ability of chemotherapy (e.g. bendamustine) to be utilized as an effective and rapid debulking agent prior to initiation of venetoclax-containing regimens in the frontline treatment of CLL.
von Tresckow J, Cramer P, Bahlo J, Robrecht S, Langerbeins P, Fink AM, et al. CLL2-BIG: sequential treatment with bendamustine, ibrutinib and obinutuzumab (GA101) in chronic lymphocytic leukemia. Leukemia. 2019;33(5):1161–72. https://doi.org/10.1038/s41375-018-0313-8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Support to Institution for Research: Abbvie, AstraZeneca, Bei-Gene, Genentech, Juno, Oncternal, Ming, TG Therapeutics
Scientific advisory boards/consulting: Abbvie, AstraZeneca, Bei-Gene, Celgene, Genentech, Gilead, Janssen, Pharmacyclics.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Chronic Lymphocytic Leukemias
Rights and permissions
About this article
Cite this article
Lee, J.C., Lamanna, N. Is There a Role for Chemotherapy in the Era of Targeted Therapies?. Curr Hematol Malig Rep 15, 72–82 (2020). https://doi.org/10.1007/s11899-020-00563-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11899-020-00563-8