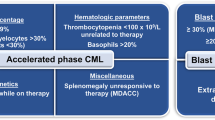
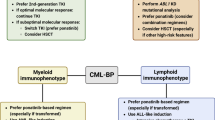
Abstract
The introduction of tyrosine kinase inhibitors (TKIs) has fundamentally changed the management of chronic myeloid leukemia (CML). Disease progression to advanced phase (accelerated or blast phase) has been reduced to 1 to 1.5 % per year from more than 20 % per year in the pre-TKI era. However, once the disease has progressed to accelerated or blast phase, there is no consensus regarding optimal therapy. The prognosis of these patients is dismal with median survival ranging from 7 to 11 months. TKIs along with allogeneic hematopoietic cell transplantation are conventional strategies in managing these patients but there are very few long-term survivors. Advanced phase CML represents the new frontier for CML treatment where research is critically needed to improve patient outcomes.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Druker BJ et al. Activity of a specific inhibitor of the BCR-ABL tyrosine kinase in the blast crisis of chronic myeloid leukemia and acute lymphoblastic leukemia with the Philadelphia chromosome. N Engl J Med. 2001;344:1038–42.
Cortes J. Natural history and staging of chronic myelogenous leukemia. Hematol Oncol Clin North Am. 2004;18:569–84.
Kantarjian H et al. Characteristics of accelerated disease in chronic myelogenous leukemia. Cancer. 1988;61:1441–46.
Cortes J, Kantarjian H. Advanced-phase chronic myeloid leukemia. Semin Hematol. 2003;40:79–86.
Howlader N et al. (eds). SEER Cancer Statistics Review, 1975–2012, National Cancer Institute. Bethesda, MD.
Kantarjian H et al. Improved survival in chronic myeloid leukemia since the introduction of imatinib therapy: a single-institution historical experience. Blood. 2012;119(9):1981–87.
Druker BJ et al. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. N Engl J Med. 2006;355(23):2408–17.
Saglio G et al. Nilotinib versus imatinib for newly diagnosed chronic myeloid leukemia. N Engl J Med. 2010;362(24):2251–59.
Kantarjian H et al. Dasatinib versus imatinib in newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med. 2010;362(24):2260–70.
Radich JP, The biology of CML blast crisis. Hematol Am Soc Hematol Educ Program. 2007:384–91.
Calabretta B, Perrotti D. The biology of CML blast crisis. Blood. 2004;103(11):4010–22.
Lugo TG et al. Tyrosine kinase activity and transformation potency of the BCR-ABL oncogene products. Science. 1990;247:1079–82.
Cortes JE et al. Staging of chronic myeloid leukemia in the imatinib era: an evaluation of the World Health Organization proposal. Cancer. 2006;106(6):1306–15.
Vardiman JWMJ, Baccarani M, Thiele J. Chronic myelogenous leukemia BCR-ABL1 positive. In: Swerdlow SHCE, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J, Vardiman JW, editors. WHO classification of tumours of haematopotietic and lymphoid tissues. 4th ed. Lyon: International Agency for Research on Cancer; 2008.
Speck B et al. Allogeneic bone-marrow transplantation for chronic myelogenous leukaemia. Lancet. 1984;8378(1):665–68.
Baccarani M et al. European LeukemiaNet recommendations for the management of chronic myeloid leukemia: 2013. Blood. 2013;122(6):872–84. Updated recommendations of the European LeukemiaNet regarding the management of chronic myeloid leukemia, including accelerated and blast phases.
Kantarjian H et al. Survival benefit with imatinib mesylate therapy inpatients with accelerated-phase chronic myelogenous leukemia – comparison with historic experience. Cancer. 2005;103(10):2099–108.
Jiang Q et al. Imatinib mesylate versus allogeneic hematopoietic stem cell transplantation for patients with chronic myelogenous leukemia in the accelerated phase. Blood. 2011;117(11):3032–40.
Hoffmann VS et al. The EUTOS prognostic score: review and validation in 1288 patients with CML treated frontline with imatinib. Leukemia. 2013;27(10):2016–22.
Furtado VF et al. Accelerated phase chronic myeloid leukemia: evaluation of clinical criteria as predictors of survival, major cytogenetic response and progression to blast phase. Rev Bras Hematol Hemoter. 2015;37(5):341-7.
Kantarjian HM et al. Treatment of Philadelphia chromosome-positive, accelerated-phase chronic myelogenous leukemia with imatinib mesylate. Clin Cancer Res. 2002;8(7):2167–76.
Talpaz M et al. Imatinib induces durable hematologic and cytogenetic responses in patients with accelerated phase chronic myeloid leukemia: results of a phase 2 study. Blood. 2002;99(6):1928–37.
Guilhot F et al. Dasatinib induces significant hematologic and cytogenetic responses in patients with imatinib-resistant or -intolerant chronic myeloid leukemia in accelerated phase. Blood. 2007;109(10):4143–50.
Kantarjian H et al. Phase 3 study of dasatinib 140 mg once daily versus 70 mg twice daily in patients with chronic myeloid leukemia in accelerated phase resistant or intolerant to imatinib: 15-month median follow-up. Blood. 2009;113(25):6322–9. 405.
Apperley JF et al. Dasatinib in the treatment of chronic myeloid leukemia in accelerated phase after imatinib failure: the START a trial. J Clin Oncol. 2009;27(21):3472–9.
le Coutre PD et al. Nilotinib in patients with Ph + chronic myeloid leukemia in accelerated phase following imatinib resistance or in- tolerance: 24-month follow-up results. Leukemia. 2012;26(6):1189–94.
Kantarjian H et al. Nilotinib in imatinib-resistant CML and Philadelphia chromosome-positive ALL. N Engl J Med. 2006;354(24):2542–51.
Nicolini FE et al. Expanding Nilotinib Access in Clinical Trials (ENACT), an open-label multicenter study of oral nilotinib in adult patients with imatinib-resistant or -intolerant chronic myeloid leukemia in accelerated phase or blast crisis. Leuk Lymphoma. 2012;53(5):907–14.
Gambacorti-Passerini C et al. Long-term efficacy and safety of bosutinib in patients with advanced leukemia following resistance/intolerance to imatinib and other tyrosine kinase inhibitors. Am J Hematol. 2015;90(9):755–68. This study established the long-term (>4 years of follow-up) efficacy and safety of bosutinib in accelerated phase CML patients refractory or resistant to prior treatment with imatinib, nilotinib, and dasatinib.
Cortes JE et al. A phase 2 trial of ponatinib in Philadelphia chromosome-positive leukemias. N Engl J Med. 2013;369(19):1783–96. This is a landmark study that demonstrated the efficacy of ponatinib as a salvage strategy in CML patients across all disease phases including accelerated phase CML with unmutated or mutated BCR-ABL who were refractory to nilotinib and dasatinib. Additionally, this study established ponatinib as the only TKI with activity in CML patients harboring T315I mutation.
Gratwohl A, Hermans J, Niederwieser D, Frassoni F, Arcese W, Gahrton G, et al. Bone marrow transplantation for chronic myeloid leukemia: long-term results. Bone Marrow Transplant. 1993;12:509–16.
McGlave P. Therapy of chronic myelogenous leukemia with related or unrelated donor bone marrow transplantation. Leukemia (Baltimore). 1992;6(Suppl):115–17.
Clift RA et al. Marrow transplantation for patients in accelerated phase of chronic myeloid leukemia. Blood. 1994;84:4368–73.
Sacchi S et al. Chronic myelogenous leukemia in nonlymphoid blastic phase. Analysis of the results of first salvage therapy with three different treatment approaches for 162 patients. Cancer. 1999;86:2632–41.
Kantarjian H et al. Treatment of chronic myelogenous leukemia in accelerated and blastic phase with daunorubicin, high-dose cytarabine and granulocyte macrophage colony stimulating factor. J Clin Oncol. 1992;10:398–405.
Kantarjian H et al. Treatment of advanced stages of Philadelphia chromosome-positive chronic myelogenous leukemia with interferon-α and low-dose cytarabine. J Clin Oncol. 1992;10:772–78.
Kantarjian H et al. Homoharringtonine and low-dose cytarabine in the management of late chronic-phase chronic myelogenous leukemia. J Clin Oncol. 2000;18:3513–21.
Giles FJ et al. Troxacitabine, a novel dioxolane nucleoside analog, has activity in patients with advanced leukemia. J Clin Oncol. 2001;19:762–71.
Silver RT et al. Sustained durability of responses and improved progression-free and overall survival with imatinib treatment for accelerated phase and blast crisis chronic myeloid leukaemia: long-term follow-up of the STI571 0102 and 0109 trials. Haematologica. 2009;94:743–44.
Kantarjian HM et al. Treatment of Philadelphia chromosome-positive, accelerated-phase chronic myelogenous leukemia with imatinib mesylate. Clin Cancer Res. 2002;8(7):2167–76.
Rea D et al. First-line imatinib mesylate in patients with newly diagnosed accelerated phase-chronic myeloid leukemia. Leukemia. 2012;26(10):2254–9.
Palandri F et al. The long-term durability of cytogenetic responses in patients with accelerated phase chronic myeloid leukemia treated with imatinib 600 mg: the GIMEMA CML Working Party experience after a 7-year follow-up. Haematologica. 2009;94(2):205–12.
Silver RT et al. Four years of follow-up of 1027 patients with late chronic phase (L-CP), accelerated phase (AP), or blast crisis (BC) chronic myeloid leukemia (CML) treated with imatinib in three large phase II trials. Blood. 2004;104:Abstract 23.
Hochhaus A et al. Imatinib therapy in chronic myelogenous leukemia: strategies to avoid and overcome resistance. Leukemia. 2004;18:1321–31.
Ohanian M et al. Tyrosine kinase inhibitors as initial therapy for patients with chronic myeloid leukemia in accelerated phase. Clin Lymphoma Myeloma Leuk. 2014;14(2):155–62 e1.
Khoury HJ, et al. Omacetaxine mepesuccinate in patients with advanced chronic myeloid leukemia with resistance or intolerance to tyrosine kinase inhibitors. Leuk Lymphoma. 2014.
Radich JP et al. Allogeneic hematopoietic stem cell transplantation for chronic myeloid leukemia. Hematol Oncol Clin North Am. 2004;18(3):685–702.
Giralt SA et al. Impact of imatinib therapy on the use of allogeneic haematopoietic progenitor cell transplantation for the treatment of chronic myeloid leukaemia. Br J Haematol. 2007;137(5):461–7.
O’Brien S et al. NCCN clinical practice guidelines in oncology: chronic myelogenous leukemia. J Natl Compr Canc Netw. 2009;7(9):984–1023.
O’Brien S, Berman E, Moore JO, et al. NCCN Task Force report: tyrosine kinase inhibitor therapy selection in the management of patients with chronic myelogenous leukemia. J Natl Compr Canc Netw. 2011;9 Suppl 2:S1–25.
Jabbour E et al. Results of allogeneic hematopoietic stem cell transplantation for chronic myelogenous leukemia patients who failed tyrosine kinase inhibitors after developing BCR-ABL1 kinase domain mutations. Blood. 2011;117(13):3641–7.
Lee SJ et al. Impact of prior imatinib mesylate on the outcome of hematopoietic cell transplantation for chronic myeloid leukemia. Blood. 2008;112(8):3500–7.
Khoury HJ et al. Prognostic factors for outcomes in allogeneic transplantation for CML in the imatinib era: a CIBMTR analysis. Bone Marrow Transplant. 2012;47(6):810–6.
Oehler VG et al. The effects of imatinib mesylate treatment before allogeneic transplantation for chronic myeloid leukemia. Blood. 2007;109(4):1782–9.
Breccia M et al. Second-generation tyrosine kinase inhibitors before allogeneic stem cell transplantation in patients with chronic myeloid leukemia resistant to imatinib. Leuk Res. 2010;34(2):143–7.
Latif AL et al. Allogeneic stem cell transplantation for chronic myeloid leukaemia is safe and effective in high risk patients following second generation tyrosine kinase inhibitors: a single centre’s experience. Leuk Res Rep. 2013;2(2):47–50.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Chronic Myeloid Leukemias
Rights and permissions
About this article
Cite this article
Mukherjee, S., Kalaycio, M. Accelerated Phase CML: Outcomes in Newly Diagnosed vs. Progression From Chronic Phase. Curr Hematol Malig Rep 11, 86–93 (2016). https://doi.org/10.1007/s11899-016-0304-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11899-016-0304-7