Abstract
Purpose of Review
Chronic inflammation, adipokines, and hepatokines have been identified as basis of insulin resistance and β cell failure in animal models. We present our current knowledge concerning the potential relationship between these cytokines, inflammation, metabolic syndrome (MetS), and type 2 diabetes mellitus (T2DM) in the pediatric population.
Recent Findings
Pro-inflammatory cytokines related to insulin resistance and MetS in children are tumor necrosis factor-alpha (TNF-α), interleukin (IL)-6, IL-1β, interferon gamma, pigment epithelium-derived factor, chemerin, vaspin, and fetuin A. Anti-inflammatory cytokines associated with insulin resistance and MetS in children are leptin, adiponectin, omentin, fibroblast growth factor (FGF)-21, osteocalcin, and irisin. These anti-inflammatory cytokines are decreased (adiponectin, omentin, and osteocalcin) or increased (leptin, FGF-21, and irisin) in obesity suggesting a resistance state. TNF-α, fetuin A, and FGF-21 are altered in obese children with T2DM suggesting an involvement in β cell failure.
Summary
These cytokines, adipokines, and hepatokines may be able to predict development of MetS and T2DM and have a potential therapeutic target ameliorating insulin resistance.
Similar content being viewed by others
Introduction
The metabolic syndrome (MetS) is defined as a clustering of obesity, hypertension, dyslipidemia, and impaired glucose tolerance [1, 2]. The MetS is diagnosed with increased frequency in children [3, 4]. The MetS leads to cardiovascular diseases and therefore to increased mortality [1, 2]. Even though the exact definition of the MetS in children is still under debate [5, 6], it is clear that the basis of MetS in childhood is insulin resistance [1, 5, 6]. When insulin resistance is associated with reduced β cell function and loss of β cell mass, then type 2 diabetes mellitus (T2DM) develops [7, 8].
It is now widely accepted that chronic inflammation plays an important role both in the development of insulin resistance and insulin secretion deficiency even if the mechanisms are not fully understood [9••, 10]. For example, high-sensitivity C-reactive protein (hsCRP) as an unspecific parameter of chronic inflammation is increased in obese insulin-resistant adults [11, 12], in obese children with MetS [13•, 14], and in obese adolescents with T2DM [15]. In addition to a direct link between chronic inflammation and insulin resistance, the involvement of adipokines, hepatokines, and cytokines of other tissues in the pathogenesis of MetS and T2DM is discussed [9••, 10, 16••, 17•].
The aim of this review is to demonstrate our current knowledge of inflammation markers in MetS and T2DM, which may offer important insights for future treatment strategies. This review focuses on the pediatric population since adverse patterns of MetS itself begin in childhood. Studies in children have also the advantage of not being influenced by other diseases, medications, or active tobacco smoking.
Inflammation as Trigger of Insulin Resistance and Insulin Secretion Deficiency
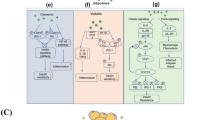
Adipose tissue produces a variety of pro- and anti-inflammatory cytokines. Proinflammatory cytokines promote insulin resistance by inducing uncontrolled insulin receptor substrates and anti-inflammatory cytokines have the opposite effect [18,19,20] (Fig. 1). Importantly, some pro-inflammatory cytokines also lead to β cell failure explaining at least in part the development of T2DM [10, 21,22,23,24,25,26]. Visceral adipose tissue produces more pro-inflammatory cytokines, whereas subcutaneous adipose tissue secretes larger quantities of anti-inflammatory and insulin-sensitizing cytokines [18, 27, 28]. Whether anti-inflammatory or pro-inflammatory cytokines are produced depends also on the regulation of a subset of T cells called CD4-positive T helper (Th) cells, which can be further differentiated into Th1 and Th2 cells [29]. Th1 cells promote a pro-inflammatory cytokine profile while Th2 cells promote an anti-inflammatory response.
Insulin and leptin signaling pathways. Binding of insulin to its receptor activates the signaling cascade of insulin receptor substrate (IRS) 1 and 2, phosphatidylinositol 3 kinase (PI3K), and protein kinase B (known as AKT). Binding of leptin to its receptor activates Janus kinase (JAK)2 which leads to activation of signal transducer and activator of transcription (STAT)3, IRS and also mitogen-activated-protein kinase (MAPK). Both pathways and also inflammatory molecules can induce the activation of suppressors of cytokine signaling 3 (SOCS3), which can inhibit insulin and leptin signaling
Furthermore, insulin resistance in muscle and liver tissue is increased by the release of free fatty acids (FFA) from adipose tissue, since FFA activate the innate immune system to release pro-inflammatory cytokines [28]. On the other hand, the increase of FFA is stimulated by proinflammatory cytokines, which enhances activity of hormone sensitive lipase in adipose tissue resulting in a vicious repetitive cycle [18, 28].
Inflammatory Markers Produced Generally
The following inflammatory markers are expressed in the adipose tissue but also generally.
Tumor Necrosis Factor-Alpha (TNF-α)
TNF-α is a pro-inflammatory cytokine synthesized as a 26-kDa transmembrane protein that undergoes cleavage by a metalloproteinase [19]. TNF-α induces phosphorylation of the insulin receptor substrate 1 (IRS-1) and thus prevents the interaction of insulin with an insulin receptor [18] (Fig. 1). Additionally, TNF-α enhances activity of hormone sensitive lipase in adipose tissue and thus increases the release of FFA into circulation [18]. Animal studies suggest that TNF-α is also related to β cell failure [10, 21,22,23,24,25,26].
In adults, an increased production of TNF-α in visceral adipose tissue correlates positively with the degree of obesity and insulin resistance [12, 30, 31]. In both cross-sectional and longitudinal pediatric studies, TNF-α is associated with parameters of insulin resistance, MetS, and degree of overweight [13•, 14, 15, 32,33,34]. Furthermore, TNF-α is higher in obese adolescents with T2DM compared to BMI-, age-, and gender- matched adolescents without T2DM [15]. These findings support the hypotheses that TNF-α is associated with insulin resistance and β cell failure.
Interleukin-6 (IL-6)
IL-6 is an interleukin circulating in multiple glycosylated forms ranging from 22 to 27 kDa [18, 27]. It acts as a pro-inflammatory cytokine inhibiting the insulin-signaling cascade by an impairment of insulin-induced insulin receptor and IRS-1 phosphorylation [18] (Fig. 1). Infusion of recombinant IL-6 in adults leads to an increase in hepatic glucose output and hyperglycemia [18]. Furthermore, IL-6 has a lipolytic effect with a consequent increase of FFA levels in circulation [18]. IL-6 concentrations are elevated in obese insulin-resistant adults [11, 35] as well as in obese insulin-resistant children [36, 37]. There is no difference of IL-6 levels between children with and without T2DM [38]. These findings support the role of IL-6 in the genesis of insulin resistance but do not confirm the hypothesis that IL-6 is involved in β cell failure.
Interleukin 1 Beta (IL-1β)
The pro-inflammatory cytokine IL-1β increases not only insulin resistance but also inhibits the function and promotes apoptosis of β cells in mouse models [21, 22, 39]. The blockade of IL-1 improves glycemia and β cell function and reduces inflammation in animal models of T2DM [40] and in humans with T2DM [21]. IL-1β concentrations are increased in adults with T2DM [11, 12, 30]. Obese children with MetS demonstrate higher concentrations of IL-1β than their lean counterparts [13•, 14]. IL-1β concentrations do not differ between obese adolescents with or without T2DM [15]. These findings do not support the hypothesis that IL-1β is involved in β cell failure but confirm the role of IL-1β in the genesis of insulin resistance and MetS in children.
Interferon Gamma (IFNγ)
Another pro-inflammatory cytokine suggested that to lead to β cell dysfunction is IFNγ. It is secreted by Th1 cells [22]. IFNγ is an important immune-activating cytokine that can prime macrophages for activation and induce inflammatory responses [41]. Obese adults with T2DM have higher concentrations of IFNγ [11, 12, 30, 31]. However, obese adolescents with T2DM do not have increased IFNγ levels [15]. Obese children have increased concentrations of IFNγ related to insulin resistance and other parameters of the MetS [42]. These findings do not support the hypothesis that IFNγ is involved in β cell failure but confirm the role of IFNγ in the genesis of insulin resistance and MetS in children.
Progranulin (PGRN)
PGRN is a widely expressed 593–amino acid glycoprotein, which has pro-inflammatory properties [43,44,45,46]. Full-length progranulin has anti-inflammatory activity [47], while proteolytic cleavage of progranulin generates granulin peptides, some of which promote inflammation [48]. Progranulin remarkably attenuate insulin sensitivity by inhibiting the insulin-signaling cascade by binding to the TNF-α receptor in cultured human adipocytes [45, 49]. PGRN promotes insulin resistance by increasing levels of IL-6 [45].
Data in humans are conflicting. Some studies report increased progranulin concentrations in obese adults with MetS or humans with T2DM, while others demonstrate no association between PGRN and MetS or T2DM [50,51,52]. Studies in obese children report no difference in respect to PGRN concentrations compared to their normal-weight counterparts as well as no relationship to parameters of the MetS or insulin resistance [53, 54]. Data in children with T2DM are lacking so far. Therefore, PGRN seems not to be related to insulin resistance or MetS in the pediatric population and the relationship to β cell failure is unclear.
Additional Inflammatory Cytokines
Besides the inflammatory cytokines mentioned above, several other widely expressed cytokines have been suggested to be related to insulin resistance. For example, obese insulin-resistant adults have significantly higher concentrations of monocyte chemoattractant protein-1 (MCP-1) and interleukin-8 (IL-8) concentrations [11, 12, 30, 31]. Obese children with parameters of the MetS demonstrate higher concentrations of MCP-1 and IL-8 than their lean counterparts [13•, 14, 32,33,34]. However, data concerning concentrations of these cytokines in adolescents with T2DM or MetS are not available so far. Therefore, it is unclear whether they play a role in insulin resistance, MetS, or T2DM in the pediatric population.
Adipokines
Cytokines secreted predominately from adipose tissue are called adipokines. Some of them also regulate the inflammation processes [55].
Leptin
Leptin is a 16-kDa peptide hormone mainly secreted by white adipose tissue [55]. It acts on the hypothalamus, leading to decreased appetite and increased energy expenditure, thereby regulating body weight [55]. Additionally, leptin acts on immune cells to stimulate the production of a spectrum anti-inflammatory cytokines [18]. Furthermore, binding of leptin to its receptor activates Janus kinase (JAK)2 which leads to activation of signal transducer and activator of transcription (STAT)3, IRS and also mitogen-activated-protein kinase (MAPK) (Fig. 1) [56]. These pathways and inflammatory molecules can induce the activation of suppressors of cytokine signaling 3 (SOCS3), which is an anti-inflammatory cytokine that suppresses cellular responses to inflammatory cytokines and also exerts an inhibitory feedback loop to leptin and insulin signaling [57]. This overlap between both signaling pathways via SOCS3 is suggested as a concomitant development of insulin and leptin resistance, potentially contributing to glucose intolerance and excess weight gain [56, 57]. On the other side, leptin improves directly insulin resistance in mouse models of T2DM through an increased oxidation of FFA [58].
Some studies report reduced leptin concentrations in adults with T2DM, while others show increased leptin levels or no difference of leptin levels between obese adults with and without T2DM [12, 59,60,61]. Two recent clinical trials show that leptin therapy is ineffective in improving diabetes and insulin resistance in obese people affected by T2DM [62, 63]. This might be explained at least in part by the observation that common obesity in humans is as a leptin resistant state [64, 65]. The potential mechanisms for leptin resistance include defective transport of leptin across the blood-brain barrier and defects in leptin receptor/post-receptor signaling cascade [64]. An increased pro-inflammatory response is reported in leptin resistance during obesity [66].
In children, hsCRP serum levels are related to leptin concentrations [67]. However, in contrast to the hypothesis of leptin resistance as a trigger of insulin resistance, serum leptin concentrations were lower in adolescents with T2DM compared to adolescents without T2DM [68]. Studies in adults [30] or children [59] reporting increased leptin levels in T2DM have not adjusted their control group to weight status, which is an important confounder since leptin is strongly associated to fat mass. Therefore, it is not proven that leptin resistance is a relevant mechanism in the development of insulin resistance, MetS, or T2DM in adolescents.
Adiponectin
Adiponectin exists as a full-length protein of 30 kDa and as a number of multimeric complexes [18]. Especially high molecular weight adiponectin are related to insulin sensitivity and T2DM [69, 70]. Adiponectin acts as a hormone with insulin-sensitizing properties in vitro and in animal models and may also promote β cell survival [71]. Even though adiponectin is secreted predominantly by adipose tissue, adiponectin concentrations are negatively correlated with fat mass in humans [70, 72]. Adiponectin levels are inversely correlated to insulin resistance and parameters of MetS in adults [70, 73, 74] and children [61, 75]. The reason for hypoadiponectinemia in obesity and insulin resistance is unclear. IL-6 and TNF-α decrease adiponectin mRNA in vitro [76]. Furthermore, prolonged exposure to insulin leads to a decrease in levels of adiponectin gene expression in adipocytes in vitro [76]. Therefore, hyperinsulinemia in insulin resistance might contribute to low adiponectin levels.
Hypoadiponectinemia correlates with the development of T2DM in adults [12, 77, 78], but the causal directions of this association is unclear. In manifest adult T2DM, some studies report reduced adiponectin concentrations, while others show no alteration of adiponectin levels [12, 30, 34, 61, 79]. In children with T2DM, reduced concentrations of adiponectin are reported [10, 59]. After adjusting for weight status however, adiponectin concentrations do not differ between obese adolescents with and without T2DM, but between obese children with and without MetS [68]. These findings suggest that adiponectin is associated with insulin resistance but not with T2DM. Mendelian randomization studies do not support a causal role for reduced circulating adiponectin levels in T2DM [80].
Adipocyte-Specific Fatty Acid-Binding Protein (A-FABP)
A-FABP belongs to the fatty acid-binding proteins and is present in adipocytes and macrophages [81]. Like most FABPs, A-FABP can bind with a variety of hydrophobic lipid ligands known to influence systemic inflammation [82]. Mice deficient in A-FABP is protected from development of hyperinsulinemia, hyperglycemia, and insulin resistance [81]. A-FABP concentrations are associated with insulin resistance and parameters of MetS in obese adults [81, 83]. In contrast, there is no relationship between A-FABP concentrations and insulin resistance or any parameters of the MetS in obese children [84]. Data concerning A-FABP in adolescents with T2DM are not available so far. Therefore, there are no data in the pediatric population supporting the hypothesis that A-FABP is related to insulin resistance, MetS, or T2DM.
Resistin
Resistin is a 12.5-kDa peptide produced by preadipocytes [18]. It was hypothesized that resistin has pro-inflammatory properties leading to insulin resistance [18]. Administration of resistin in healthy mice impairs glucose tolerance, whereas immunoneutralization of resistin in obese mice improves insulin sensitivity [85]. However, the role of resistin in human insulin resistance is less clear. Some studies documented that plasma resistin levels are elevated in obese adults with insulin resistance [86], whereas others reported that high-insulin-sensitive athletes have higher plasma resistin levels than obese subjects [87]. Data concerning resistin in adolescents with T2DM are not available so far. Resistin concentrations are not associated with parameters of the MetS or insulin resistance in obese children [88]. This is in line with the current hypothesis suggesting that the main significance of resistin in humans appears to regulate the inflammatory process rather than directly influencing insulin sensitivity [18].
Apelin
Apelin exists in at least three bioactive forms, consisting of 13, 17, or 36 amino acids, all originating from a common 77-amino acid precursor. Apelin is the endogenous ligand of the orphan G protein-coupled receptor [89]. Although synthesized in several tissues, apelin is expressed and secreted predominately by human adipocytes [90]. The most documented functions of apelin concern the regulation of fluid homeostasis and the modification of cardiac contractility and blood pressure [89, 91, 92]. Additionally, apelin inhibits insulin secretion in mice [89] suggesting a link between apelin and glucose homeostasis [93].
The situation in humans concerning apelin and its relation to insulin resistance is less clear. Some studies in adults demonstrate a positive correlation between apelin, insulin resistance, and parameters of MetS [94, 95], while other studies report the opposite [96, 97]. In children, no association between apelin and insulin resistance or parameters of MetS could be detected [98, 99]. Data concerning apelin in adolescents with T2DM are not available so far. Therefore, there are no data in the pediatric population supporting the hypothesis that apelin is related to insulin resistance, MetS, or T2DM.
Visfatin
Visfatin is a 52-kDa pre-B cell colony-enhancing factor expressed in peripheral blood lymphocytes [18] and visceral adipose tissue [100]. Similar to insulin, visfatin in vitro enhances glucose uptake by adipocytes and myocytes and inhibits hepatocyte glucose release [100]. Visfatin’s insulin-like effects are observed in the phosphorylation of insulin receptors IRS-1 and IRS-2. Interestingly, visfatin and insulin have the same affinity for the insulin receptor but interact with the insulin receptor at different site. Furthermore, visfatin regulates intracellular activity of the NAD/NADH dependent enzymes that are critical for glucose-stimulated insulin secretion in pancreatic β cells [101]. Clinical studies in adults provide controversial findings concerning the role of visfatin glucose metabolism with positive, negative, or no associations found [102,103,104]. In children, no association between visfatin and insulin resistance is reported [105]. Data concerning visfatin in adolescents with T2DM are not available so far. Therefore, there are no data in children supporting the hypothesis that visfatin is related to insulin resistance, MetS, or T2DM.
Omentin
Omentin is a 38–40-kDa adipokine preferentially produced by visceral adipose tissue [106]. Omentin enhances insulin-stimulated glucose uptake in human adipocytes [106, 107]. Expression of omentin in visceral adipose tissue is reduced in insulin resistance [106, 107]. Omentin leads to suppression of TNF-α-induced vascular inflammation in cell models [108]. Omentin upregulates adiponectin gene expression [109]. In children, serum omentin levels correlate negatively with insulin resistance [110]. Obese children with MetS have lower omentin serum levels compared to obese children without MetS [111, 112]. Data concerning omentin in adolescents with T2DM are not available so far. These findings support the role of omentin in the genesis of insulin resistance and MetS, while its protective effect on β cells is unclear in the pediatric population.
Pigment Epithelium-Derived Factor (PEDF)
PEDF is a 50-kDa secreted glycoprotein belonging to the serine protease inhibitor (serpin) family [18]. Recombinant PEDF activates macrophages to release TNF-α and IL-1 [113]. Additionally, PEDF promotes lipolysis in an adipose triglyceride lipase-dependent manner and mobilizes FFA into systemic circulation leading to inflammation [18]. PEDF provokes kinase-mediated inhibitory phosphorylation cascade of IRS that attenuates insulin signaling and induces insulin resistance in peripheral tissues [18]. Furthermore, PEDF might be the link between insulin resistance and acanthosis nigricans [114].
Administration of recombinant PEDF reduces insulin sensitivity during hyperinsulinemic-euglycemic clamp in mice, whereas neutralization of PEDF restores insulin sensitivity [115]. PEDF expression in adipose tissue positively correlates with obesity and insulin resistance in mice [115]. In adults, PEDF correlates better to insulin resistance than to the degree of obesity [116]. In children, plasma PEDF is positively associated with insulin resistance [117]. However, PEDF concentrations are similar in obese children with and without T2DM [118]. These findings support the role of PEDF in the genesis of insulin resistance and MetS, but do not confirm the hypothesis that PEDF is involved in β cell failure.
Chemerin
Chemerin is an adipokine highly expressed in white adipose tissue and in the liver [119, 120]. It is unclear whether chemerin increases insulin resistance or if chemerin is an adaptive hormone to improve insulin resistance since conflicting data exist regarding the effect of chemerin on insulin signaling in adipocytes in vitro. Chemerin has been shown to downregulate insulin-stimulated glucose uptake in adipocytes [121], while another study reported the opposite [122]. Chemerin might also influence β cell function [123]. TNF-α stimulates mRNA levels of chemerin in visceral adipocytes from obese patients [124]. Injection of recombinant human chemerin exacerbates glucose intolerance, lowers serum insulin levels, and decreases tissue glucose uptake in obese mice [125].
Chemerin is associated with components of MetS in some studies of adults [126, 127], while another study does not find any relationship between parameters of MetS, insulin resistance, and chemerin [128]. In children, serum concentrations of chemerin are related both cross-sectionally and longitudinally to the degree of insulin resistance, amount of fat mass, and severity of MetS [54, 129,130,131,132,133]. Data concerning chemerin in adolescents with T2DM are not available so far. These findings support the role of chemerin in the genesis of insulin resistance and MetS, while the situation in T2DM is less clear in children.
Vaspin
Vaspin belongs to the serpin superfamily and is serine protease inhibitor with insulin-sensitizing effects [134]. It is produced predominately in the visceral adipose tissue [134]. Recombinant vaspin administration in obese mice improves insulin sensitivity [134]. In adults, vaspin correlates with fat mass and insulin resistance [135, 136]. Obese children with MetS have higher vaspin concentrations than obese children without MetS [111, 137]. T2DM seems to abrogate the correlation between increased circulating vaspin, higher body weight, and decreased insulin sensitivity in adults [138]. Data concerning vaspin in adolescents with T2DM are not available so far. These findings support the role of vaspin in the genesis of insulin resistance and MetS, while the situation in T2DM is unclear in children.
Other Adipokines
There are several other adipokines which have been proposed to be linked to insulin resistance, parameters of MetS, and T2DM such as adipolin [18, 139]. However, today, there are only few studies of these adipokines in insulin-resistant humans and studies in children are especially missing.
Hepatokines
Cytokine secreted by the liver, so-called hepatokines, have also been proposed to be related to chronic inflammation, insulin resistance, parameters of MetS, and T2DM [140,141,142,143,144,145,146,147,148]. For example, hepatocyte-secreted dipeptidyl peptidase 4 (DPP4) in obese mice promotes adipose inflammation and insulin resistance [149].
Fibroblast Growth Factor (FGF)-21
FGF-21 is an insulin-sensitizing hepatokine. It is mainly produced by the liver but also by other tissues including white adipose tissue, skeletal muscle, and pancreatic β cells [150]. FGF-21 induces glucose uptake and decreases glucose concentrations in obese animals [142, 151]. Adults with insulin resistance or T2DM have higher FGF-21 serum levels pointing to FGF-21 resistance [143, 152, 153]. FGF-21 resistance can be mediated through altered expression of both the FGF-21 receptor and the adapter molecule β-klotho [154]. Interestingly, TNF-α represses β-klotho expression and impairs FGF-21 action in adipose cells [148]. In concordance, FGF-21 concentrations are higher in obese adolescents with T2DM compared to obese adolescents without T2DM [155]. These findings suggest a FGF-21 resistance state in the genesis of insulin resistance and β cell failure in the pediatric population.
Fetuin A
Another protein secreted by the liver, fetuin A, is also proposed as a link between insulin resistance, MetS, and T2DM [156]. Fetuin A inhibits insulin receptor tyrosine kinase activity in muscles [157, 158] and induces cytokine expression and low-grade inflammation in animal models [147]. Additionally, fetuin A represses adiponectin production in animals [147]. Higher fetuin A concentrations are associated with the degree of insulin resistance, severity of MetS, and the presence of T2DM in adults [145, 159]. Furthermore, fetuin A is associated both cross-sectionally and longitudinally with insulin resistance and parameters of the MetS in obese children [15, 160]. Fetuin A serum levels are higher in obese adolescents with T2DM compared to age-, gender-, and BMI-matched adolescents without T2DM [155]. These findings support the role of fetuin A in the development of insulin resistance and β cell failure in the pediatric population.
Other Tissues
The skeleton and muscle tissue are now regarded as endocrine organs secreting cytokines that affect glucose metabolism.
Osteocalcin
Osteocalcin, a marker of bone formation specifically synthesized and secreted by osteoblasts, has also insulin-sensitizing properties [161]. Adults with T2DM have low circulating osteocalcin levels [162, 163]. Osteocalcin is associated negatively with insulin resistance and parameters of MetS in cross-sectional and longitudinal analyses of obese children [164]. Data concerning osteocalcin in adolescents with T2DM are not available so far. These findings support the role of osteocalcin in the genesis of insulin resistance and MetS in children, while the situation in T2DM is less clear.
Irisin
Irisin, a myokine induced by exercise, is another cytokine with insulin-sensitizing properties [165, 166]. Circulating irisin results from C-terminal cleavage of the fibronectin type III domain containing five transmembrane protein [167]. This process is induced by the peroxisome proliferator-activated receptor (PPAR)-γ coactivator (PGC)-1α [168]. Irisin concentrations increase immediately after exercise and correlate with exercise intensity [169]. Since irisin induces glucose and fatty acid uptake in human muscle, the increase of irisin in insulin-resistant obese humans in most studies suggests a state of irisin resistance [170, 171]. Data concerning irisin in adolescents with T2DM are not available so far. Accordingly to most previous studies in adults [165, 166, 172], irisin is associated with insulin resistance and other parameters of the MetS both in cross-sectional and longitudinal analyses in obese children [173]. These findings support an irisin resistant state in the genesis of insulin resistance and MetS in children, while the situation in T2DM is unclear.
Conclusions
There are a growing number of discovered inflammatory markers, adipokines, and hepatokines, which have been hypothesized to be linked to inflammation and consequently insulin resistance, MetS, and development of T2DM. While for some of these parameters, this hypothesis could not be confirmed in the pediatric population (no impact of progranulin, A-FABP, resistin, apelin, or visfatin on MetS, insulin resistance, or T2DM), a significant association of other parameters to MetS, and T2DM were confirmed in the children (Fig. 2): Pro-inflammatory cytokines related to insulin resistance and MetS in children are TNF-α, IL-6, IL-1β, IFNγ, PEDF, leptin, chemerin, vaspin, and fetuin A. The serum concentrations of all of them are increased in obesity. Anti-inflammatory cytokines associated with insulin resistance and MetS in children are adiponectin, omentin, FGF-21, osteocalcin, and irisin. These anti-inflammatory are decreased (adiponectin, omentin, and osteocalcin) or increased (FGF-21 and irisin) in obesity, the latter suggesting a resistance state. The pro-inflammatory cytokines TNF-α and fetuin A, and the anti-inflammatory metabolic factor FGF-21 are altered in obese children with T2DM pointing toward an involvement in β cell failure.
The presented data in the pediatric population support the hypothesis that systemic inflammation is one causative link between obesity, insulin resistance, MetS, and T2DM. In the future, elucidating pathways of these cytokines, adipokines, and hepatokines may yield a potential therapeutic target in ameliorating insulin resistance and preventing T2DM.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Eckel RH, Grundy SM, Zimmet PZ. The metabolic syndrome. Lancet. 2005;365(9468):1415–28.
Franks PW, Hanson RL, Knowler WC, Sievers ML, Bennett PH, Looker HC. Childhood obesity, other cardiovascular risk factors, and premature death. N Engl J Med. 2010;362(6):485–93.
Viner RM, Segal TY, Lichtarowicz-Krynska E, Hindmarsh P. Prevalence of the insulin resistance syndrome in obesity. Arch Dis Child. 2005;90(1):10–4.
Zimmet P, Alberti KG, Kaufman F, et al. The metabolic syndrome in children and adolescents - an IDF consensus report. Pediatr Diabetes. 2007;8(5):299–306.
Reinehr T. Metabolic syndrome in children and adolescents: a critical approach considering the interaction between pubertal stage and insulin resistance. Curr Diab Rep. 2016;16(1):8.
Magge SN, Goodman E, Armstrong SC. The metabolic syndrome in children and adolescents: shifting the focus to cardiometabolic risk factor clustering. Pediatrics. 2017;140:e20171603.
Stumvoll M, Goldstein BJ, van Haeften TW. Type 2 diabetes: principles of pathogenesis and therapy. Lancet. 2005;365(9467):1333–46.
Kahn SE. The relative contributions of insulin resistance and beta-cell dysfunction to the pathophysiology of Type 2 diabetes. Diabetologia. 2003;46(1):3–19.
•• Donath MY, Shoelson SE. Type 2 diabetes as an inflammatory disease. Nat Rev Immunol. 2011;11(2):98–107 This paper summarizes our knowledge concerning the relationship between inflammation and type 2 diabetes.
Rempel JD, Packiasamy J, Dean HJ, McGavock J, Janke A, Collister M, et al. Preliminary analysis of immune activation in early onset type 2 diabetes. Int J Circumpolar Health. 2013;72.
Lucas R, Parikh SJ, Sridhar S, Guo DH, Bhagatwala J, Dong Y, et al. Cytokine profiling of young overweight and obese female African American adults with prediabetes. Cytokine. 2013;64(1):310–5.
Al-Hamodi Z, Al-Habori M, Al-Meeri A, Saif-Ali R. Association of adipokines, leptin/adiponectin ratio and C-reactive protein with obesity and type 2 diabetes mellitus. Diabetol Metab Syndr. 2014;6(1):99.
• Roth CL, Kratz M, Ralston MM, Reinehr T. Changes in adipose-derived inflammatory cytokines and chemokines after successful lifestyle intervention in obese children. Metabolism. 2011;60(4):445–52 This paper demonstrates the change of adipokines in obese children with weight loss.
Gungor N, Thompson T, Sutton-Tyrrell K, Janosky J, Arslanian S. Early signs of cardiovascular disease in youth with obesity and type 2 diabetes. Diabetes Care. 2005;28(5):1219–21.
Reinehr T, Karges B, Meissner T, Wiegand S, Stoffel-Wagner B, Holl RW, et al. Inflammatory markers in obese adolescents with type 2 diabetes and their relationship to hepatokines and adipokines. J Pediatr. 2016;173:131–5.
•• Bluher M. Adipokines - removing road blocks to obesity and diabetes therapy. Mol Metab. 2014;3(3):230–40 This paper shows the potential of modifying adipokines as a treatment option in obesity and diabetes.
• Roth CL, Reinehr T. Roles of gastrointestinal and adipose tissue peptides in childhood obesity and changes after weight loss due to lifestyle intervention. Arch Pediatr Adolesc Med. 2010;164(2):131–8 This review summarizes the changes of adipokine and GI hormones in obese children with successful weight loss.
Smitka K, Maresova D. Adipose tissue as an endocrine organ: an update on pro-inflammatory and anti-inflammatory microenvironment. Prague Med Rep. 2015;116(2):87–111.
Hotamisligil GS, Spiegelman BM. Tumor necrosis factor alpha: a key component of the obesity-diabetes link. Diabetes. 1994;43(11):1271–8.
Tanti JF, Jager J. Cellular mechanisms of insulin resistance: role of stress-regulated serine kinases and insulin receptor substrates (IRS) serine phosphorylation. Curr Opin Pharmacol. 2009;9(6):753–62.
Larsen CM, Faulenbach M, Vaag A, Vølund A, Ehses JA, Seifert B, et al. Interleukin-1-receptor antagonist in type 2 diabetes mellitus. N Engl J Med. 2007;356(15):1517–26.
Ibarra UA, Friberg J, Christensen DP, Lund CG, Billestrup N. Inflammatory cytokines stimulate bone morphogenetic protein-2 expression and release from pancreatic beta cells. J Interf Cytokine Res. 2015.
Eder K, Baffy N, Falus A, Fulop AK. The major inflammatory mediator interleukin-6 and obesity. Inflamm Res. 2009;58(11):727–36.
Vanderford NL. Defining the regulation of IL-1beta- and CHOP-mediated beta-cell apoptosis. Islets. 2010;2(5):334–6.
Fogeda M, Gallart L, Gutierrez C, Vendrell J, Simón I, García-España A, et al. High expression of tumor necrosis factor alpha receptors in peripheral blood mononuclear cells of obese type 2 diabetic women. Eur Cytokine Netw. 2004;15(1):60–6.
Dinarello CA. A clinical perspective of IL-1beta as the gatekeeper of inflammation. Eur J Immunol. 2011;41(5):1203–17.
Fontana L, Eagon JC, Trujillo ME, Scherer PE, Klein S. Visceral fat adipokine secretion is associated with systemic inflammation in obese humans. Diabetes. 2007;56(4):1010–3.
Sjoholm A, Nystrom T. Inflammation and the etiology of type 2 diabetes. Diabetes Metab Res Rev. 2006;22(1):4–10.
Moss RB, Moll T, El-Kalay M, et al. Th1/Th2 cells in inflammatory disease states: therapeutic implications. Expert Opin Biol Ther. 2004;4(12):1887–96.
Rajkovic N, Zamaklar M, Lalic K, Jotic A, Lukic L, Milicic T, et al. Relationship between obesity, adipocytokines and inflammatory markers in type 2 diabetes: relevance for cardiovascular risk prevention. Int J Environ Res Public Health. 2014;11(4):4049–65.
Popko K, Gorska E, Stelmaszczyk-Emmel A, Plywaczewski R, Stoklosa A, Gorecka D, et al. Proinflammatory cytokines Il-6 and TNF-alpha and the development of inflammation in obese subjects. Eur J Med Res. 2010;15(Suppl 2):120–2.
Su SC, Pei D, Hsieh CH, Hsiao FC, Wu CZ, Hung YJ. Circulating pro-inflammatory cytokines and adiponectin in young men with type 2 diabetes. Acta Diabetol. 2011;48(2):113–9.
Nadeau KJ, Zeitler PS, Bauer TA, Brown MS, Dorosz JL, Draznin B, et al. Insulin resistance in adolescents with type 2 diabetes is associated with impaired exercise capacity. J Clin Endocrinol Metab. 2009;94(10):3687–95.
West NA, Hamman RF, Mayer-Davis EJ, D’Agostino RB, Marcovina SM, Liese AD, et al. Cardiovascular risk factors among youth with and without type 2 diabetes: differences and possible mechanisms. Diabetes Care. 2009;32(1):175–80.
Klover PJ, Clementi AH, Mooney RA. Interleukin-6 depletion selectively improves hepatic insulin action in obesity. Endocrinology. 2005;146(8):3417–27.
Hershkop K, Besor O, Santoro N, Pierpont B, Caprio S, Weiss R. Adipose insulin resistance in obese adolescents across the spectrum of glucose tolerance. J Clin Endocrinol Metab. 2016;101(6):2423–31.
De Filippo G, Rendina D, Moccia F, Rocco V, Campanozzi A. Interleukin-6, soluble interleukin-6 receptor/interleukin-6 complex and insulin resistance in obese children and adolescents. J Endocrinol Investig. 2015;38(3):339–43.
Shalitin S, Deutsch V, Tauman R. Hepcidin, soluble transferrin receptor and IL-6 levels in obese children and adolescents with and without type 2 diabetes mellitus/impaired glucose tolerance and their association with obstructive sleep apnea. J Endocrinol Investig. 2018;41(8):969–75.
Sopasakis VR, Nagaev I, Smith U. Cytokine release from adipose tissue of nonobese individuals. Int J Obes. 2005;29(9):1144–7.
Ehses JA, Lacraz G, Giroix MH, Schmidlin F, Coulaud J, Kassis N, et al. IL-1 antagonism reduces hyperglycemia and tissue inflammation in the type 2 diabetic GK rat. Proc Natl Acad Sci U S A. 2009;106(33):13998–4003.
Hansson GK. Inflammation and immune response in atherosclerosis. Curr Atheroscler Rep. 1999;1(2):150–5.
Pacifico L, Di RL, Anania C, Osborn JF, Ippoliti F, Schiavo E, et al. Increased T-helper interferon-gamma-secreting cells in obese children. Eur J Endocrinol. 2006;154(5):691–7.
Wei Z, Huang Y, Xie N, Ma Q. Elevated expression of secreted autocrine growth factor progranulin increases cervical cancer growth. Cell Biochem Biophys. 2015;71(1):189–93.
Luo L, Lu L, Lu Y, Zhang L, Li B, Guo K, et al. Effects of hypoxia on progranulin expression in HT22 mouse hippocampal cells. Mol Med Rep. 2014;9(5):1675–80.
Matsubara T, Mita A, Minami K, Hosooka T, Kitazawa S, Takahashi K, et al. PGRN is a key adipokine mediating high fat diet-induced insulin resistance and obesity through IL-6 in adipose tissue. Cell Metab. 2012;15(1):38–50.
Youn BS, Bang SI, Kloting N, Park JW, Lee N, Oh JE, et al. Serum progranulin concentrations may be associated with macrophage infiltration into omental adipose tissue. Diabetes. 2009;58(3):627–36.
Eriksen JL, Mackenzie IR. Progranulin: normal function and role in neurodegeneration. J Neurochem. 2008;104(2):287–97.
Tolkatchev D, Malik S, Vinogradova A, Wang P, Chen Z, Xu P, et al. Structure dissection of human progranulin identifies well-folded granulin/epithelin modules with unique functional activities. Protein Sci. 2008;17(4):711–24.
Tang W, Lu Y, Tian QY, Zhang Y, Guo FJ, Liu GY, et al. The growth factor progranulin binds to TNF receptors and is therapeutic against inflammatory arthritis in mice. Science. 2011;332(6028):478–84.
Qu H, Deng H, Hu Z. Plasma progranulin concentrations are increased in patients with type 2 diabetes and obesity and correlated with insulin resistance. Mediat Inflamm. 2013;2013:360190.
Li H, Zhou B, Xu L, Liu J, Zang W, Wu S, et al. Circulating PGRN is significantly associated with systemic insulin sensitivity and autophagic activity in metabolic syndrome. Endocrinology. 2014;155(9):3493–507.
Esposito K, Giugliano D. The metabolic syndrome and inflammation: association or causation? Nutr Metab Cardiovasc Dis. 2004;14(5):228–32.
Alissa EM, Sutaih RH, Kamfar HZ, Alagha AE, Marzouki ZM. Serum progranulin levels in relation to insulin resistance in childhood obesity. J Pediatr Endocrinol Metab. 2017;30(12):1251–6.
Daxer J, Herttrich T, Zhao YY, Vogel M, Hiemisch A, Scheuermann K, et al. Nocturnal levels of chemerin and progranulin in adolescents: influence of sex, body mass index, glucose metabolism and sleep. J Pediatr Endocrinol Metab. 2017;30(1):57–61.
Koerner A, Kratzsch J, Kiess W. Adipocytokines: leptin--the classical, resistin--the controversical, adiponectin--the promising, and more to come. Best Pract Res Clin Endocrinol Metab. 2005;19(4):525–46.
Lustig RH. Childhood obesity: behavioral aberration or biochemical drive? Reinterpreting the first law of thermodynamics. Nat Clin Pract Endocrinol Metab. 2006;2(8):447–58.
Iikuni N, Lam QL, Lu L, Matarese G, La CA. Leptin and inflammation. Curr Immunol Rev. 2008;4(2):70–9.
Berglund ED, Vianna CR, Donato J Jr, Kim MH, Chuang JC, Lee CE, et al. Direct leptin action on POMC neurons regulates glucose homeostasis and hepatic insulin sensitivity in mice. J Clin Invest. 2012;122(3):1000–9.
Morales A, Wasserfall C, Brusko T, Carter C, Schatz D, Silverstein J, et al. Adiponectin and leptin concentrations may aid in discriminating disease forms in children and adolescents with type 1 and type 2 diabetes. Diabetes Care. 2004;27(8):2010–4.
Abdelgadir M, Elbagir M, Eltom M, Berne C, Ahren B. Reduced leptin concentrations in subjects with type 2 diabetes mellitus in Sudan. Metabolism. 2002;51(3):304–6.
Stringer DM, Sellers EA, Burr LL, Taylor CG. Altered plasma adipokines and markers of oxidative stress suggest increased risk of cardiovascular disease in First Nation youth with obesity or type 2 diabetes mellitus. Pediatr Diabetes. 2009;10(4):269–77.
Moon HS, Matarese G, Brennan AM, Chamberland JP, Liu X, Fiorenza CG, et al. Efficacy of metreleptin in obese patients with type 2 diabetes: cellular and molecular pathways underlying leptin tolerance. Diabetes. 2011;60(6):1647–56.
Mittendorfer B, Horowitz JF, DePaoli AM, McCamish MA, Patterson BW, Klein S. Recombinant human leptin treatment does not improve insulin action in obese subjects with type 2 diabetes. Diabetes. 2011;60(5):1474–7.
Bjorbaek C, Kahn BB. Leptin signaling in the central nervous system and the periphery. Recent Prog Horm Res. 2004;59:305–31.
Martin SS, Qasim A, Reilly MP. Leptin resistance: a possible interface of inflammation and metabolism in obesity-related cardiovascular disease. J Am Coll Cardiol. 2008;52(15):1201–10.
Vazquez-Vela ME, Torres N, Tovar AR. White adipose tissue as endocrine organ and its role in obesity. Arch Med Res. 2008;39(8):715–28.
Navarro P, de DO, Gavela-Perez T, Jois A, Garces C, Soriano-Guillen L. High-sensitivity C-reactive protein and leptin levels related to body mass index changes throughout childhood. J Pediatr. 2016;178:178–82.
Reinehr T, Woelfle J, Wiegand S, et al. Leptin but not adiponectin is related to type 2 diabetes mellitus in obese adolescents. Pediatr Diabetes. 2016 Jun;17(4):281–8. https://doi.org/10.1111/pedi.12276.
Waki H, Yamauchi T, Kamon J, Ito Y, Uchida S, Kita S, et al. Impaired multimerization of human adiponectin mutants associated with diabetes. Molecular structure and multimer formation of adiponectin. J Biol Chem. 2003;278(41):40352–63.
Lau DC, Dhillon B, Yan H, Szmitko PE, Verma S. Adipokines: molecular links between obesity and atheroslcerosis. Am J Physiol Heart Circ Physiol. 2005;288(5):H2031–41.
Turer AT, Scherer PE. Adiponectin: mechanistic insights and clinical implications. Diabetologia. 2012;55(9):2319–26.
Statnick MA, Beavers LS, Conner LJ, Corominola H, Johnson D, Hammond CD, et al. Decreased expression of apM1 in omental and subcutaneous adipose tissue of humans with type 2 diabetes. Int J Exp Diabetes Res. 2000;1(2):81–8.
Weyer C, Funahashi T, Tanaka S, Hotta K, Matsuzawa Y, Pratley RE, et al. Hypoadiponectinemia in obesity and type 2 diabetes: close association with insulin resistance and hyperinsulinemia. J Clin Endocrinol Metab. 2001;86(5):1930–5.
Herder C, Schneitler S, Rathmann W, Haastert B, Schneitler H, Winkler H, et al. Low-grade inflammation, obesity, and insulin resistance in adolescents. J Clin Endocrinol Metab. 2007;92(12):4569–74.
Reinehr T, Roth C, Menke T, Andler W. Adiponectin before and after weight loss in obese children. J Clin Endocrinol Metab. 2004;89(8):3790–4.
Fasshauer M, Klein J, Neumann S, Eszlinger M, Paschke R. Hormonal regulation of adiponectin gene expression in 3T3-L1 adipocytes. Biochem Biophys Res Commun. 2002;290(3):1084–9.
Tabak AG, Brunner EJ, Miller MA, Karanam S, McTernan PG, Cappuccio FP, et al. Low serum adiponectin predicts 10-year risk of type 2 diabetes and HbA1c independently of obesity, lipids, and inflammation: Whitehall II study. Horm Metab Res. 2009;41(8):626–9.
Li S, Shin HJ, Ding EL, van Dam RM. Adiponectin levels and risk of type 2 diabetes: a systematic review and meta-analysis. JAMA. 2009;302(2):179–88.
Coimbra S, Brandao PJ, Santos-Silva A, Neuparth MJ. Adiponectin, leptin, and chemerin in elderly patients with type 2 diabetes mellitus: a close linkage with obesity and length of the disease. Biomed Res Int. 2014;2014:701915.
Yaghootkar H, Lamina C, Scott RA, Dastani Z, Hivert MF, Warren LL, et al. Mendelian randomization studies do not support a causal role for reduced circulating adiponectin levels in insulin resistance and type 2 diabetes. Diabetes. 2013;62(10):3589–98.
Boord JB, Fazio S, Linton MF. Cytoplasmic fatty acid-binding proteins: emerging roles in metabolism and atherosclerosis. Curr Opin Lipidol. 2002;13(2):141–7.
Maeda K, Cao H, Kono K, Gorgun CZ, Furuhashi M, Uysal KT, et al. Adipocyte/macrophage fatty acid binding proteins control integrated metabolic responses in obesity and diabetes. Cell Metab. 2005;1(2):107–19.
Makowski L, Hotamisligil GS. Fatty acid binding proteins--the evolutionary crossroads of inflammatory and metabolic responses. J Nutr. 2004;134(9):2464S–8S.
Reinehr T, Stoffel-Wagner B, Roth CL. Adipocyte fatty acid-binding protein in obese children before and after weight loss. Metabolism. 2007;56(12):1735–41.
Steppan CM, Lazar MA. Resistin and obesity-associated insulin resistance. Trends Endocrinol Metab. 2002;13(1):18–23.
Norata GD, Ongari M, Garlaschelli K, Raselli S, Grigore L, Catapano AL. Plasma resistin levels correlate with determinants of the metabolic syndrome. Eur J Endocrinol. 2007;156(2):279–84.
Perseghin G, Burska A, Lattuada G, Alberti G, Costantino F, Ragogna F, et al. Increased serum resistin in elite endurance athletes with high insulin sensitivity. Diabetologia. 2006;49(8):1893–900.
Reinehr T, Roth CL, Menke T, Andler W. Resistin concentrations before and after weight loss in obese children. Int J Obes. 2006;30(2):297–301.
Castan-Laurell I, Boucher J, Dray C, Daviaud D, Guigne C, Valet P. Apelin, a novel adipokine over-produced in obesity: friend or foe? Mol Cell Endocrinol. 2005;245(1–2):7–9.
Boucher J, Masri B, Daviaud D, Gesta S, Guigné C, Mazzucotelli A, et al. Apelin, a newly identified adipokine up-regulated by insulin and obesity. Endocrinology. 2005;146(4):1764–71.
Karmazyn M, Purdham DM, Rajapurohitam V, Zeidan A. Signalling mechanisms underlying the metabolic and other effects of adipokines on the heart. Cardiovasc Res. 2008;79(2):279–86.
Wei L, Hou X, Tatemoto K. Regulation of apelin mRNA expression by insulin and glucocorticoids in mouse 3T3-L1 adipocytes. Regul Pept. 2005;132(1–3):27–32.
Yue P, Jin H, Aillaud M, Deng AC, Azuma J, Asagami T, et al. Apelin is necessary for the maintenance of insulin sensitivity. Am J Physiol Endocrinol Metab. 2010;298(1):E59–67.
Soriguer F, Garrido-Sanchez L, Garcia-Serrano S, Garcia-Almeida JM, Garcia-Arnes J, Tinahones FJ, et al. Apelin levels are increased in morbidly obese subjects with type 2 diabetes mellitus. Obes Surg. 2009;19(11):1574–80.
Li L, Yang G, Li Q, Tang Y, Yang M, Yang H, et al. Changes and relations of circulating visfatin, apelin, and resistin levels in normal, impaired glucose tolerance, and type 2 diabetic subjects. Exp Clin Endocrinol Diabetes. 2006;114(10):544–8.
Heinonen MV, Laaksonen DE, Karhu T, Karhunen L, Laitinen T, Kainulainen S, et al. Effect of diet-induced weight loss on plasma apelin and cytokine levels in individuals with the metabolic syndrome. Nutr Metab Cardiovasc Dis. 2009;19(9):626–33.
Castan-Laurell I, Vitkova M, Daviaud D, Dray C, Kováciková M, Kovacova Z, et al. Effect of hypocaloric diet-induced weight loss in obese women on plasma apelin and adipose tissue expression of apelin and APJ. Eur J Endocrinol. 2008;158(6):905–10.
Tapan S, Tascilar E, Abaci A, Sonmez A, Kilic S, Erbil MK, et al. Decreased plasma apelin levels in pubertal obese children. J Pediatr Endocrinol Metab. 2010;23(10):1039–46.
Reinehr T, Woelfle J, Roth CL. Lack of association between apelin, insulin resistance, cardiovascular risk factors, and obesity in children: a longitudinal analysis. Metabolism. 2011;60:1349–54.
Fukuhara A, Matsuda M, Nishizawa M, Segawa K, Tanaka M, Kishimoto K, et al. Visfatin: a protein secreted by visceral fat that mimics the effects of insulin. Science. 2005;307(5708):426–30.
Revollo JR, Korner A, Mills KF, et al. Nampt/PBEF/Visfatin regulates insulin secretion in beta cells as a systemic NAD biosynthetic enzyme. Cell Metab. 2007;6(5):363–75.
Kiess W, Petzold S, Topfer M, et al. Adipocytes and adipose tissue. Best Pract Res Clin Endocrinol Metab. 2008;22(1):135–53.
Berndt J, Kloting N, Kralisch S, Kovacs P, Fasshauer M, Schon MR, et al. Plasma visfatin concentrations and fat depot-specific mRNA expression in humans. Diabetes. 2005;54(10):2911–6.
Retnakaran R, Youn BS, Liu Y, Hanley AJG, Lee NS, Park JW, et al. Correlation of circulating full-length visfatin (PBEF/NAMPT) with metabolic parameters in subjects with and without diabetes: a cross-sectional study. Clin Endocrinol. 2008;69(6):885–93.
Friebe D, Neef M, Kratzsch J, Erbs S, Dittrich K, Garten A, et al. Leucocytes are a major source of circulating nicotinamide phosphoribosyltransferase (NAMPT)/pre-B cell colony (PBEF)/visfatin linking obesity and inflammation in humans. Diabetologia. 2011;54:1200–11.
Yang RZ, Lee MJ, Hu H, Pray J, Wu HB, Hansen BC, et al. Identification of omentin as a novel depot-specific adipokine in human adipose tissue: possible role in modulating insulin action. Am J Physiol Endocrinol Metab. 2006;290(6):E1253–61.
de Souza Batista CM, Yang RZ, Lee MJ, Glynn NM, Yu DZ, Pray J, et al. Omentin plasma levels and gene expression are decreased in obesity. Diabetes. 2007;56(6):1655–61.
Yamawaki H, Tsubaki N, Mukohda M, Okada M, Hara Y. Omentin, a novel adipokine, induces vasodilation in rat isolated blood vessels. Biochem Biophys Res Commun. 2010;393(4):668–72.
Herder C, Ouwens DM, Carstensen M, Kowall B, Huth C, Meisinger C, et al. Adiponectin may mediate the association between omentin, circulating lipids and insulin sensitivity: results from the KORA F4 study. Eur J Endocrinol. 2015;172(4):423–32.
Oswiecimska J, Suwala A, Swietochowska E, et al. Serum omentin levels in adolescent girls with anorexia nervosa and obesity. Physiol Res. 2015;64(5):701–9.
Buyukinan M, Atar M, Can U, Pirgon O, Guzelant A, Deniz I. The association between serum vaspin and omentin-1 levels in obese children with metabolic syndrome. Metab Syndr Relat Disord. 2018;16(2):76–81.
Zhang M, Tan X, Yin C, Wang L, Tie Y, Xiao Y. Serum levels of omentin-1 are increased after weight loss and are particularly associated with increases in obese children with metabolic syndrome. Acta Paediatr. 2017;106(11):1851–6.
Chavan SS, Hudson LK, Li JH, Ochani M, Harris Y, Patel NB, et al. Identification of pigment epithelium-derived factor as an adipocyte-derived inflammatory factor. Mol Med. 2012;18:1161–8.
Galhardo J, Hunt LP, Shield JP. Serum levels of pigment epithelium-derived factor (PEDF) are positively associated with acanthosis nigricans in obese adolescents. Diabet Med. 2012;29(7):e117–20.
Crowe S, Wu LE, Economou C, Turpin SM, Matzaris M, Hoehn KL, et al. Pigment epithelium-derived factor contributes to insulin resistance in obesity. Cell Metab. 2009;10(1):40–7.
Gattu AK, Birkenfeld AL, Jornayvaz F, et al. Insulin resistance is associated with elevated serum pigment epithelium-derived factor (PEDF) levels in morbidly obese patients. Acta Diabetol. 2012;49(Suppl 1):S161–9.
Sunderland KL, Tryggestad JB, Wang JJ, Teague AM, Pratt LV, Zhang SX, et al. Pigment epithelium-derived factor (PEDF) varies with body composition and insulin resistance in healthy young people. J Clin Endocrinol Metab. 2012;97(11):E2114–8.
Tryggestad JB, Wang JJ, Zhang SX, Thompson DM, Short KR. Elevated plasma pigment epithelium-derived factor in children with type 2 diabetes mellitus is attributable to obesity. Pediatr Diabetes. 2015;16(8):600–5.
Goralski KB, McCarthy TC, Hanniman EA, et al. Chemerin, a novel adipokine that regulates adipogenesis and adipocyte metabolism. J Biol Chem. 2007;282(38):28175–88.
Bozaoglu K, Bolton K, McMillan J, Zimmet P, Jowett J, Collier G, et al. Chemerin is a novel adipokine associated with obesity and metabolic syndrome. Endocrinology. 2007;148(10):4687–94.
Kralisch S, Weise S, Sommer G, Lipfert J, Lossner U, Bluher M, et al. Interleukin-1beta induces the novel adipokine chemerin in adipocytes in vitro. Regul Pept. 2009;154(1–3):102–6.
Takahashi M, Takahashi Y, Takahashi K, Zolotaryov FN, Hong KS, Kitazawa R, et al. Chemerin enhances insulin signaling and potentiates insulin-stimulated glucose uptake in 3T3-L1 adipocytes. FEBS Lett. 2008;582(5):573–8.
Hatziagelaki E, Herder C, Tsiavou A, Teichert T, Chounta A, Nowotny P, et al. Serum chemerin concentrations associate with beta-cell function, but not with insulin resistance in individuals with non-alcoholic fatty liver disease (NAFLD). PLoS One. 2015;10(5):e0124935.
Catalan V, Gomez-Ambrosi J, Rodriguez A, et al. Increased levels of chemerin and its receptor, chemokine-like receptor-1, in obesity are related to inflammation: tumor necrosis factor-alpha stimulates mRNA levels of chemerin in visceral adipocytes from obese patients. Surg Obes Relat Dis. 2013;9(2):306–14.
Ernst MC, Issa M, Goralski KB, Sinal CJ. Chemerin exacerbates glucose intolerance in mouse models of obesity and diabetes. Endocrinology. 2010;151(5):1998–2007.
Bozaoglu K, Segal D, Shields KA, Cummings N, Curran JE, Comuzzie AG, et al. Chemerin is associated with metabolic syndrome phenotypes in a Mexican-American population. J Clin Endocrinol Metab. 2009;94(8):3085–8.
Ye Z, Wang S, Yang Z, He M, Zhang S, Zhang W, et al. Serum lipocalin-2, cathepsin S and chemerin levels and nonalcoholic fatty liver disease. Mol Biol Rep. 2014;41(3):1317–23.
Weigert J, Neumeier M, Wanninger J, Filarsky M, Bauer S, Wiest R, et al. Systemic chemerin is related to inflammation rather than obesity in type 2 diabetes. Clin Endocrinol. 2010;72(3):342–8.
Landgraf K, Friebe D, Ullrich T, Kratzsch J, Dittrich K, Herberth G, et al. Chemerin as a mediator between obesity and vascular inflammation in children. J Clin Endocrinol Metab. 2012;97(4):E556–64.
Redondo MJ, Rodriguez LM, Haymond MW, Hampe CS, Smith EO, Balasubramanyam A, et al. Serum adiposity-induced biomarkers in obese and lean children with recently diagnosed autoimmune type 1 diabetes. Pediatr Diabetes. 2014;15(8):543–9.
Sledzinska M, Szlagatys-Sidorkiewicz A, Brzezinski M, Kazmierska K, Sledzinski T, Kaminska B. Serum chemerin in children with excess body weight may be associated with ongoing metabolic complications - a pilot study. Adv Med Sci. 2017;62(2):383–6.
Maghsoudi Z, Kelishadi R, Hosseinzadeh-Attar MJ. The comparison of chemerin, adiponectin and lipid profile indices in obese and non-obese adolescents. Diabetes Metab Syndr. 2016;10(2 Suppl 1):S43–6.
Niklowitz P, Rothermel J, Lass N, Barth A, Reinehr T. Link between chemerin, central obesity, and parameters of the metabolic syndrome: findings from a longitudinal study in obese children participating in a lifestyle intervention. Int J Obes. 2018.
Dimova R, Tankova T. The role of vaspin in the development of metabolic and glucose tolerance disorders and atherosclerosis. Biomed Res Int. 2015;2015:823481.
Chang HM, Lee HJ, Park HS, Kang JH, Kim KS, Song YS, et al. Effects of weight reduction on serum vaspin concentrations in obese subjects: modification by insulin resistance. Obesity (Silver Spring). 2010;18(11):2105–10.
Handisurya A, Riedl M, Vila G, Maier C, Clodi M, Prikoszovich T, et al. Serum vaspin concentrations in relation to insulin sensitivity following RYGB-induced weight loss. Obes Surg. 2010;20(2):198–203.
Korner A, Neef M, Friebe D, et al. Vaspin is related to gender, puberty and deteriorating insulin sensitivity in children. Int J Obes. 2011;35(4):578–86.
Youn BS, Kloting N, Kratzsch J, Lee N, Park JW, Song ES, et al. Serum vaspin concentrations in human obesity and type 2 diabetes. Diabetes. 2008;57(2):372–7.
Wei Z, Peterson JM, Lei X, Cebotaru L, Wolfgang MJ, Baldeviano GC, et al. C1q/TNF-related protein-12 (CTRP12), a novel adipokine that improves insulin sensitivity and glycemic control in mouse models of obesity and diabetes. J Biol Chem. 2012;287(13):10301–15.
Stefan N, Haring HU. The role of hepatokines in metabolism. Nat Rev Endocrinol. 2013;9(3):144–52.
Fisher FM, Chui PC, Antonellis PJ, et al. Obesity is a fibroblast growth factor 21 (FGF21)-resistant state. Diabetes. 2010;59(11):2781–9.
Kharitonenkov A, Shiyanova TL, Koester A, Ford AM, Micanovic R, Galbreath EJ, et al. FGF-21 as a novel metabolic regulator. J Clin Invest. 2005;115(6):1627–35.
Zhang X, Yeung DC, Karpisek M, et al. Serum FGF21 levels are increased in obesity and are independently associated with the metabolic syndrome in humans. Diabetes. 2008;57(5):1246–53.
So WY, Cheng Q, Chen L, Evans-Molina C, Xu A, Lam KSL, et al. High glucose represses beta-klotho expression and impairs fibroblast growth factor 21 action in mouse pancreatic islets: involvement of peroxisome proliferator-activated receptor gamma signaling. Diabetes. 2013;62(11):3751–9.
Song A, Xu M, Bi Y, Xu Y, Huang Y, Li M, et al. Serum fetuin-A associates with type 2 diabetes and insulin resistance in Chinese adults. PLoS One. 2011;6(4):e19228.
Iyidir OT, Degertekin CK, Yilmaz BA, Altinova AE, Toruner FB, Bozkurt N, et al. Serum levels of fetuin A are increased in women with gestational diabetes mellitus. Arch Gynecol Obstet. 2014;291(4):933–7.
Hennige AM, Staiger H, Wicke C, Machicao F, Fritsche A, Häring HU, et al. Fetuin-A induces cytokine expression and suppresses adiponectin production. PLoS One. 2008;3(3):e1765.
Diaz-Delfin J, Hondares E, Iglesias R, Giralt M, Caelles C, Villarroya F. TNF-alpha represses beta-Klotho expression and impairs FGF21 action in adipose cells: involvement of JNK1 in the FGF21 pathway. Endocrinology. 2012;153(9):4238–45.
Ghorpade DS, Ozcan L, Zheng Z, Nicoloro SM, Shen Y, Chen E, et al. Hepatocyte-secreted DPP4 in obesity promotes adipose inflammation and insulin resistance. Nature. 2018;555(7698):673–7.
Beenken A, Mohammadi M. The FGF family: biology, pathophysiology and therapy. Nat Rev Drug Discov. 2009;8(3):235–53.
Kharitonenkov A, Wroblewski VJ, Koester A, Chen YF, Clutinger CK, Tigno XT, et al. The metabolic state of diabetic monkeys is regulated by fibroblast growth factor-21. Endocrinology. 2007;148(2):774–81.
Chen C, Cheung BM, Tso AW, et al. High plasma level of fibroblast growth factor 21 is an independent predictor of type 2 diabetes: a 5.4-year population-based prospective study in Chinese subjects. Diabetes Care. 2011;34(9):2113–5.
Xiao Y, Xu A, Law LS, et al. Distinct changes in serum fibroblast growth factor 21 levels in different subtypes of diabetes. J Clin Endocrinol Metab. 2012;97(1):E54–8.
Kharitonenkov A, Dunbar JD, Bina HA, Bright S, Moyers JS, Zhang C, et al. FGF-21/FGF-21 receptor interaction and activation is determined by betaKlotho. J Cell Physiol. 2008;215(1):1–7.
Reinehr T, Karges B, Meissner T, Wiegand S, Fritsch M, Holl RW, et al. Fibroblast growth factor 21 and fetuin-A in obese adolescents with and without type 2 diabetes. J Clin Endocrinol Metab. 2015;100(8):3004 jc20152192.
Stefan N, Fritsche A, Weikert C, Boeing H, Joost HG, Haring HU, et al. Plasma fetuin-A levels and the risk of type 2 diabetes. Diabetes. 2008;57(10):2762–7.
Kalabay L, Chavin K, Lebreton JP, Robinson KA, Buse MG, Arnaud P. Human recombinant alpha 2-HS glycoprotein is produced in insect cells as a full length inhibitor of the insulin receptor tyrosine kinase. Horm Metab Res. 1998;30:1):1–6.
Mathews ST, Singh GP, Ranalletta M, Cintron VJ, Qiang X, Goustin AS, et al. Improved insulin sensitivity and resistance to weight gain in mice null for the Ahsg gene. Diabetes. 2002;51(8):2450–8.
Ix JH, Biggs ML, Mukamal KJ, Kizer JR, Zieman SJ, Siscovick DS, et al. Association of fetuin-a with incident diabetes mellitus in community-living older adults: the cardiovascular health study. Circulation. 2012;125(19):2316–22.
Reinehr T, Roth CL. Fetuin-A and its relation to metabolic syndrome and fatty liver disease in obese children before and after weight loss. J Clin Endocrinol Metab. 2008;93(11):4479–85.
Lee NK, Sowa H, Hinoi E, Ferron M, Ahn JD, Confavreux C, et al. Endocrine regulation of energy metabolism by the skeleton. Cell. 2007;130(3):456–69.
Bouillon R, Bex M, Van HE, et al. Influence of age, sex, and insulin on osteoblast function: osteoblast dysfunction in diabetes mellitus. J Clin Endocrinol Metab. 1995;80(4):1194–202.
Pietschmann P, Schernthaner G, Woloszczuk W. Serum osteocalcin levels in diabetes mellitus: analysis of the type of diabetes and microvascular complications. Diabetologia. 1988;31(12):892–5.
Reinehr T, Roth CL. A new link between skeleton, obesity and insulin resistance: relationships between osteocalcin, leptin and insulin resistance in obese children before and after weight loss. Int J Obes. 2010;34(5):852–8.
Park KH, Zaichenko L, Brinkoetter M, et al. Circulating irisin in relation to insulin resistance and the metabolic syndrome. J Clin Endocrinol Metab. 2013;98(12):4899–907.
Sesti G, Andreozzi F, Fiorentino TV, Mannino GC, Sciacqua A, Marini MA, et al. High circulating irisin levels are associated with insulin resistance and vascular atherosclerosis in a cohort of nondiabetic adult subjects. Acta Diabetol. 2014;51(5):705–13.
Staiger H, Bohm A, Scheler M, et al. Common genetic variation in the human FNDC5 locus, encoding the novel muscle-derived ‘browning’ factor irisin, determines insulin sensitivity. PLoS One. 2013;8(4):e61903.
Bostrom P, Wu J, Jedrychowski MP, et al. A PGC1-alpha-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature. 2012;481(7382):463–8.
Anastasilakis AD, Polyzos SA, Saridakis ZG, Kynigopoulos G, Skouvaklidou EC, Molyvas D, et al. Circulating irisin in healthy, young individuals: day-night rhythm, effects of food intake and exercise, and associations with gender, physical activity, diet, and body composition. J Clin Endocrinol Metab. 2014;99(9):3247–55.
Moreno-Navarrete JM, Ortega F, Serrano M, Guerra E, Pardo G, Tinahones F, et al. Irisin is expressed and produced by human muscle and adipose tissue in association with obesity and insulin resistance. J Clin Endocrinol Metab. 2013;98(4):E769–78.
Huh JY, Mougios V, Kabasakalis A, Fatouros I, Siopi A, Douroudos II, et al. Exercise-induced irisin secretion is independent of age or fitness level and increased irisin may directly modulate muscle metabolism through AMPK activation. J Clin Endocrinol Metab. 2014;99(11):E2154–61.
Stengel A, Hofmann T, Goebel-Stengel M, Elbelt U, Kobelt P, Klapp BF. Circulating levels of irisin in patients with anorexia nervosa and different stages of obesity--correlation with body mass index. Peptides. 2013;39:125–30.
Reinehr T, Elfers C, Lass N, Roth CL. Irisin and its relation to insulin resistance and puberty in obese children: a longitudinal analysis. J Clin Endocrinol Metab. 2015;100(5):2123–30.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Thomas Reinehr and Christian Ludwig Roth declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Pediatric Type 2 and Monogenic Diabetes
Rights and permissions
About this article
Cite this article
Reinehr, T., Roth, C.L. Inflammation Markers in Type 2 Diabetes and the Metabolic Syndrome in the Pediatric Population. Curr Diab Rep 18, 131 (2018). https://doi.org/10.1007/s11892-018-1110-5
Published:
DOI: https://doi.org/10.1007/s11892-018-1110-5