Abstract
Purpose of Review
Colorectal cancer (CRC) is a leading cause of cancer-related death and additional treatment options are urgently needed. Cytotoxic chemotherapy has been the mainstay of treatment options for patients for many years, including FOLFOX (leucovorin, 5-fluorouracil (5-FU), and oxaliplatin) or FOLFIRI (5-FU, leucovorin, and irinotecan) Here we review the current clinical use of systemic therapies for metastatic CRC and mechanisms of resistance to these agents.
Recent Findings
Biologic therapies, including anti-angiogenic and anti-epidermal growth factor monoclonal antibodies, have shown increased efficacy for patients with metastatic CRC. Most recently, immunotherapies have also been an option for some patients.
Summary
Identification of molecular markers predictive of response or resistance has led to enhanced ability to treat patients with metastatic CRC in a more personalized fashion.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Marley AR, Nan H. Epidemiology of colorectal cancer. Int J Mol Epidemiol Genet. 2016;7(3):105–14.
Laufman L, Bukowski RM, Collier MA, et al. A randomized double-blind trial of fluorouracil plus placebo versus fluorouracil plus oral leucovorin in patients with metastatic colorectal cancer. J Clin Oncol. 1993;11(10):1888.
Venook A, Niedzwiecki D, Lenz HJ, et al. CALGB/SWOG 80405: phase III trial of irinotecan/5-FU/leucovorin (FOLFIRI) or oxaliplatin/5-FU/leucovorin (mFOLFOX6) with bevacizumab (BV) or cetuximab (CET) for patients (pts) with KRAS wild-type (wt) untreated metastatic adenocarcinoma of the colon or rectum (MCRC). J Clin Oncol. 2017;32:5s (suppl; abstr LBA3).
Heidelberger C, Chaudhuri NK, Danenberg PV, et al. Fluorinated pyrimidines: a new class of tumor inhibitory compounds. Nature. 1957;179:663.
de Gramont A, Bosset JF, Milan C, et al. Randomized trial comparing monthly low-dose leucovorin and fluorouracil bolus with bimonthly high-dose leucovorin and fluorouracil bolus plus continuous infusion for advanced colorectal cancer: a French intergroup study. J Clin Oncol. 1997;15(2):808.
Van Kuilenburg ABP. Dihydropyrimidine dehydrogenase and the efficacy and toxicity of 5-fluorouracil. Eur J Cancer. 2004;40(7):939–50.
Parker WB, Cheng YC. Metabolism and mechanism of action of 5-fluorouracil. Pharmacol Ther. 1990;48:381–95.
Salonga D, Danenberg KD, Johnson MR, et al. Colorectal tumors responding to 5-fluorouracil have low gene expression levels of dihydropyrimidine dehydrogenase, thymidylate synthase, and thymidine phosphorylase. Clin Cancer Res. 2000;6:1322–7.
Leichman CG, Lenz HJ, Leichman L, et al. Quantitation of intratumoral thymidylate synthase expression predicts for disseminated colorectal cancer response and resistance to protracted-infusion fluorouracil and weekly leucovorin. J Clin Oncol. 1997;15:3223–9.
Lacopetta B, Grieu F, Joseph D, Elsaleh H. A polymorphism in the enhancer region of the thymidylate synthase promoter influences the survival of colorectal cancer patients treated with 5-fluorouracil. Br J Cancer. 2001;85:827–30.
Wei X, Wang W, Wang L, Zhang Y, Zhang X, Chen M, et al. MicroRNA-21 induces 5-fluorouracil resistance in human pancreatic cancer cells by regulating PTEN and PDC. Cancer Med. 2016;5(4):693–702.
Tomimaru Y, Eguchi H, Nagano H, Wada H, Tomokuni A, Kobayashi S, et al. MicroRNA-21 induces resistance to the anti-tumour effect of interferon-α/5-fluorouracil in hepatocellular carcinoma cells. Br J Cancer. 2010;103(10):1617–26.
Valeri N, Gasparini P, Braconi C, Paone A, Lovat F, Fabbri M, et al. MicroRNA-21 induces resistance to 5-fluorouracil by down-regulating human DNA MutS homolog 2 (hMSH2). Proc Natl Acad Sci USA. 2010;107(49):21098–103.
Tournigand C, Andre T, Achille E, et al. FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol. 2004;22(2):229.
Woynarowski JM, Faivre S, Herzig MC, Arnett B, Chapman WG, Trevino AV, et al. Oxaliplatin-induced damage of cellular DNA. Mol Pharm. 2000;58(5):920–7.
Bruno PM, Liu Y, Park GY, Murai J, Koch CE, Eisen TJ, et al. A supset of platinum-containing chemotherapeutic agents kill cells by inducing ribosome biogenesis stress. Nat Med. doi:10.1038/nm.4291.
Arnould S, Hennebelle I, Canal P, Bugat R, Guichard S. Cellular determinants of oxaliplatin sensitivity in colon cancer cell lines. Eur J Cancer. 2003;39(1):112–9.
Lenz HJ, Lee FC, Yau L, et al. MAVERICC, a phase 2 study of mFOLFOX6-bevacizumab (BV) vs FOLFIRI-BV with biomarker stratification as first-line (1L) chemotherapy (CT) in patients (pts) with metastatic colorectal cancer (mCRC). J Clin Oncol. 2016;34:(suppl 4S; abstr 493).
Martinez-Balibrea E, et al. Tumor-related molecular mechanisms of oxaliplatin resistance. Mol Cancer Ther. 2015;14(8):1767–76.
Samimi G, Katano K, Holzer AK, Safaei R, Howell SB. Modulation of the cellular pharmacology of cisplatin and its analogs by the copper exporters ATP7A and ATP7B. Mol Pharmacol. 2004;66:25–32.
Plasencia C, Martinez-Balibrea E, Martinez-Cardus A, Quinn DI, Abad A, Neamati N. Expression analysis of genes involved in oxaliplatin response and development of oxaliplatin-resistant HT29 colon cancer cells. Int J Oncol. 2006;29:225–35.
Martinez-Balibrea E, Martinez-Cardus A, Musulen E, Gines A, Manzano JL, Aranda E, et al. Increased levels of copper efflux transporter ATP7B are associated with poor outcome in colorectal cancer patients receiving oxaliplatin-based chemotherapy. Int J Cancer. 2009;124:2905–10.
Pommier Y, Leo E, Zhang H, Marchand C. DNA topoisomerases and their poisoning by anticancer and antibacterial drugs. Chem Biol. 2010;17:421–33.
Pommier Y. DNA topoisomerase I inhibitors: chemistry, biology, and interfacial inhibition. Chem Rev. 2009;109:2894–902.
Andre T, Louvet C, Maindrault-Goebel F, et al. CPT-11 (irinotecan) addition to bimonthly, high-dose leucovorin and bolus and continuous-infusion 5-fluorouracil (FOLFIRI) for pretreated metastatic colorectal cancer. GERCOR Eur J Cancer. 1999;35:1343.
Van Cutsem E, Lenz HJ, Kohne CH, et al. Fluorouracil, leucovorin, and irinotecan plus cetuximab treatment and RAS mutations in colorectal cancer. J Clin Oncol. 2015;33(7):692–700.
Fujita K, Kubota Y, Ishida H, Sasaki Y. Irinotecan, a key chemotherapeutic drug for metastatic colorectal cancer. World J Gastroenterol. 2015;21(43):12234–48.
Kawato Y, Aonuma M, Hirota Y, et al. Intracellular roles of SN-38, a metabolite of the camptothecin derivative CPT-11, in the antitumor effect of CPT-11. Cancer Res. 1991;51:4187–91.
Xu Y, Villalona-Calero MA. Irinotecan: mechanisms of tumor resistance and novel strategies for modulating its activity. Ann Oncol. 2002;13:1841–51.
van Ark-Otte J, Kedde MA, van der Vijgh WJ, et al. Determinants of CPT-11 and SN-38 activities in human lung cancer cells. Br J Cancer. 1998;77:2171–6.
Chu XY, Suzuki H, Ueda K, et al. Active efflux of CPT-11 and its metabolites in human KB-derived cell lines. J Pharmacol Exp Ther. 1999;288:735–41.
Temmink OH, Emura T, de Bruin M, Fukushima M, Peters GJ. Therapeutic potential of the dual-targeted TAS-102 formulation in the treatment of gastrointestinal malignancies. Cancer Sci. 2007;98:779–89.
Dexter DL, Wolberg WH, Ansfield FJ, Helson L, Heidelberger C. The clinical pharmacology of 5-trifluoromethyl-2′-deoxyuridine. Cancer Res. 1972;32:247–53.
Fukushima M, Suzuki N, Emura T, Yano S, Kazuno H, Tada Y, et al. Structure and activity of specific inhibitors of thymidine phosphorylase to potentiate the function of antitumor 2′-deoxyribonucleosides. Biochem Parmacol. 2000;59:1227–36.
Murakami Y, Kazano H, Emura T, Tsujimoto H, Suzuki N, Fukushima M. Different mechanisms of acquired resistance to fluorinated pyrimidines in human colorectal cancer cells. Int J Oncol. 2000;17:277–83.
Mayer RJ, Van Cutsem E, Falcone A, et al. Randomized trial of TAS-102 for refractory metastatic colorectal cancer. N Engl J Med. 2015;372(20):1909–19.
Olaf H, et al. Trifluorothymidine resistance is associated with decreased thymidine kinase and equilibrative nucleoside transporter expression or increased secretory phospholipase A2. Mol Cancer Ther. 2010;9(4):1047–57.
Fakih M. The evolving role of VEGF-targeted therapies in the treatment of metastatic colorectal cancer. Expert Rev Anticancer Ther. 2013;4:427–38.
Gambardella V, Tarazona N, Cejalvo JM, Roselló S, Certantes A. Clinical pharmacokinetics and pharmacodynamics of ramicirumab in the treatment of colorectal cancer. Expert Opin Drug Metab Toxicol. 2016;12(4):449–56.
Qu CY, Zheng Y, Zhang Y, Shen F, Cao J, Xu LM. Value of bevacizumab in treatment of colorectal cancer: a meta-analysis. World J Gastroenterol. 2015;21(16):50-72–5080.
Bergers G, Hanahan D. Modes of resistance to anti-angiogenic therapy. Nat Rev Cancer. 2008;8:592–603.
Casanovas O, Hicklin DJ, Bergers G, Hanahan D. Drug resistance by evasion of antiangiogenic targeting of VEGF signaling in late-stage pancreatic islet tumors. Cancer Cell. 2005;8:299–309.
Blouw B, et al. The hypoxic response of tumors is dependent on their microenvironment. Cancer Cell. 2003;4:133–46.
Kurai J, Chikumi H, Hashimoto K, et al. Antibody-dependent cellular cytotoxicity mediated by cetuximab against lung cancer cell lines. Clin Cancer Res. 2007;13(5):1552–61.
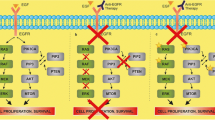
• Deming D, Holen K. KRAS mutation analysis prior to EGFR-directed therapy for metastatic colorectal cancer: a review and cost analysis. Curr Cancer Ther Rev. 2010;6(4):256–61. This manuscript reviews the ability of KRAS testing to predict resistance to anti-EGFR directed therapies and the immense cost savings as the result of this becoming a standard practice.
Tran NH, Cavalcante LL, Lubner SJ, et al. Precision medicine in colorectal cancer: the molecular profile alters treatment strategies. Ther Adv Med Oncol. 2015;7(5):252–62.
Al-Shamsi HO, Alhazzani W, Wolff RA. Extended RAS testing in metastatic colorectal cancer-refining the predictive molecular biomarkers. J Gastrointest Oncol. 2015;6(3):314–21.
Turk A, Deming DA. BRAF mutation in colorectal cancer. Personalized Medicine in Oncology. 2016;5(1).
• Venook AP, Niedzwiecki D, Innocenti F, et al. Impact of primary (1°) tumor location on overall survival (OS) and progression-free survival (PFS) in patients (pts) with metastatic colorectal cancer (mCRC): analysis of CALGB/SWOG 80405 (Alliance). J Clin Oncol. 2016;34:(suppl; abstr 3504). This study demonstrates the importance of sidedness in the use of anti-EGFR directed therapies for the treatment of metastatic CRC.
Benson AB 3rd, Venook AP, Cederquist L, et al. Colon cancer, version 1.2017, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw. 2017;15(3):370–98.
Lee MS, Advani SM, Morris J, et al. Association of primary site and molecular featurs with progression-free survival and overall survival of metastatic colorectal cancer after anti-epidermal growth factor therapy. J Clin Oncol. 34:(suppl;abstr 3506).
Seow HF, Yip WK, Fifis T. Advances in targeted and immunobased therapies for colorectal cancer in the genomic era. Oncol Targets Ther. 2016;9:1899–920.
•• Le DT, Uram JN, Wang H, et al. PD-1 blockade in tumors with mismatch-repair deficiency. N Engl J Med. 2015;372(26):2509–20. This was the first description of the use of anti-PD1 therapies for the treatment of patients with mismatch repair deficient cancers.
Overman MJ, Lonardi S, Leone F, et al. Nivolumab in patients with DNA mismatch repair deficient/microsatellite instability high metastatic colorectal cancer: update from CheckMate 142. J Clin Oncol. 2017;35:(suppl 4S;abstract 519).
Restifo NP, Smyth MJ, Snyder A. Acquired resistance to immunotherapy and future challenges. Nat Rev Cancer. 2016;16:121–6.
Restifo NP, Marnicola FM, Kawakami Y, Taubenberger J, Yannelli JR, Rosenber SA. Loss of functional beta 2-microglobulin in metastatic melanomas from five patients receiving immunotherapy. J Natl Cancer Inst. 1996;88(2):100–8.
Garrido F, Aptsiauri N, Doordujin EM, Lora AMG, van Hall T. The urgent need to recover MHC class I in cancers for effective immunotherapy. Curr Opin Immunol. 2016;39:44–51.
Zhang X, Kelaria S, Kerstetter J, Wang J. The functional and prognostic implications of regulatory T cells in colorectal carcinoma. J Gastrointest Oncol. 2015;6(3):307–13.
Hope C, Foulcer S, Jagodinsky J, et al. Immunoregulatory roles of versican in the myeloma microenvironment. Blood. 2016;128(5):680–5.
Acknowledgments
The authors wish to thank the UW Cellular and Molecular Pathology Graduate program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Philip Emmerich declares that he has no conflict of interest.
Linda Clipson declares that she has no conflict of interest.
Dustin A. Deming has received research funding through grants from Merck and Millennium.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Financial Support
This project was supported by P30 CA014520 (Core Grant, University of Wisconsin Carbone Cancer Center).
Additional information
This article is part of the Topical Collection on Basic Science Foundations in Colorectal Cancer
Rights and permissions
About this article
Cite this article
Emmerich, P., Clipson, L. & Deming, D.A. Resistance Mechanisms to Colorectal Cancer Therapeutics and the Clinical Implications. Curr Colorectal Cancer Rep 13, 334–340 (2017). https://doi.org/10.1007/s11888-017-0374-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11888-017-0374-5