Abstract
Purpose of Review
This review describes recent data supporting locoregional ablative radiation in the treatment of oligometastatic colorectal cancer liver metastases.
Recent Findings
Stereotactic body radiotherapy (SBRT) demonstrates high rates of local control in colorectal cancer liver metastases when a biologically equivalent dose of > 100 Gy is delivered. Future innovations to improve the efficacy of SBRT include MRI-guided radiotherapy (MRgRT) to enhance target accuracy, systemic immune activation to treat extrahepatic disease, and genomic customization. Selective internal radiotherapy (SIRT) with y-90 is an intra-arterial therapy that delivers high doses to liver metastases internally which has shown to increase liver disease control in phase 3 trials. Advancements in transarterial radioembolization (TARE) dosimetry could improve local control and decrease toxicity.
Summary
SBRT and SIRT are both promising options in treating unresectable metastatic colorectal cancer liver metastases. Identification of oligometastatic patients who receive long-term disease control from either therapy is essential. Future advancements focusing on improving radiation design and customization could further improve efficacy and toxicity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
An estimated 150,000 people are diagnosed with colorectal cancer (CRC) each year in the USA [1]. Approximately 25–30% of CRC patients present with either synchronous or metachronous CRC liver metastases (CLMs) [2, 3]. For those with oligometastatic disease, usually defined as fewer than 5 sites of disease, the treatment paradigm includes definitive local treatment of all disease sites as clinical studies have demonstrated that metastatic control increases progression-free survival (PFS) and overall survival (OS) in various disease sites [4]. If feasible, surgical resection of CLMs can produce clinical outcomes comparable to earlier stages [5, 6].
However, up to 90% of newly diagnosed liver metastases are inoperable [7]. While multiagent chemotherapy can also convert some patients into surgical candidates, a significant number of CLMs will remain inoperable, emphasizing the need for nonsurgical focal therapy options [8, 9].
Radiation therapy is a non-invasive modality that can provide extended local control (LC) when delivered at high doses [10]. Internal beam radiation and external beam radiation (EBRT) are commonly used to achieve disease control within the liver. Within EBRT, SBRT treats liver lesions with millimeter precision. Meanwhile, selective internal radiotherapy (SIRT) delivers high doses of radioactive yttrium-90 (y-90) to CLMs utilizing TARE.
The main purpose of this review is to detail the most current data examining both forms of radiotherapy towards treating oligometastatic CLMs. We also discuss future advancements including improving radiation precision towards minimizing toxicity, activating systemic immunity for extrahepatic disease control, genomic tailoring of radiation delivery, and the potential to combine SBRT and SIRT.
Introduction to SBRT
SBRT refers to submillimeter delivery of ablative radiation to tumors outside of the central nervous system in one to five treatments. It involves integration of high-resolution imaging, non-invasive positioning techniques, and respiratory motion management to design conformal beams targeted around a tumor with an additional goal to reduce the amount of radiation to adjacent normal tissue. Currently, it is an essential part of the treatment paradigm for lung, pancreas, and prostate cancers, as well as the palliation of metastatic lesions from a broad array of primary malignancies.
SBRT for CLMs
Investigating the optimal SBRT dose to liver metastases, retrospective studies looking at hepatic metastases of various etiologies have noted the importance of achieving a biological effective dose (BED) of at least 100 Gy to achieve durable LC [11, 12]. Published reports using SBRT to treat liver metastases have shown actuarial LC rates ranging from 50 to 100% with higher doses associated with better LC. A multi-institutional phase I/II study of SBRT for liver metastases showed the safety of dose escalation from 36 Gy up to 60 Gy in 3 fractions with 2-year actuarial in-field LC rates of 92% [13]. Lesions smaller than 3 cm had a 100% LC at 2 years.
The few retrospective studies that have focused solely on CLMs treated with SBRT have found a possible benefit of BED greater than 110 Gy [13,14,15,16,17,18,19,20]. A multi-institutional pooled analysis of 102 CLMs in 65 patients with 1 to 4 hepatic sites appreciated significant differences in LC when utilizing a BED cutoff value of 75 Gy [21]. On tumor control probability modeling, they estimated a BED of 117 Gy was needed to achieve 90% LC at 1 year. Joo et al. stratified lesions by BED (< 80 Gy, 100–112 Gy, > 132 Gy) and noted an improvement in the 2-year LC from 83 to 89% with BED > 130 Gy compared to 100–112 Gy, a small but not statistically significant difference [22•]. BED greater than 132 was predictive of OS and LC on univariate analysis. However, no differences in PFS were noted between the three groups.
A large prospective Dutch-Belgian registry followed 515 patients with 668 liver metastases treated using SBRT with a variety of fractionations ranging from 54 to 60 Gy in 3, 5, 8, and 12 fractions [23]. Eighty percent of patients presented with CLMs and approximately 50% completed prior therapy. Most patients completed SBRT to 1 liver lesion. Overall LC after RT was 87% at 1 year, 75% at 2 years, and 68% at 3 years. No differences in LC were appreciated by SBRT fractionation scheme. OS at 1, 2, and 3 years was 84%, 63%, and 44%. The rate of grade 3 or higher CTCAE gastrointestinal toxicity was 3.9% including one event of grade 5 hepatobiliary toxicity. Analysis of treated lesions by liver segment did not reveal differences in toxicity outcomes but an association between segment 3 lesions and lower LC approached significance (HR 3.72, 0.94–14,63, P = 0.06). Only 14 segments and 3 livers were treated making these results prone to sample bias. Recent retrospective studies note similar findings with 3-year LC rates ranging from 68 to 79% (Table 1).
Few prospective trials have investigated SBRT solely in the setting of oligometastatic CLMs (Table 2). Rusthoven et al. at the University of Colorado first demonstrated 2-year LC rates greater than 90% with SBRT to 1 to 3 liver metastases after 60 Gy in 3 fractions but only included 32% CRC patients [13]. Shortly thereafter, Erasmus University published their prospective experience of SBRT for CLMs looking at patients with limited hepatic involvement unamenable to radiofrequency ablation and treated with 37.5 Gy in 3 fractions [20]. Nine local failures were seen in the 31 CLMs treated, which is higher compared to prior large retrospective studies but may be explained by the lower dose given (BED = 84.4 Gy). A phase 2 Italian trial conducted by Scorsetti et al. used 75 Gy in 3 fractions (BED = 262.5 Gy) to treat one to three CLMs [27••]. Of the 52 lesions treated, 43% demonstrated a complete response and 3-year LC was 85%. Of the 26% of patients presenting with extrahepatic disease, the median PFS was 12 months and OS was 29 months.
Predicting which patients with oligometastatic CLMs achieve durable disease control after SBRT remains unclear. Utilizing clinical risk score (CRS) based on the number of metastases, size of the lesion(s), serum CEA level, disease-free interval, and presence of lymph nodes at diagnosis, Creasy et al. appreciated significantly longer disease-free interval after resection in those with lower CRS [31]. Future SBRT studies implementing a multifactorial stratification tool like CRS could help identify those that would significantly benefit from ablative radiation.
The importance of patient selection is further demonstrated in a recently published phase I trial from UT Southwestern looking at single-fraction SBRT of 35 to 40 Gy [28••]. Eligible patients had good performance status with less than 5 liver metastases that were at least 2 cm away from the portal vein and its bifurcation within the liver due to concern for radiation toxicity. At median follow-up of 28.3 months, only two local failures were noted. Despite 42.5% patients having extrahepatic disease, 4-year OS was 49.7%, and only three grade 2 gastrointestinal toxicities were noted. None of the patients developed radiation-induced liver disease. The high LC is likely explained by the high BED delivered (> 150 Gy) although BED estimates for single-fraction treatment can be inaccurate. The total disease burden was also limited with 87.9% of patients having fewer than 5 sites of disease and 66.6% having 2 or fewer. All patients in this study only received treatment to 1 to 2 liver metastases with a median maximal tumor length of 2 cm (0.5–5 cm). Additionally, only allowing tumors further from major vessels may select for patients with a lower risk of further disease spread. A limitation of this study is low incidence of inoperable patients with limited hepatic involvement reflected by 9-year accrual of only 33 patients. Nonetheless, this trial encourages further investigation into SBRT providing long-term cure for those with oligometastatic CLMs.
In summary, retrospective evidence shows that SBRT provides durable LC of CLMs supported by increased LC with higher BED delivered (> 110–130 Gy). The low rates of PFS may be explained by higher overall disease burden and extrahepatic disease. Future studies may benefit from scoring systems to estimate overall disease burden to select patients most likely to benefit from SBRT.
Future Directions of SBRT for CLMs
Proton-Based SBRT
The use of protons with SBRT is potentially attractive given the advantage of lack of exit dose when compared to photon-based techniques. Several treatment planning comparison studies have been reported showing improved normal tissue dosimetry of proton-based radiotherapy compared to photon-based radiotherapy in the treatment of liver, lung, and adrenal lesions [30••, 32, 33]. SBRT with protons have been used to treat hepatocellular carcinoma (HCC) using various hypofractionated regimens with minimal acute toxicity, no reported radiation-induced liver disease, and 3-year PFS of 60% [34,35,36,37]. Phase I data of ablative proton SBRT (BED 180 Gy) shows it to be well tolerated in patients with limited liver metastases with no acute toxicities grade 2 or higher [38].
Combination SBRT and Immunotherapy for Extrahepatic Control
Despite high LC rates with SBRT, median OS remains less than 3 years. This is attributed to the high rates of out-of-field disease progression within the first 2 years emphasizing the importance of systemic control [39]. Examining changes in PD-L1 expression and CD8 + T-cell infiltration before and after chemoradiation in rectal cancer patients, Lim et al. found that persistently high tumor surface PD-L1 and low CD8 + T-cells after treatment correlated with an increased risk of distant disease relapse, suggesting anti-PD-1 therapy could improve outcomes in rectal cancer [40]. SBRT has been shown to promote tumor antigen release and increase T-cell receptor diversity [41] and the combination of checkpoint inhibitors and SBRT has been extensively investigated in preclinical and early phase trials [42] (Fig. 1A). However, the dose and fractionation of radiation to prime the immune system against colorectal cancer remain unknown. A recent multi-institutional phase 2 trial examined 8 Gy per day for 3 days versus 2 Gy twice a day for 2 days for those with 1 to 2 microsatellite stable CLMs given in combination with anti-CTLA-4 and anti-PD-1 therapy [43•]. Despite noting significant changes in immune cell populations within treated tumor specimens, tumor regression outside of the radiation field did not occur in either arm. Further trials examining checkpoint blockade with SBRT for CLMs are underway and will provide more information (NCT02837263, NCT03927898).
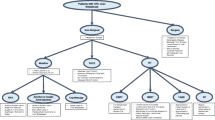
A Radiation promotes release of diverse tumor antigens which are taken by APC in regional lymphatics resulting in greater CD8 + T-cell diversity in conjunction with immunotherapy to act on distant tumor via the abscopal effect. B Genomic sequencing of primary colorectal cancer reveals potential radioresistance which can be overcome with higher doses of radiation or radiosensitizers. C With higher soft tissue resolution, MR-guided radiotherapy results in higher precision for tumor targeting and lower doses to nearby normal organs including the stomach and small bowel. CT-based treatment required fiducials to generate Gross Tumor Volume (GTV) (red), then Internal Target Volume (ITV) (yellow) for motion management, and then Planning Target Volume (PTV) (green). The MRI-based treatment directly visualizes the tumor GTV without the aid of fiducials, is treated via Deep Inspiratory Breath Hold (DIBH) for respiratory motion management, and then is expanded to form a smaller PTV in comparison to CT. D Fusion of pretreatment Tm-99 imaging with diagnostic CT could reveal areas for dosimetric improvement to reduce toxicity and increase efficacy
Integrating Genomics to Improve SBRT for CLM
Previous retrospective studies have noted a BED > 110 Gy needed to achieve durable LC, suggesting that CLMs may be more radioresistant than other primary tumor types. Ahmed et al. developed a radiation sensitivity index (RSI), with higher scores indicating more radioresistance, based on RNA expression of 10 genes correlated with radiation response [44]. They evaluated RSI values for 1362 primary and 704 metastatic colon cancer lesions. They found a higher RSI for CLMs compared to CRC lung metastases, which translated to inferior rates of LC after SBRT. RSI scoring was used to classify 372 resected liver metastases (84.4% CRC) and CRC displayed a higher RSI (0.43) than breast (0.35), pancreas (0.27), lung adenocarcinoma (0.31), and anal cancer (0.22). CLMs demonstrated worse LC than non-colorectal metastases at 2 years after SBRT with BED of 100 Gy or higher (59% vs 100%, p = 0.019).
A phase 2 proton SBRT trial studied the effect of various genomic alterations after SBRT (30 to 50 Gy in 5 fractions) in 91 patients with limited liver metastases of which 34 were CLMs [30••]. Like Ahmed et al., they noted higher rates of local failure in CLMs. Genetic predictors of local failure after SBRT included KRAS mutations and hotpot TP53 mutations. Patients with both mutations had only 20% 1-year LC.
These studies raise the question of whether radiation dose should be customized based on genomic alterations of CLMs (Fig. 1B). They also open further exploration to improve outcomes after SBRT of CLMs including combination locoregional therapy to overcome radioresistance (NCT03963726) and the addition of radiosensitizers to augment SBRT (NCT01569984, NCT03223779).
MRI-Guided Radiotherapy for CLMs
Most EBRT machines utilize x-ray imaging during SBRT to guide treatment. Due to poor soft tissue delineation of liver targets on x-rays, fiducials are often placed within metastatic liver lesions as surrogate markers for targeting and assessing motion [45]. However, fiducial placement carries risk of pain, pneumothorax, bleeding, and fiducial migration [46].
The integration of magnetic resonance imaging (MRI) into radiation treatment machines allows for real-time MRI acquisition during SBRT, resulting in improved soft tissue visualization, commonly referred to as MRgRT [47]. Cine-MRI captures images every 4 to 8 s to allow for real-time treatment monitoring of tumor motion without the need for fiducials [48]. Stereotactic MR-guided online-adaptive radiotherapy uses daily MRI to modify dose and treatment plan in response to changes in tumor response, target motion, and organs at risk motion.
Studies exploring high-dose focal RT using MRgRT for CLMs are in the early stages. In a multi-institutional retrospective review of 26 patients with both primary and secondary liver malignancies treated to 50 Gy in 10 fractions, Rosenberg et al. showed freedom from local progression of CLMs at 75%. Only 2 patients experienced grade 3 toxicities and none experienced grade 4 or higher toxicity [49]. van Dams et al. recently published their phase I results exploring the safety and feasibility of MR-guided SBRT in 20 patients treated with 25 liver tumors (8 primary, 12 secondary) [50•]. This trial examined a 3-dose tier for single and multiple lesion plans while prioritizing limiting the dose to normal liver. They appreciated a LC benefit when liver tumors were treated with a BED > 100 Gy consistent with previous literature [11, 12, 51]. No acute grade 3 toxicities were noted and only 1 patient had late grade 3 duodenal toxicity. With the ability to deliver ablative doses of radiotherapy with higher precision, MRgRT is a promising tool with the potential to overcome CLM radioresistance while minimizing toxicity due to real-time target and organs at risk visualization (Fig. 1C). Prospective trials comparing CT-based SBRT and MRgRT outcomes and toxicity are eagerly awaited.
Selective Internal Radiotherapy for CLMs
Transarterial Radioembolization
Transarterial radioembolization (TARE) with y-90 has been shown to be an effective treatment for CRC metastases to the liver. The beta radiation’s mean tissue penetration of 2.5 mm allows for high doses to be administered with doses > 100 Gy frequently delivered.
The ideal dose to allow for disease control without significant toxicity to the normal liver or lung is still unclear. In the setting of HCC, several studies have shown that a higher absorbed tumor dose is a superior prognostic marker of survival with one study even suggesting 205 Gy as an appropriate target dose [52, 53]. Dosage data in CRC is less robust, but there are also studies that suggest improved benefit with higher doses. Alsultan et al. looked at dose–response in patients with CLMs treated with y-90. They reported median absorbed dose by tumor response of 196 Gy for complete response, 177 Gy for partial response, 72 Gy for stable disease, and 95 Gy for progressive disease. Based on their data, they concluded that a tumor absorbed dose of > 189 Gy was recommended as a target to achieve the best response [54]. Further study is needed in this arena, particularly in the setting of CRC metastases.
Resin vs Glass TARE
Two devices are available for y-90 delivery, glass sphere-based y-90 (TheraSphere™, Boston Scientific Corporation, Marlborough, MA) and resin sphere-based y-90 (SIR-Spheres®, Sirtex Medical, Sydney, New South Wales, Australia). The main difference between the two devices lies in the activity per sphere with glass spheres having an activity on the order of 50 times that of resin spheres. This translates into a significantly larger amount of resin beads required to deliver the same amount of radioactivity which, in turn, can affect the biodistribution of the spheres and confers a potentially higher embolic effect to resin spheres, potentially precluding delivery of the entire prescribed dose if stasis is met first. The dosimetry of the two devices has historically been calculated differently, as well. The details are beyond the scope of this review, but briefly, resin-based therapy dosimetry has largely been based on a body surface area (BSA) calculation and glass-based therapy dosimetry on a medical internal radiation dose (MIRD) calculation.
SIRT for CLMs
While resin-based therapy is specifically FDA approved for CRC metastases, and glass-based therapy is only FDA approved for HCC, several studies have shown the safety and efficacy of both of these devices in the chemorefractory or salvage setting of CRC. A single-center study of 302 patients treated with resin-based y-90 radioembolization demonstrated a median survival of 10.5 months after radioembolization performed in the salvage setting with relatively limited clinical toxicity. Hepatic tumor involvement was found to be a statistically significant factor in OS with 0–25%, 26–50%, and 51–75% having 11.6, 9.1, and 5.6 months of median survival [55]. Another study looking at 531 patients at multiple centers who were treated with glass-based y-90 radioembolization similarly showed a median OS of 10.6 months with low toxicity. This study also showed better survival outcomes for hepatic tumor burden no more than 25% on univariate analysis [56]. These studies show the benefit of y-90 in this setting and suggest improved outcomes with better patient selection, such as those with lesser disease burden.
The favorable results of these studies and earlier smaller studies prompted investigation into the use of TARE in the first-line setting in an attempt to improve patient outcomes. Three multicenter, randomized, worldwide phase 3 trials (FOXFIRE, SIRFLOX, and FOXFIRE-Global) with 1103 total patients were performed, with a combined analysis, comparing y-90 radioembolization plus chemotherapy to chemotherapy alone in treatment-naïve patients. While the study did show better liver control in the radioembolization group with a lower cumulative incidence of the first progression in the liver (31% vs. 49%) and higher objective response (72% vs. 63%), there was no statistically significant difference in the primary endpoint of OS (22.6 months vs. 23.3 months) [57••].
However, post hoc analysis of some of this data yielded interesting results for potential use of TARE in the first-line setting. The data from the SIRFLOX and FOXFIRE-Global trials was looked at in the setting of patients with right-sided metastatic CRC, based on right-sided primary being a poor prognostic factor. Looking at these 2 studies alone, there was again no statistically significant difference in OS when looking at all 739 patients (24.3 months vs. 24.6 months), but in the 179 patients with right-sided primary, an improved OS was seen in those receiving y-90 radioembolization in addition to chemotherapy (22.0 months vs. 17.1 months) [58]. Another post hoc analysis of 472 patients from SIRFLOX looked at secondary technical resectability of the two arms. While there was no difference in resectability at baseline (11.9% vs. 11.0%), both arms demonstrated an increase in resectability compared to baseline, with more patients resectable in the radioembolization group compared to the control (38.1% vs. 28.9%) [59]. While these findings are somewhat limited due to the post hoc nature of these studies, their findings suggest that TARE may still have a role in the first-line setting in select patients. Table 3 summarizes recent studies examining SIRT for CLMs.
Radiation Segmentectomy for Oligometastatic Disease
Another potential use for y-90 in the treatment of CRC is radiation segmentectomy (RS). RS uses higher, ablative doses of radiation, often > 200 Gy, injected into a smaller territory, to treat tumors primarily when they are in only 1 or 2 segments of the liver. This technique is best suited for patients with limited disease, but who are not candidates for resection or ablation. The higher doses to a smaller area, segmental or subsegmental delivery, result in improved necrosis to the tumor and surrounding liver. This technique has been studied extensively in the treatment of HCC where it has been shown to be safe, efficacious, and even potentially curative [61,62,63]. However, its role in CRC metastases remains unclear given their hypovascularity, frequent multifocality, and potential for micrometastases not visible on imaging. Small studies have shown excellent results in the safety and efficacy of this technique in CRC and other metastases. One study looking at initial efficacy treated 10 patients with this technique with 5 showing complete response, 1 with partial response, and 4 with stable disease with a mean dose of 261 Gy [64]. Another study treating 14 tumors in 10 patients showed 2- and 3-year local tumor PFS of 83% and 69% and OS of 41.5 months with a mean delivered dose of 293 Gy [60]. These results are promising for the use of this technique in treatment of patients with oligometastatic disease.
Improving TARE Dosimetry
Advances in dosimetry may further improve the efficacy and safety of TARE. Much of the early data acquired on TARE is in the setting of previously mentioned dosimetry methods which have significant limitations. More centers are now using the partition model which is more work-intensive, but theoretically more accurate and patient-specific taking into consideration tumor volume, normal liver volume, tumor to normal ratio, and lung shunt. Currently, technetium-99 m macroaggregated albumin (Tc-99 m MAA) is injected into the hepatic artery during a mapping angiogram primarily to evaluate the lung shunt. While previously calculated with planar imaging, this is now performed via SPECT/CT imaging for more accurate calculations. These Tc-99 m MAA SPECT/CT images can also be used to improve dosimetry with more accurate tumor to normal absorbed dose ratios. This is, unfortunately, somewhat limited by the differential distribution patterns of Tc-99 m MAA and y-90 microspheres. There is ongoing research into other radiolabeled particles which may better approximate the y-90 distribution pattern. Technological advancements in y-90 PET/CT imaging have allowed for a more accurate assessment of post-treatment y-90 distribution and, as a result, a more accurate calculation of tumor absorbed dose [65•] (Fig. 1D). This information can be used to determine undertreated areas of tumor which may require additional therapy, such as a more selective TARE, SBRT, or other locoregional therapies. A recent study combining EBRT and SIRT utilizing y-90-based dosimetry in 10 HCC patients appeared safe [66]. Further studies are needed regarding the safety and efficacy of TARE using these more advanced dosimetry methodologies.
Comparison of SBRT and SIRT Outcomes
Currently, there are no clinical trials comparing SBRT and SIRT for limited CLMs. A previous trial examining checkpoint inhibitors in combination with y-90 or SBRT was withdrawn (NCT03802747). While retrospective studies have examined the efficacy of both treatments for primary hepatobiliary malignancies, none exists for oligometastatic liver disease [67, 68]. These retrospective studies contain a low number of patients making it difficult to conclude superiority of either SBRT or SIRT outcomes. The LEGACY study retrospectively examined LC of SIRT in solitary HCC and noted excellent 24-month LC of 100% by modified RECIST criteria although 20% of these patients had received 2 or more courses of y-90 [69]. The median size of tumor treated was 2.6 cm. Prospective SBRT studies treating small tumor sizes (median 2 to 3 cm) demonstrated LC rates of 68 to 96.6% [23, 28••]. Larger population studies such as meta-analyses are needed to assess the effectiveness of SIRT and SBRT depending on various CLM characteristics (i.e., segment location, proximity to vessels, tumor volume, number of lesions genomic profile).
Conclusions
SBRT and SIRT are ablative locoregional therapies that provide durable LC with minimal toxicity, but their role in oligometastatic CLMs requires further investigation. Several promising areas of SBRT research for CLMs are underway including combination with immunotherapy to combat extrahepatic disease, genomic sequencing to reveal customized approaches to overcoming radioresistance, and enhanced precision targeting with MgRT. TARE in combination with chemotherapy has been shown in multicenter phase 3 trials to improve liver disease control compared to chemotherapy alone. Subgroup analysis reveals a possible OS benefit for those with reduced liver involvement. Radiation segmentectomy delivers even higher ablative doses (> 200 Gy) and has been utilized in treatment of HCC. Y-90 TARE dosimetry with PET could reveal opportunities to combine SIRT and SBRT together. Further refinement of those with minimal metastatic burden could result in a prolonged disease-free interval after SBRT and/or SIRT of CLMs.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer Statistics, 2021. CA Cancer J Clin. 2021;71:7–33.
Hackl C, Neumann P, Gerken M, Loss M, Klinkhammer-Schalke M, Schlitt HJ. Treatment of colorectal liver metastases in Germany: a ten-year population-based analysis of 5772 cases of primary colorectal adenocarcinoma. BMC Cancer. 2014;14:810.
Manfredi S, Lepage C, Hatem C, Coatmeur O, Faivre J, Bouvier A-M. Epidemiology and management of liver metastases from colorectal cancer. Ann Surg. 2006;244:254–9.
Palma DA, Olson R, Harrow S, et al. Stereotactic ablative radiotherapy versus standard of care palliative treatment in patients with oligometastatic cancers (SABR-COMET): a randomised, phase 2, open-label trial. Lancet. 2019;393:2051–8.
Aloia TA, Vauthey J-N, Loyer EM, Ribero D, Pawlik TM, Wei SH, Curley SA, Zorzi D, Abdalla EK. Solitary colorectal liver metastasis: resection determines outcome. Arch Surg. 2006;141:460–6 (discussion 466-467).
Morris EJA, Forman D, Thomas JD, Quirke P, Taylor EF, Fairley L, Cottier B, Poston G. Surgical management and outcomes of colorectal cancer liver metastases. Br J Surg. 2010;97:1110–8.
Van Cutsem E, Nordlinger B, Adam R, Köhne C-H, Pozzo C, Poston G, Ychou M, Rougier P, European Colorectal Metastases Treatment Group. Towards a pan-European consensus on the treatment of patients with colorectal liver metastases. Eur J Cancer. 2006;42:2212–21.
Ye L-C, Liu T-S, Ren L, et al. Randomized controlled trial of cetuximab plus chemotherapy for patients with KRAS wild-type unresectable colorectal liver-limited metastases. J Clin Oncol. 2013;31:1931–8.
Folprecht G, Gruenberger T, Bechstein WO, et al. Tumour response and secondary resectability of colorectal liver metastases following neoadjuvant chemotherapy with cetuximab: the CELIM randomised phase 2 trial. Lancet Oncol. 2010;11:38–47.
Chang BK, Timmerman RD. Stereotactic body radiation therapy: a comprehensive review. Am J Clin Oncol. 2007;30:637–44.
Mahadevan A, Blanck O, Lanciano R, Peddada A, Sundararaman S, D’Ambrosio D, Sharma S, Perry D, Kolker J, Davis J. Stereotactic body radiotherapy (SBRT) for liver metastasis – clinical outcomes from the international multi-institutional RSSearch® Patient Registry. Radiat Oncol. 2018;13:1–11.
Kok END, Jansen EPM, Heeres BC, Kok NFM, Janssen T, van Werkhoven E, Sanders FRK, Ruers TJM, Nowee ME, Kuhlmann KFD. High versus low dose stereotactic body radiation therapy for hepatic metastases. Clin Transl Radiat Oncol. 2019;20:45–50.
Rusthoven KE, Kavanagh BD, Cardenes H, et al. Multi-institutional phase I/II trial of stereotactic body radiation therapy for liver metastases. JCO. 2009;27:1572–8.
Lee MT, Kim JJ, Dinniwell R, et al. Phase I study of individualized stereotactic body radiotherapy of liver metastases. J Clin Oncol. 2009;27:1585–91.
Rule W, Timmerman R, Tong L, Abdulrahman R, Meyer J, Boike T, Schwarz RE, Weatherall P, Chinsoo Cho L. Phase I dose-escalation study of stereotactic body radiotherapy in patients with hepatic metastases. Ann Surg Oncol. 2011;18:1081–7.
Goodman KA, Wiegner EA, Maturen KE, Zhang Z, Mo Q, Yang G, Gibbs IC, Fisher GA, Koong AC. Dose-escalation study of single-fraction stereotactic body radiotherapy for liver malignancies. Int J Radiat Oncol Biol Phys. 2010;78:486–93.
Herfarth KK, Debus J, Wannenmacher M. Stereotactic radiation therapy of liver metastases: update of the initial phase-I/II trial. Front Radiat Ther Oncol. 2004;38:100–5.
Wulf J, Guckenberger M, Haedinger U, Oppitz U, Mueller G, Baier K, Flentje M. Stereotactic radiotherapy of primary liver cancer and hepatic metastases. Acta Oncol. 2006;45:838–47.
Hoyer M, Roed H, Traberg Hansen A, Ohlhuis L, Petersen J, Nellemann H, Kiil Berthelsen A, Grau C, Aage Engelholm S, Von der Maase H. Phase II study on stereotactic body radiotherapy of colorectal metastases. Acta Oncol. 2006;45:823–30.
van der Pool AEM, Méndez Romero A, Wunderink W, Heijmen BJ, Levendag PC, Verhoef C, Ijzermans JNM. Stereotactic body radiation therapy for colorectal liver metastases. Br J Surg. 2010;97:377–82.
Chang DT, Swaminath A, Kozak M, et al. Stereotactic body radiotherapy for colorectal liver metastases: a pooled analysis. Cancer. 2011;117:4060–9.
Joo JH, Park J-H, Kim JC, et al. Local control outcomes using stereotactic body radiation therapy for liver metastases from colorectal cancer. Int J Radiat Oncol Biol Phys. 2017;99:876–883. CLM SBRT cohort study that showed LC benefit with BED >130 Gy. This study shows higher ablative radiation doses may lead to improved LC.
Méndez Romero A, Schillemans W, van Os R, et al. The Dutch-Belgian Registry of stereotactic body radiation therapy for liver metastases: clinical outcomes of 515 patients and 668 metastases. Int J Radiat Oncol Biol Phys. 2021;109:1377–86.
Clerici E, Comito T, Franzese C, et al. Role of stereotactic body radiation therapy in the treatment of liver metastases: clinical results and prognostic factors. Strahlenther Onkol. 2020;196:325–33.
Andratschke N, Alheid H, Allgäuer M, et al. The SBRT database initiative of the German Society for Radiation Oncology (DEGRO): patterns of care and outcome analysis of stereotactic body radiotherapy (SBRT) for liver oligometastases in 474 patients with 623 metastases. BMC Cancer. 2018;18:283.
Franzese C, Comito T, Clerici E, et al. Liver metastases from colorectal cancer: propensity score-based comparison of stereotactic body radiation therapy vs. microwave ablation. J Cancer Res Clin Oncol. 2018;144:1777–83.
Scorsetti M, Comito T, Tozzi A, et al. Final results of a phase II trial for stereotactic body radiation therapy for patients with inoperable liver metastases from colorectal cancer. J Cancer Res Clin Oncol. 2015;141:543–553. Phase II study of SBRT to CLM 75 Gy in 3 fractions (BED 262.5) that showed excellent rates of LC-CR 43% and 3 years LC 85%.
Folkert MR, Meyer JJ, Aguilera TA, et al. Long-term results of a phase 1 dose-escalation trial and subsequent institutional experience of single-fraction stereotactic ablative radiation therapy for liver metastases. Int J Radiat Oncol Biol Phys. 2021;109:1387–1395. Phase I dose-escalation study 35 to 40 Gy in 1 fraction (BED >150) SBRT that showed high LC with low toxicity — 4-year LC 96% with no G3+ toxicities. This study demonstrates the feasibility, high LC, and low toxicity with single fraction ablative SBRT.
McPartlin A, Swaminath A, Wang R, et al. Long-term outcomes of phase 1 and 2 studies of SBRT for hepatic colorectal metastases. Int J Radiat Oncol Biol Phys. 2017;99:388–95.
Hong TS, Wo JY, Borger DR, et al. Phase II study of proton-based stereotactic body radiation therapy for liver metastases: importance of tumor genotype. JNCI J Natl Cancer Inst. 2017. https://doi.org/10.1093/jnci/djx031. Phase II proton SBRT study that showed higher LF in CLMs as well as those with KRAS and TP54 mutations. This study shows that CLMs have higher rates of LF and that certain genomic markers may help predict treatment response.
Creasy JM, Sadot E, Koerkamp BG, et al. Actual 10-year survival after hepatic resection of colorectal liver metastases: what factors preclude cure? Surgery. 2018;163:1238–44.
Westover KD, Seco J, Adams JA, Lanuti M, Choi NC, Engelsman M, Willers H. Proton SBRT for medically inoperable stage I NSCLC. J Thorac Oncol. 2012;7:1021–5.
Toesca DAS, Koong AJ, von Eyben R, Koong AC, Chang DT. Stereotactic body radiation therapy for adrenal gland metastases: outcomes and toxicity. Adv Radiat Oncol. 2018;3:621–9.
Bush DA, Kayali Z, Grove R, Slater JD. The safety and efficacy of high-dose proton beam radiotherapy for hepatocellular carcinoma: a phase 2 prospective trial. Cancer. 2011;117:3053–9.
Fukumitsu N, Sugahara S, Nakayama H, Fukuda K, Mizumoto M, Abei M, Shoda J, Thono E, Tsuboi K, Tokuuye K. A prospective study of hypofractionated proton beam therapy for patients with hepatocellular carcinoma. Int J Radiat Oncol Biol Phys. 2009;74:831–6.
Kawashima M, Furuse J, Nishio T, Konishi M, Ishii H, Kinoshita T, Nagase M, Nihei K, Ogino T. Phase II study of radiotherapy employing proton beam for hepatocellular carcinoma. J Clin Oncol. 2005;23:1839–46.
Nakayama H, Sugahara S, Tokita M, Fukuda K, Mizumoto M, Abei M, Shoda J, Sakurai H, Tsuboi K, Tokuuye K. Proton beam therapy for hepatocellular carcinoma: the University of Tsukuba experience. Cancer. 2009;115:5499–506.
Kang JI, Sufficool DC, Hsueh C-T, Wroe AJ, Patyal B, Reeves ME, Slater JD, Yang GY. A phase I trial of proton stereotactic body radiation therapy for liver metastases. J Gastrointest Oncol. 2019;10:112–7.
Petrelli F, Comito T, Barni S, Pancera G, Scorsetti M, Ghidini A, SBRT for CRC liver metastases. Stereotactic body radiotherapy for colorectal cancer liver metastases: a systematic review. Radiother Oncol. 2018;129:427–34.
Lim YJ, Koh J, Kim S, et al. Chemoradiation-induced alteration of programmed death-ligand 1 and CD8+ tumor-infiltrating lymphocytes identified patients with poor prognosis in rectal cancer: a matched comparison analysis. Int J Radiat Oncol Biol Phys. 2017;99:1216–24.
Twyman-Saint Victor C, Rech AJ, Maity A, et al. Radiation and dual checkpoint blockade activate non-redundant immune mechanisms in cancer. Nature. 2015;520:373–7.
Schoenhals JE, Seyedin SN, Tang C, Cortez MA, Niknam S, Tsouko E, Chang JY, Hahn SM, Welsh JW. Preclinical rationale and clinical considerations for radiotherapy plus immunotherapy: going beyond local control. Cancer J. 2016;22:130–7.
Monjazeb AM, Giobbie-Hurder A, Lako A, et al. A randomized trial of combined PD-L1 and CTLA-4 inhibition with targeted low-dose or hypofractionated radiation for patients with metastatic colorectal cancer. Clin Cancer Res. 2021;27:2470–2480. Phase II randomized study 8 Gy per day for 3 days vs. 2 Gy BID for 2 days combined with microsatellite stable CLM given in combination with anti-CTLA-4 and anti-PD-1 therapy. Although there was no improvement in extrahepatic disease, significant changes were noted in immune cell populations with treated tumor.
Ahmed KA, Fulp WJ, Berglund AE, Hoffe SE, Dilling TJ, Eschrich SA, Shridhar R, Torres-Roca JF. Differences between colon cancer primaries and metastases using a molecular assay for tumor radiation sensitivity suggest implications for potential oligometastatic SBRT patient selection. Int J Radiat Oncol Biol Phys. 2015;92:837–42.
Eccles C, Brock KK, Bissonnette J-P, Hawkins M, Dawson LA. Reproducibility of liver position using active breathing coordinator for liver cancer radiotherapy. Int J Radiat Oncol Biol Phys. 2006;64:751–9.
Valentine K, Cabrera T, Roberge D. Implanting metal fiducials to guide stereotactic liver radiation: McGill experience and review of current devices, techniques and complications. Technol Cancer Res Treat. 2014;13:253–8.
Boldrini L, Corradini S, Gani C, Henke L, Hosni A, Romano A, Dawson L. MR-guided radiotherapy for liver malignancies. Front Oncol. 2021. https://doi.org/10.3389/fonc.2021.616027.
Gani C, Boeke S, McNair H, et al. Marker-less online MR-guided stereotactic body radiotherapy of liver metastases at a 1.5 T MR-Linac - feasibility, workflow data and patient acceptance. Clin Transl Radiat Oncol. 2021;26:55–61.
Rosenberg SA, Henke LE, Shaverdian N, et al. A multi-institutional experience of MR-guided liver stereotactic body radiation therapy. Adv Radiat Oncol. 2019;4:142–9.
van Dams R, Wu TC, Kishan AU, et al. Ablative radiotherapy for liver tumors using stereotactic MRI guidance: a prospective phase I trial. Radiother Oncol. 2021;S0167–8140(21):06577–84. The first phase I study of MRgRT in treating liver lesions. High rates of LC – 1 year 94.7% and 2 year 79.6%. Improved LC was seen when BED >100 was delivered. No acute G3 or higher toxicities seen.
Ohri N, Tomé WA, Méndez Romero A, Miften M, Ten Haken RK, Dawson LA, Grimm J, Yorke E, Jackson A. Local control after stereotactic body radiation therapy for liver tumors. Int J Radiat Oncol Biol Phys. 2021;110:188–95.
Kokabi N, Galt JR, Xing M, Camacho JC, Barron BJ, Schuster DM, Kim HS. A simple method for estimating dose delivered to hepatocellular carcinoma after yttrium-90 glass-based radioembolization therapy: preliminary results of a proof of concept study. J Vasc Interv Radiol. 2014;25:277–87.
Garin E, Rolland Y, Edeline J, et al. Personalized dosimetry with intensification using 90Y-loaded glass microsphere radioembolization induces prolonged overall survival in hepatocellular carcinoma patients with portal vein thrombosis. J Nucl Med. 2015;56:339–46.
Alsultan AA, van Roekel C, Barentsz MW, Smits MLJ, Kunnen B, Koopman M, Bruijnen RCG, de Keizer B, Lam MGEH. Dose-response and dose-toxicity relationships for yttrium-90 glass radioembolization in patients with colorectal cancer liver metastases. J Nucl Med. 2021. https://doi.org/10.2967/jnumed.120.255745.
Saxena A, Meteling B, Kapoor J, Golani S, Morris DL, Bester L. Is yttrium-90 radioembolization a viable treatment option for unresectable, chemorefractory colorectal cancer liver metastases? A large single-center experience of 302 patients. Ann Surg Oncol. 2015;22:794–802.
Hickey R, Lewandowski RJ, Prudhomme T, et al. 90Y radioembolization of colorectal hepatic metastases using glass microspheres: safety and survival outcomes from a 531-patient multicenter study. J Nucl Med. 2016;57:665–71.
Wasan HS, Gibbs P, Sharma NK, et al. First-line selective internal radiotherapy plus chemotherapy versus chemotherapy alone in patients with liver metastases from colorectal cancer (FOXFIRE, SIRFLOX, and FOXFIRE-Global): a combined analysis of three multicentre, randomised, phase 3 trials. Lancet Oncol. 2017;18:1159–1171. Combined analysis of 3 phase III trials of SIRT combined with chemotherapy that demonstrated improved liver disease control although no improved survival compared to chemotherapy alone.
Gibbs P, Heinemann V, Sharma NK, et al. Effect of primary tumor side on survival outcomes in untreated patients with metastatic colorectal cancer when selective internal radiation therapy is added to chemotherapy: combined analysis of two randomized controlled studies. Clin Colorectal Cancer. 2018;17:e617–29.
Garlipp B, Gibbs P, Van Hazel GA, et al. Secondary technical resectability of colorectal cancer liver metastases after chemotherapy with or without selective internal radiotherapy in the randomized SIRFLOX trial. Br J Surg. 2019;106:1837–46.
Kurilova I, Bendet A, Fung EK, et al. Radiation segmentectomy of hepatic metastases with Y-90 glass microspheres. Abdom Radiol (NY). 2021;46:3428–36.
Riaz A, Gates VL, Atassi B, et al. Radiation segmentectomy: a novel approach to increase safety and efficacy of radioembolization. Int J Radiat Oncol Biol Phys. 2011;79:163–71.
Vouche M, Habib A, Ward TJ, et al. Unresectable solitary hepatocellular carcinoma not amenable to radiofrequency ablation: multicenter radiology-pathology correlation and survival of radiation segmentectomy. Hepatology. 2014;60:192–201.
Lewandowski RJ, Gabr A, Abouchaleh N, et al. Radiation segmentectomy: potential curative therapy for early hepatocellular carcinoma. Radiology. 2018;287:1050–8.
Meiers C, Taylor A, Geller B, Toskich B. Safety and initial efficacy of radiation segmentectomy for the treatment of hepatic metastases. J Gastrointest Oncol. 2018;9:311–5.
Tafti BA, Padia SA. Dosimetry of Y-90 microspheres utilizing Tc-99m SPECT and Y-90 PET. Semin Nucl Med. 2019;49:211–217. Discusses quantitative imaging with Tc-99m SPECT and Y-90 PET to help improve TARE dosimetry.
Abbott E, Young RS, Hale C, Mitchell K, Falzone N, Vallis KA, Kennedy A. Stereotactic inverse dose planning after yttrium-90 selective internal radiation therapy in hepatocellular cancer. Adv Radiat Oncol. 2021. https://doi.org/10.1016/j.adro.2020.11.002.
Mohamed M, Katz AW, Tejani MA, et al. Comparison of outcomes between SBRT, yttrium-90 radioembolization, transarterial chemoembolization, and radiofrequency ablation as bridge to transplant for hepatocellular carcinoma. Adv Radiat Oncol. 2015;1:35–42.
Sebastian N, Miller ED, Williams TM, Pardo DAD. Transarterial radioembolization (TARE) vs stereotactic body radiation therapy (SBRT) in the treatment of unresectable intrahepatic cholangiocarcinoma. Int J Radiat Oncol Biol Phys. 2018;102:e61.
Salem R, Johnson GE, Kim E, Riaz A, Bishay V, Boucher E, Fowers K, Lewandowski R, Padia SA. Yttrium-90 radioembolization for the treatment of solitary, unresectable HCC: the LEGACY study. Hepatology. 2021. https://doi.org/10.1002/hep.31819.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Radiation Therapy and Radiation Therapy Innovations in Colorectal Cancer
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ku, E., Yeakel, J., Gan, M. et al. The Role of Ablative Radiotherapy to Liver Oligometastases from Colorectal Cancer. Curr Colorectal Cancer Rep 17, 103–112 (2021). https://doi.org/10.1007/s11888-021-00472-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11888-021-00472-9