Conclusions
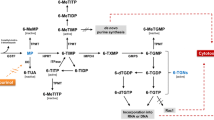
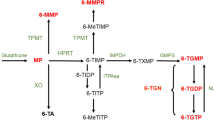
New strategies for optimizing thiopurine therapy of IBD are promising. TDM and TPMT genotyping seem to be important tools for fine tuning thiopurine therapy, thereby reducing toxicity and improving efficacy. ITPA genotyping and the use of 6-TG in IBD need further research before they can be advocated or banned.
Similar content being viewed by others
References
Adler DJ, Korelitz BI: The therapeutic efficacy of 6-mercaptopurine in refractory ulcerative colitis. Am J Gastroenterol 1990, 85:717–722.
Pearson DC, May GR, Fick GH, et al.: Azathioprine and 6-mercaptopurine in Crohn disease: a meta-analysis. Ann Intern Med 1995, 123:132–142.
Sandborn WJ: A review of immune modifier therapy for inflammatory bowel disease: azathioprine, 6-mercaptopurine, cyclosporine, and methotrexate. Am J Gastroenterol 1996, 91:423–433.
Kirschner BS: Safety of azathioprine and 6-mercaptopurine in pediatric patients with inflammatory bowel disease. Gastroenterology 1998, 115:813–821.
Present DH, Meltzer SJ, Krumholz MP, et al.: 6-Mercaptopurine in the management of inflammatory bowel disease: short- and long-term toxicity. Ann Intern Med 1989, 111:641–649.
Connell WR, Kamm MA, Ritchie JK, et al.: Bone marrow toxicity caused by azathioprine in inflammatory bowel disease: 27 years of experience. Gut 1993, 34:1081–1085.
Lennard L, Singleton HJ: High-performance liquid chromatographic assay of the methyl and nucleotide metabolites of 6-mercaptopurine: quantitation of red blood cell 6-thioguanine nucleotide, 6-thioinosinic acid and 6-methylmercaptopurine metabolites in a single sample. J Chromatogr 1992, 583:83–90.
Dervieux T, Boulieu R: Simultaneous determination of 6-thioguanine and methyl 6-mercaptopurine nucleotides of azathioprine in red blood cells by HPLC. Clin Chem 1998, 44:551–555.
Erdmann GR, France LA, Bostrom BC, et al.: A reversed phase high performance liquid chromatography approach in determining total red blood cell concentrations of 6-thioguanine, 6-mercaptopurine, methylthioguanine, and methylmercaptopurine in a patient receiving thiopurine therapy. Biomed Chromatogr 1990, 4:47–51.
Cuffari C, Theoret Y, Latour S, et al.: 6-Mercaptopurine metabolism in Crohn’s disease: correlation with efficacy and toxicity. Gut 1996, 39:401–406.
Dubinsky MC, Lamothe S, Yang HY, et al.: Pharmacogenomics and metabolite measurement for 6-mercaptopurine therapy in inflammatory bowel disease. Gastroenterology 2000, 118:705–713.
Cuffari C, Hunt S, Bayless T: Utilisation of erythrocyte 6- thioguanine metabolite levels to optimise azathioprine therapy in patients with inflammatory bowel disease. Gut 2001, 48:642–646.
Achkar JP, Stevens T, Easley K, et al.: Indicators of clinical response to treatment with six-mercaptopurine or azathioprine in patients with inflammatory bowel disease. Inflamm Bowel Dis 2004, 10:339–345.
Hindorf U, Lyrenas E, Nilsson A, et al.: Monitoring of long-term thiopurine therapy among adults with inflammatory bowel disease. Scand J Gastroenterol 2004, 39:1105–1112.
Dubinsky MC, Yang H, Hassard PV, et al.: 6-MP metabolite profiles provide a biochemical explanation for 6-MP resistance in patients with inflammatory bowel disease. Gastroenterology 2002, 122:904–915.
Belaiche J, Desager JP, Horsmans Y, et al.: Therapeutic drug monitoring of azathioprine and 6-mercaptopurine metabolites in Crohn disease. Scand J Gastroenterol 2001, 36:71–76.
Lowry PW, Franklin CL, Weaver AL, et al.: Measurement of thiopurine methyltransferase activity and azathioprine metabolites in patients with inflammatory bowel disease. Gut 2001, 49:665–670.
Gupta P, Gokhale R, Kirschner BS: 6-mercaptopurine metabolite levels in children with inflammatory bowel disease. J Pediatr Gastroenterol Nutr 2001, 33:450–454.
Goldenberg BA, Rawsthorne P, Bernstein CN: The utility of 6-thioguanine metabolite levels in managing patients with inflammatory bowel disease. Am J Gastroenterol 2004, 99:1744–1748.
Sandborn WJ: 6-MP metabolite levels: a potential guide to Crohn’s disease therapy. Gastroenterology 1997, 113:690–692.
Cohen RD: Forecast for using metabolite measurements in the dosing of azathioprine or 6-mercaptopurine for IBD patients: "partly cloudy". Gastroenterology 2002, 122:2082–2084, discussion 2084.
Derijks LJ, Gilissen LP, Engels LG, et al.: Pharmacokinetics of 6-mercaptopurine in patients with inflammatory bowel disease: implications for therapy. Ther Drug Monit 2004, 26:311–318.
Gilissen LP, Derijks LJ, Bos LP, et al.: Some cases demonstrating the clinical usefulness of therapeutic drug monitoring in thiopurine-treated inflammatory bowel disease patients. Eur J Gastroenterol Hepatol 2004, 16:705–710.
Gilissen LP, Derijks LJJ, Bos LP, et al.: Therapeutic drug monitoring in patients with inflammatory bowel disease and established azathioprine therapy. Clin Drug Invest 2004, 24:479–486.
Ansari A, Hassan C, Duley J, et al.: Thiopurine methyltransferase activity and the use of azathioprine in inflammatory bowel disease. Aliment Pharmacol Ther 2002, 16:1743–1750.
Black AJ, McLeod HL, Capell HA: Thiopurine methyltransferase genotype predicts therapy-limiting severe toxicity from azathioprine. Ann Intern Med 1998, 129:716–718.
Lennard L: TPMT in the treatment of Crohn’s disease with azathioprine. Gut 2002, 51:143–146.
Louis E, Belaiche J: Optimizing treatment with thioguanine derivatives in inflammatory bowel disease. Best Pract Res Clin Gastroenterol 2003, 17:37–46.
Colombel JF, Ferrari N, Debuysere H, et al.: Genotypic analysis of thiopurine S-methyltransferase in patients with Crohn’s disease and severe myelosuppression during azathioprine therapy. Gastroenterology 2000, 118:1025–1030.
Marinaki AM, Ansari A, Duley JA, et al.: Adverse drug reactions to azathioprine therapy are associated with polymorphism in the gene encoding inosine triphosphate pyrophosphatase (ITPase). Pharmacogenetics 2004, 14:181–187.
Gearry RB, Roberts RL, Barclay ML, et al.: Lack of association between the ITPA 94C< A polymorphism and adverse effects from azathioprine. Pharmacogenetics 2004, 14:779–781.
Allorge D, Hamdan R, Broly F, et al.: ITPA genotyping test does not improve detection of Crohn’s disease patients at risk of azathioprine/6-mercaptopurine induced myelosuppression. Gut 2005, 54:565.
Dubinsky MC, Hassard PV, Seidman EG, et al.: An openlabel pilot study using thioguanine as a therapeutic alternative in Crohn’s disease patients resistant to 6-mercaptopurine therapy. Inflamm Bowel Dis 2001, 7:181–189.
Baert F, Rutgeerts P: 6-Thioguanine: a naked bullet? (Or how pharmacogenomics can make old drugs brand new). Inflamm Bowel Dis 2001, 7:190–191.
Derijks LJ, De Jong DJ, Gilissen LP, et al.: 6-Thioguanine seems promising in azathioprineor 6-mercaptopurine-intolerant inflammatory bowel disease patients: a short-term safety assessment. Eur J Gastroenterol Hepatol 2003, 15:63–67.
Dubinsky MC, Feldman EJ, Abreu MT, et al.: Thioguanine: a potential alternate thiopurine for IBD patients allergic to 6-mercaptopurine or azathioprine. Am J Gastroenterol 2003, 98:1058–1063.
Bonaz B, Boitard J, Marteau P, et al.: Tioguanine in patients with Crohn’s disease intolerant or resistant to azathioprine/mercaptopurine. Aliment Pharmacol Ther 2003, 18:401–408.
Lancaster DL, Patel N, Lennard L, et al.: Leucocyte versus erythrocyte thioguanine nucleotide concentrations in children taking thiopurines for acute lymphoblastic leukaemia. Cancer Chemother Pharmacol 2002, 50:33–36.
Dervieux T, Blanco JG, Krynetski EY, et al.: Differing contribution of thiopurine methyltransferase to mercaptopurine versus thioguanine effects in human leukemic cells. Cancer Res 2001, 61:5810–5816.
Herrlinger KR, Kreisel W, Schwab M, et al.: 6-thioguanine-efficacy and safety in chronic active Crohn’s disease. Aliment Pharmacol Ther 2003, 17:503–508.
Herrlinger KR, Deibert P, Schwab M, et al.: Remission maintenance by tioguanine in chronic active Crohn’s disease. Aliment Pharmacol Ther 2003, 17:1459–1464.
Dubinsky MC, Vasiliauskas EA, Singh H, et al.: 6-thioguanine can cause serious liver injury in inflammatory bowel disease patients. Gastroenterology 2003, 125:298–303.
Geller SA, Dubinsky MC, Poordad FF, et al.: Early hepatic nodular hyperplasia and submicroscopic fibrosis associated with 6-thioguanine therapy in inflammatory bowel disease. Am J Surg Pathol 2004, 28:1204–1211.
Naber AH, Van Haelst U, Yap SH: Nodular regenerative hyperplasia of the liver: an important cause of portal hypertension in non-cirrhotic patients. J Hepatol 1991, 12:94–99.
Stromeyer FW, Ishak KG: Nodular transformation (nodular "regenerative "hyperplasia) of the liver: a clinicopathologic study of 30 cases. Hum Pathol 1981, 12:60–71.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Derijks, L.J.J., Hommes, D.W. Thiopurines in inflammatory bowel disease: New strategies for optimization of pharmacotherapy?. Curr Gastroenterol Rep 8, 89–92 (2006). https://doi.org/10.1007/s11894-006-0001-0
Issue Date:
DOI: https://doi.org/10.1007/s11894-006-0001-0