Abstract
Purpose of Review
Our understanding of the fundamental cellular and molecular factors leading to atrial fibrillation (AF) remains stagnant despite significant advancement in ablation and device technologies. Diagnosis and prevention strategies fall behind that of treatment, but expanding knowledge in AF genetics holds the potential to drive progress. We aim to review how an understanding of the genetic contributions to AF can guide an approach to individualized risk stratification and novel avenues in drug discovery.
Recent Findings
Rare familial forms of AF identified monogenic contributions to the development of AF. Genome-wide association studies (GWAS) further identified single-nucleotide polymorphisms (SNPs) suggesting polygenic and multiplex nature of this common disease. Polygenic risk scores accounting for the multitude of associated SNPs that each confer mildly elevated risk have been developed to translate genetic information into clinical practice, though shortcomings remain. Additionally, novel laboratory methods have been empowered by recent genetic findings to enhance drug discovery efforts.
Summary
AF is increasingly recognized as a disease with a significant genetic component. With expanding sequencing technologies and accessibility, polygenic risk scores can help identify high risk individuals. Advancement in digital health tools, artificial intelligence and machine learning based on standard electrocardiograms, and genomic driven drug discovery may be integrated to deliver a sophisticated level of precision medicine in this modern era of emphasis on prevention. Randomized, prospective studies to demonstrate clinical benefits of these available tools are needed to validate this approach.
Similar content being viewed by others
Abbreviations
- AI:
-
Artificial intelligence
- AF:
-
Atrial fibrillation
- CAD:
-
Coronary artery disease
- ECG:
-
Electrocardiogram
- EHR:
-
Electronic health record
- HT:
-
High throughput
- iPSC:
-
Induced pluripotent stem cell
- GWAS:
-
Genome-wide association study
- LD:
-
Linkage disequilibrium
- PRS:
-
Polygenic risk score
References
Papers of particular interest, published recently, have been highlighted as:
• Of importance
Kaarisalo MM, Immonen-Raiha P, Marttila RJ, Salomaa V, Kaarsalo E, Salmi K, et al. Atrial fibrillation and stroke. Mortality and causes of death after the first acute ischemic stroke. Stroke. 1997;28(2):311–5. https://doi.org/10.1161/01.str.28.2.311.
Staerk L, Sherer JA, Ko D, Benjamin EJ, Helm RH. Atrial Fibrillation: Epidemiology, Pathophysiology, and Clinical Outcomes. Circ Res. 2017;120(9):1501–17. https://doi.org/10.1161/CIRCRESAHA.117.309732.
Williams BA, Chamberlain AM, Blankenship JC, Hylek EM, Voyce S. Trends in Atrial Fibrillation Incidence Rates Within an Integrated Health Care Delivery System, 2006 to 2018. JAMA Netw Open. 2020;3(8):e2014874. https://doi.org/10.1001/jamanetworkopen.2020.14874.
Colilla S, Crow A, Petkun W, Singer DE, Simon T, Liu X. Estimates of current and future incidence and prevalence of atrial fibrillation in the U.S. adult population. Am J Cardiol. 2013;112(8):1142–7. https://doi.org/10.1016/j.amjcard.2013.05.063.
Chung MK, Eckhardt LL, Chen LY, Ahmed HM, Gopinathannair R, Joglar JA, et al. Lifestyle and Risk Factor Modification for Reduction of Atrial Fibrillation: A Scientific Statement From the American Heart Association. Circulation. 2020;141(16):e750–72. https://doi.org/10.1161/CIR.0000000000000748.
Kannel WB, Wolf PA, Benjamin EJ, Levy D. Prevalence, incidence, prognosis, and predisposing conditions for atrial fibrillation: population-based estimates. Am J Cardiol. 1998;82(8A):2N–9N. https://doi.org/10.1016/s0002-9149(98)00583-9.
Dzeshka MS, Shantsila A, Shantsila E, Lip GYH. Atrial Fibrillation and Hypertension. Hypertension. 2017;70(5):854–61. https://doi.org/10.1161/HYPERTENSIONAHA.117.08934.
Kanagala R, Murali NS, Friedman PA, Ammash NM, Gersh BJ, Ballman KV, Shamsuzzaman ASM, Somers VK. Obstructive sleep apnea and the recurrence of atrial fibrillation. Circulation. 2003;107(20):2589–94. https://doi.org/10.1161/01.CIR.0000068337.25994.21.
Nalliah CJ, Sanders P, Kottkamp H, Kalman JM. The role of obesity in atrial fibrillation. Eur Heart J. 2016;37(20):1565–72. https://doi.org/10.1093/eurheartj/ehv486.
Middeldorp ME, Pathak RK, Meredith M, Mehta AB, Elliott AD, Mahajan R, et al. Prevention and regressive effect of weight-loss and risk factor modification on atrial fibrillation: the REVERSE-AF study. Europace. 2018;20(12):1929–35. https://doi.org/10.1093/europace/euy117.
Haissaguerre M, Jais P, Shah DC, Takahashi A, Hocini M, Quiniou G, et al. Spontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins. N Engl J Med. 1998;339(10):659–66. https://doi.org/10.1056/NEJM199809033391003.
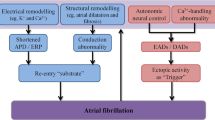
Iwasaki YK, Nishida K, Kato T, Nattel S. Atrial fibrillation pathophysiology: implications for management. Circulation. 2011;124(20):2264–74. https://doi.org/10.1161/CIRCULATIONAHA.111.019893.
Steinberg JS, Varma N, Cygankiewicz I, Aziz P, Balsam P, Baranchuk A, et al. 2017 ISHNE-HRS expert consensus statement on ambulatory ECG and external cardiac monitoring/telemetry. Heart Rhythm. 2017;14(7):e55–e96. https://doi.org/10.1016/j.hrthm.2017.03.038.
Tayal AH, Tian M, Kelly KM, Jones SC, Wright DG, Singh D, Jarouse J, Brillman J, Murali S, Gupta R. Atrial fibrillation detected by mobile cardiac outpatient telemetry in cryptogenic TIA or stroke. Neurology. 2008;71(21):1696–701. https://doi.org/10.1212/01.wnl.0000325059.86313.31.
Bhatt A, Majid A, Razak A, Kassab M, Hussain S, Safdar A. Predictors of occult paroxysmal atrial fibrillation in cryptogenic strokes detected by long-term noninvasive cardiac monitoring. Stroke Res Treat. 2011;2011:172074. https://doi.org/10.4061/2011/172074.
Sanna T, Diener H-C, Passman RS, Di Lazzaro V, Bernstein RA, Morillo CA, et al. Cryptogenic stroke and underlying atrial fibrillation. N Engl J Med. 2014;370(26):2478–86. https://doi.org/10.1056/NEJMoa1313600.
Flint AC, Banki NM, Ren X, Rao VA, Go AS. Detection of paroxysmal atrial fibrillation by 30-day event monitoring in cryptogenic ischemic stroke: the stroke and monitoring for PAF in real time (SMART) registry. Stroke. 2012;43(10):2788–90. https://doi.org/10.1161/STROKEAHA.112.665844.
Stahrenberg R, Weber-Krüger M, Seegers J, Edelman F, Lahno R, Haase B, et al. Enhanced detection of paroxysmal atrial fibrillation by early and prolonged continuous holter monitoring in patients with cerebral ischemia presenting in sinus rhythm. Stroke. 2010;41(12):2884–8. https://doi.org/10.1161/STROKEAHA.110.591958.
Buck BH, Hill MD, Quinn FR, Butcher KS, Menon BK, Gulamhusein S, et al. Effect of Implantable vs Prolonged External Electrocardiographic Monitoring on Atrial Fibrillation Detection in Patients With Ischemic Stroke: The PER DIEM Randomized Clinical Trial. JAMA. 2021;325(21):2160–8. https://doi.org/10.1001/jama.2021.6128.
Bernstein RA, Kamel H, Granger CB, Piccini JB, Sethi PP, Katz JM, et al. Effect of Long-term Continuous Cardiac Monitoring vs Usual Care on Detection of Atrial Fibrillation in Patients With Stroke Attributed to Large- or Small-Vessel Disease: The STROKE-AF Randomized Clinical Trial. JAMA. 2021;325(21):2169–77. https://doi.org/10.1001/jama.2021.6470.
Ha A, Verma S, Mazer D, Quan A, Latter D, Yanagawa B, et al. Abstract 18618: Enhanced monitoring for atrial fibrillation following cardiac surgery-SEARCH-AF Cardiolink Randomized Trial. Circulation. 2020;142(24). https://doi.org/10.1161/CIR.0000000000000940.
Healey JS, Connolly SJ, Gold MR, Israel CW, Van Gelder IC, Capucci A, et al. Subclinical atrial fibrillation and the risk of stroke. N Engl J Med. 2012;366(2):120–9. https://doi.org/10.1056/NEJMoa1105575.
Svennberg E, Engdahl J, Al-Khalili F, Friberg L, Frykman V, Rosenqvist M. Mass Screening for Untreated Atrial Fibrillation: The STROKESTOP Study. Circulation. 2015;131(25):2176–84. https://doi.org/10.1161/CIRCULATIONAHA.114.014343.
Chan N, Choy C. Screening for atrial fibrillation in 13 122 Hong Kong citizens with smartphone electrocardiogram. Heart. 2017;103(1):24–31. https://doi.org/10.1136/heartjnl-2016-309993.
Kaasenbrood F, Hollander M, Rutten FH, Gerhards LJ, Hoes AW, Tieleman RG. Yield of screening for atrial fibrillation in primary care with a hand-held, single-lead electrocardiogram device during influenza vaccination. Europace. 2016;18(10):1514–20. https://doi.org/10.1093/europace/euv426.
Tison GH, Sanchez JM, Ballinger B, Singh A, Olgin JE, Pletcher MJ, et al. Passive Detection of Atrial Fibrillation Using a Commercially Available Smartwatch. JAMA Cardiol. 2018;3(5):409–16. https://doi.org/10.1001/jamacardio.2018.0136.
Lowres N, Neubeck L, Salkeld G, Krass I, McLachlan AJ, Redfern J, et al. Feasibility and cost-effectiveness of stroke prevention through community screening for atrial fibrillation using iPhone ECG in pharmacies. The SEARCH-AF study. Thromb Haemost. 2014;111(6):1167–76. https://doi.org/10.1160/TH14-03-0231.
• Attia ZI, Noseworthy PA, Lopez-Jimenez F, Asirvatham SJ, Deshmukh AJ, Gersh BJ, et al. An artificial intelligence-enabled ECG algorithm for the identification of patients with atrial fibrillation during sinus rhythm: a retrospective analysis of outcome prediction. Lancet. 2019;394(10201):861–7. https://doi.org/10.1016/S0140-6736(19)31721-0. This study highlights the power of AI/ML driven algorithms to identify patients with AF from electrocardiograms even when in normal sinus rhythm.
Raghunath S, Pfeifer JM, Ulloa-Cerna AE, Nemani A, Carbonati T, Jing L, et al. Deep Neural Networks Can Predict New-Onset Atrial Fibrillation From the 12-Lead ECG and Help Identify Those at Risk of Atrial Fibrillation-Related Stroke. Circulation. 2021;143(13):1287–98. https://doi.org/10.1161/CIRCULATIONAHA.120.047829.
Verweij N, Benjamins JW, Morley MP, van de Vegte YJ, Teumer A, Trenkwalder T, et al. The Genetic Makeup of the Electrocardiogram. Cell Syst. 2020;11(3):229-238 e5. https://doi.org/10.1016/j.cels.2020.08.005.
Alonso A, Norby FL. Predicting Atrial Fibrillation and Its Complications. Circ J. 2016;80(5):1061–6. https://doi.org/10.1253/circj.CJ-16-0239.
Schnabel RB, Sullivan LM, Levy D, Pencina MJ, Massaro JM, D’Agostino RB Sr., et al. Development of a risk score for atrial fibrillation (Framingham Heart Study): a community-based cohort study. Lancet. 2009;373(9665):739–45. https://doi.org/10.1016/S0140-6736(09)60443-8.
Chamberlain AM, Agarwal SK, Folsom AR, Soliman EZ, Chambless LE, Crow R, Ambrose M, Alonso A. A clinical risk score for atrial fibrillation in a biracial prospective cohort (from the Atherosclerosis Risk in Communities [ARIC] study). Am J Cardiol. 2011;107(1):85–91. https://doi.org/10.1016/j.amjcard.2010.08.049.
Alonso A, Roetker NS, Soliman EZ, Chen LY, Greenland P, Heckbert SR. Prediction of Atrial Fibrillation in a Racially Diverse Cohort: The Multi-Ethnic Study of Atherosclerosis (MESA). J Am Heart Assoc. 2016;5(2). https://doi.org/10.1161/JAHA.115.003077.
Everett BM, Cook NR, Conen D, Chasman DI, Ridker PM, Albert CM. Novel genetic markers improve measures of atrial fibrillation risk prediction. Eur Heart J. 2013;34(29):2243–51. https://doi.org/10.1093/eurheartj/eht033.
Christophersen IE, Yin X, Larson MG, Lubitz SA, Magnani JW, McManus DD, Ellinor PT, Benjamin EJ. A comparison of the CHARGE-AF and the CHA2DS2-VASc risk scores for prediction of atrial fibrillation in the Framingham heart study. Am Heart J. 2016;178:45–54. https://doi.org/10.1016/j.ahj.2016.05.004.
Kolek MJ, Graves AJ, Xu M, Bian A, Teixeira PL, Shoemaker MB, et al. Evaluation of a Prediction Model for the Development of Atrial Fibrillation in a Repository of Electronic Medical Records. JAMA Cardiol. 2016;1(9):1007–13. https://doi.org/10.1001/jamacardio.2016.3366.
Kolek MJ, Muehlschlegel JD, Bush WS, Parvez B, Murray KT, Stein CM, et al. Genetic and clinical riskprediction model for postoperative atrial fibrillation. Circ ArrhythmElectrophysiol. 2015;8(1):25–31. https://doi.org/10.1161/CIRCEP.114.002300.
Lubitz SA, Yin X, Lin HJ, Kolek M, Smith JG, Trompet S, et al. Genetic Risk Prediction of Atrial Fibrillation. Circulation. 2017;135(14):1311–20. https://doi.org/10.1161/CIRCULATIONAHA.116.024143.
• Muse ED, Wineinger NE, Spencer EG, Peters M, Henderson R, Zhang Y, et al. Validation of a genetic risk score for atrial fibrillation: A prospective multicenter cohort study. PLoS Med. 2018;15(3):e1002525. https://doi.org/10.1371/journal.pmed.1002525. A prospective evaluation of an AF PRS in a clinical setting.
Okubo Y, Nakano Y, Ochi H, Onohara Y, Tokuyama T, Motoda C, et al. Predicting atrial fibrillation using a combination of genetic risk score and clinical risk factors. Hear Rhythm. 2020;17(5 Pt A):699–705. https://doi.org/10.1016/j.hrthm.2020.01.006.
Holmes DR, Reddy VY, Turi ZG, Doshi SK, Sievert H, Buchbinder M, Mullin CM, Sick P. Percutaneous closure of the left atrial appendage versus warfarin therapy for prevention of stroke in patients with atrial fibrillation: a randomised non-inferiority trial. Lancet. 2009;374(9689):534–42. https://doi.org/10.1016/S0140-6736(09)61343-X.
Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgren J, Parekh A, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;361(12):1139–51. https://doi.org/10.1056/NEJMoa0905561.
Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, et al. Rivaroxaban versus warfarin in nonvalvularatrial fibrillation. N Engl J Med. 2011;365(10):883–91. https://doi.org/10.1056/NEJMoa1009638.
Granger CB, Alexander JH, McMurray JJV, Lopes RD, Hylek EM, Hanna M, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2011;365(11):981–92. https://doi.org/10.1056/NEJMoa1107039.
Giugliano RP, Ruff CT, Braunwald E, Murphy SA, Wiviott SD, Halperin JL, et al. Edoxaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2013;369(22):2093–104. https://doi.org/10.1056/NEJMoa1310907.
Kampouraki E, Kamali F. Pharmacogenetics of anticoagulants used for stroke prevention in patients with atrial fibrillation. Expert Opin Drug Metab Toxicol. 2019;15(6):449–58. https://doi.org/10.1080/17425255.2019.1623878.
Raymond J, Imbert L, Cousin T, Duflot T, Varin R, Wils J, et al. Pharmacogenetics of Direct Oral Anticoagulants: A Systematic Review. J Pers Med. 2021;11(1). https://doi.org/10.3390/jpm11010037.
Parameswaran R, Al-Kaisey AM, Kalman JM. Catheter ablation for atrial fibrillation: current indications and evolving technologies. Nat Rev Cardiol. 2021;18(3):210–25. https://doi.org/10.1038/s41569-020-00451-x.
• Roselli C, Chaffin MD, Weng LC, Aeschbacher S, Ahlberg G, Albert CM, et al. Multi-ethnic genome-wide association study for atrial fibrillation. Nat Genet. 2018;50(9):1225–33. https://doi.org/10.1038/s41588-018-0133-9. This study represents the largest meta-analaysis of AF GWAS to date and identified a large number of loci associated with AF.
Choi SH, Weng LC, Roselli C, Lin H, Haggerty CM, Shoemaker MB, et al. Association Between Titin Loss-of-Function Variants and Early-Onset Atrial Fibrillation. JAMA. 2018;320(22):2354–64. https://doi.org/10.1001/jama.2018.18179.
Caglayan AO. Different aspects of atrial fibrillation genetics. Interact Cardiovasc Thorac Surg. 2010;11(6):779–83. https://doi.org/10.1510/icvts.2010.245910.
Wolff L. Familial Auricular Fibrillation. N Engl J Med. 1943;229(10):396–8. https://doi.org/10.1056/nejm194309022291002.
Kopecky SL, Gersh BJ, McGoon MD, Whisnant JP, Holmes DR Jr., Ilstrup DM. The natural history of lone atrial fibrillation. A population-based study over three decades. N Engl J Med. 1987;317(11):669–74. https://doi.org/10.1056/NEJM198709103171104.
Brand FN, Abbott RD, Kannel WB, Wolf PA. Characteristics and prognosis of lone atrial fibrillation. 30-year follow-up in the Framingham study. JAMA. 1985;254(24):3449–53.
Furberg CD, Psaty BM, Manolio TA, Gardin JM, Smith VE, Rautaharju PM. Prevalence of atrial fibrillation in elderly subjects (the cardiovascular health study). Am J Cardiol. 1994;74(3):236–41. https://doi.org/10.1016/0002-9149(94)90363-8.
Weng LC, Choi SH, Klarin D, Smith JG, Loh PR, Chaffin M, et al. Heritability of Atrial Fibrillation. Circ Cardiovasc Genet. 2017;10(6). https://doi.org/10.1161/CIRCGENETICS.117.001838.
Torkamani A, Wineinger NE, Topol EJ. The personal and clinical utility of polygenic risk scores. Nat Rev Genet. 2018;19(9):581–90. https://doi.org/10.1038/s41576-018-0018-x.
Fox CS, Parise H, D’Agostino RBS, Lloyd-Jones DM, Vasan RS, Wang TJ, et al. Parental atrial fibrillation as a risk factor for atrial fibrillation in offspring. JAMA. 2004;291(23):2851–5. https://doi.org/10.1001/jama.291.23.2851.
Ellinor PT, Yoerger DM, Ruskin JN, MacRae CA. Familial aggregation in lone atrial fibrillation. Hum Genet. 2005;118(2):179–84. https://doi.org/10.1007/s00439-005-0034-8.
Yang YQ, Zhang XL, Wang XH, Tan HW, Shi HF, Fang WY, et al. Familial aggregation of lone atrial fibrillation in the Chinese population. Intern Med. 2010;49(22):2385–91. https://doi.org/10.2169/internalmedicine.49.4130.
Arnar DO, Thorvaldsson S, Manolio TA, Thorgeirsson G, Kristjansson K, Hakonarson H, et al. Familial aggregation of atrial fibrillation in Iceland. Eur Heart J. 2006;27(6):708–12. https://doi.org/10.1093/eurheartj/ehi727.
Darbar D, Herron KJ, Ballew JD, Jahangir A, Gersh BJ, Shen WK, et al. Familial atrial fibrillation is a genetically heterogeneous disorder. J Am Coll Cardiol. 2003;41(12):2185–92. https://doi.org/10.1016/s0735-1097(03)00465-0.
Campuzano O, Brugada R. Genetics of familial atrial fibrillation. Europace. 2009;11(10):1267–71. https://doi.org/10.1093/europace/eup199.
Lubitz SA, Yi BA, Ellinor PT. Genetics of atrial fibrillation. Cardiol Clin. 2009;27(1):25–33. https://doi.org/10.1016/j.ccl.2008.09.007.
Lubitz SA, Yin X, Fontes JD, Magnani JW, Rienstra M, Pai M, et al. Association between familial atrial fibrillation and risk of new-onset atrial fibrillation. JAMA. 2010;304(20):2263–9. https://doi.org/10.1001/jama.2010.1690.
Brugada R, Tapscott T, Czernuszewicz GZ, Marian AJ, Iglesias A, Mont L, et al. Identification of a genetic locus for familial atrial fibrillation. N Engl J Med. 1997;336(13):905–11. https://doi.org/10.1056/NEJM199703273361302.
Ellinor PT, Shin JT, Moore RK, Yoerger DM, MacRae CA. Locus for atrial fibrillation maps to chromosome 6q14-16. Circulation. 2003;107(23):2880–3. https://doi.org/10.1161/01.CIR.0000077910.80718.49.
Chen Y-H, Xu S-J, Bendahhou S, et al. KCNQ1 gain-of-function mutation in familial atrial fibrillation. Science. 2003;299:251–4. https://doi.org/10.1126/science.1077771.
Olson TM, Michels VV, Ballew JD, Reyna SP, Karst ML, Herron KJ, Horton SC, Rodeheffer RJ, Anderson JL. Sodium channel mutations and susceptibility to heart failure and atrial fibrillation. JAMA. 2005;293:447–54. https://doi.org/10.1001/jama.293.4.447.
Makiyama T, Akao M, Shizuta S, et al. A novel SCN5A gain-of-function mutation M1875T associated with familial atrial fibrillation. J Am Coll Cardiol. 2008;52:1326–34. https://doi.org/10.1016/j.jacc.2008.07.013.
Watanabe H, Darbar D, Kaiser DW, Jiramongkolchai K, Chopra S, Donahue BS, Kannankeril PJ, Roden DM. Mutations in sodium channel β1- and β2-subunits associated with atrial fibrillation. Circ Arrhythm Electrophysiol. 2009;2:268–75. https://doi.org/10.1161/CIRCEP.108.779181.
Oberti C, Wang L, Li L, Dong J, Rao S, Du W, Wang Q. Genome-wide linkage scan identifies a novel genetic locus on chromosome 5p13 for neonatal atrial fibrillation associated with sudden death and variable cardiomyopathy. Circulation. 2004;110:3753–9. https://doi.org/10.1161/01.CIR.0000150333.87176.C7.
Gollob MH, Jones DL, Krahn AD, et al. Somatic mutations in the connexin 40 gene (GJA5) in atrial fibrillation. N Engl J Med. 2006;354:2677–88. https://doi.org/10.1056/NEJMoa052800.
Jiang J-Q, Shen F-F, Fang W-Y, Liu X, Yang Y-Q. Novel GATA4 mutations in lone atrial fibrillation. Int J Mol Med. 2011;28:1025–32. https://doi.org/10.3892/ijmm.2011.783.
Zhabyeyev P, Hiess F, Wang R, Liu Y, Wayne Chen SR, Oudit GY. S4153R is a gain-of-function mutation in the cardiac Ca(2+) release channel ryanodine receptor associated with catecholaminergic polymorphic ventricular tachycardia and paroxysmal atrial fibrillation. Can J Cardiol. 2013;29:993–6. https://doi.org/10.1016/j.cjca.2012.12.019.
Wang X-H, Huang C-X, Wang Q, Li R-G, Xu Y-J, Liu X, Fang W-Y, Yang Y-Q. A novel GATA5 loss-of-function mutation underlies lone atrial fibrillation. Int J Mol Med. 2013;31:43–50. https://doi.org/10.3892/ijmm.2012.1189.
Postma AV, van de Meerakker JBA, Mathijssen IB, Barnett P, Christoffels VM, Ilgun A, Lam J, Wilde AAM, Lekanne Deprez RH, Moorman AFM. A gain-of-function TBX5 mutation is associated with atypical Holt-Oram syndrome and paroxysmal atrial fibrillation. Circ Res. 2008;102:1433–42. https://doi.org/10.1161/CIRCRESAHA.107.168294.
Kurabayashi M, Komuro I, Tsuchimochi H, Takaku F, Yazaki Y. Molecular cloning and characterization of human atrial and ventricular myosin alkali light chain cDNA clones. J Biol Chem. 1988;263:13930–6.
Hodgson-Zingman DM, Karst ML, Zingman LV, Heublein DM, Darbar D, Herron KJ, Ballew JD, de Andrade M, Burnett JCJ, Olson TM. Atrial natriuretic peptide frameshift mutation in familial atrial fibrillation. N Engl J Med. 2008;359:158–65. https://doi.org/10.1056/NEJMoa0706300.
Orr N, Arnaout R, Gula LJ, et al. A mutation in the atrial-specific myosin light chain gene (MYL4) causes familial atrial fibrillation. Nat Commun. 2016;7:11303. https://doi.org/10.1038/ncomms11303.
Visscher PM, Wray NR, Zhang Q, Sklar P, McCarthy MI, Brown MA, Yang J. 10 years of GWAS discovery: biology, function, and translation. Am J Hum Genet. 2017;101:5–22. https://doi.org/10.1016/j.ajhg.2017.06.005.
Gudbjartsson DF, Arnar DO, Helgadottir A, et al. Variants conferring risk of atrial fibrillation on chromosome 4q25. Nature. 2007;448:353–7. https://doi.org/10.1038/nature06007.
Kääb S, Darbar D, van Noord C, et al. Large scale replication and meta-analysis of variants on chromosome 4q25 associated with atrial fibrillation. Eur Heart J. 2009;30:813–9. https://doi.org/10.1093/eurheartj/ehn578.
Kirchhof P, Kahr PC, Kaese S, et al. PITX2c is expressed in the adult left atrium, and reducing Pitx2c expression promotes atrial fibrillation inducibility and complex changes in gene expression. Circ Cardiovasc Genet. 2011;4:123–33. https://doi.org/10.1161/CIRCGENETICS.110.958058.
Lubitz SA, Sinner MF, Lunetta KL, et al. Independent susceptibility markers for atrial fibrillation on chromosome 4q25. Circulation. 2010;122:976–84. https://doi.org/10.1161/CIRCULATIONAHA.109.886440.
Lubitz SA, Lunetta KL, Lin H, et al. Novel genetic markers associate with atrial fibrillation risk in Europeans and Japanese. J Am Coll Cardiol. 2014;63:1200–10. https://doi.org/10.1016/j.jacc.2013.12.015.
Body SC, Collard CD, Shernan SK, et al. Variation in the 4q25 chromosomal locus predicts atrial fibrillation after coronary artery bypass graft surgery. Circ Cardiovasc Genet. 2009;2:499–506. https://doi.org/10.1161/CIRCGENETICS.109.849075.
Husser D, Adams V, Piorkowski C, Hindricks G, Bollmann A. Chromosome 4q25 variants and atrial fibrillation recurrence after catheter ablation. J Am Coll Cardiol. 2010;55:747–53. https://doi.org/10.1016/j.jacc.2009.11.041.
Ellinor PT, Lunetta KL, Glazer NL, et al. Common variants in KCNN3 are associated with lone atrial fibrillation. Nat Genet. 2010;42:240–4. https://doi.org/10.1038/ng.537.
Ellinor PT, Lunetta KL, Albert CM, et al. Meta-analysis identifies six new susceptibility loci for atrial fibrillation. Nat Genet. 2012;44:670–5. https://doi.org/10.1038/ng.2261.
Sinner MF, Tucker NR, Lunetta KL, et al. Integrating genetic, transcriptional, and functional analyses to identify 5 novel genes for atrial fibrillation. Circulation. 2014;130:1225–35. https://doi.org/10.1161/CIRCULATIONAHA.114.009892.
Gudbjartsson DF, Holm H, Gretarsdottir S, et al. A sequence variant in ZFHX3 on 16q22 associates with atrial fibrillation and ischemic stroke. Nat Genet. 2009;41:876–8. https://doi.org/10.1038/ng.417.
Benjamin EJ, Rice KM, Arking DE, et al. Variants in ZFHX3 are associated with atrial fibrillation in individuals of European ancestry. Nat Genet. 2009;41:879–81. https://doi.org/10.1038/ng.416.
Christophersen IE, Rienstra M, Roselli C, et al. Large-scale analyses of common and rare variants identify 12 new loci associated with atrial fibrillation. Nat Genet. 2017;49:946–52. https://doi.org/10.1038/ng.3843.
• Nielsen JB, Thorolfsdottir RB, Fritsche LG, et al. Biobank-driven genomic discovery yields new insight into atrial fibrillation biology. Nat Genet. 2018;50:1234–39. PMID 30061737. https://doi.org/10.1038/s41588-018-0171-3. A large scale GWAS identifying novel AF risk variants with function analysis to suggest possible biologic role.
Tucker NR, Clauss S, Ellinor PT. Common variation in atrial fibrillation: navigating the path from genetic association to mechanism. Cardiovasc Res. 2016;109:493–501. https://doi.org/10.1093/cvr/cvv283.
Nadadur RD, Broman MT, Boukens B, et al. Pitx2 modulates a Tbx5-dependent gene regulatory network to maintain atrial rhythm. Sci Transl Med. 2016;8:354ra115. PMID 27582060. https://doi.org/10.1126/scitranslmed.aaf4891.
O’Sullivan JW, Shcherbina A, Justesen JM, Turakhia M, Perez M, Wand H, et al. Combining clinical and polygenic risk improves stroke prediction among individuals with atrial fibrillation. Circ Genomic Precis Med. 2021. https://doi.org/10.1161/CIRCGEN.120.003168.
• Khera AV, Chaffin M, Aragam KG, et al. Genome-wide polygenic scores for common diseases identify individuals with risk equivalent to monogenic mutations. Nat Genet. 2018;50:1219–24. PMID 30104762. https://doi.org/10.1038/s41588-018-0183-z. This study illustrates the clinical utility of multiple PRS including AF.
Choe WS, Kang JH, Choi EK, Shin SY, Lubitz SA, Ellinor PT, Oh S, Lim HE. A genetic risk score for atrial fibrillation predicts the response to catheter ablation. Korean Circ J. 2019;49:338–49. https://doi.org/10.4070/kcj.2018.0161.
Kertai MD, Mosley JD, He J, Ramakrishnan A, Abdelmalak M, Hong Y, et al. Predictive accuracy of a polygenic risk score for postoperative atrial fibrillation after cardiac surgery. Circ Genomic Precis Med. 2021. https://doi.org/10.1161/CIRCGEN.120.003269.
Weng LC, Preis SR, Hulme OL, et al. Genetic predisposition, clinical risk factor burden, and lifetime risk of atrial fibrillation. Circulation. 2018;137:1027–38. https://doi.org/10.1161/CIRCULATIONAHA.117.031431.
Lazarte J, Dron JS, Mcintyre AD, Allan C, Gula LJ, Tang AS, et al. l P. CJC Open. 2021. https://doi.org/10.1016/j.cjco.2021.02.001. PMID 34169254.
Gudbjartsson DF, Helgason H, Gudjonsson SA, et al. Large-scale whole-genome sequencing of the Icelandic population. Nat Genet. 2015;47:435–44. https://doi.org/10.1038/ng.3247.
Lubitz SA, Brody JA, Bihlmeyer NA, et al. Whole exome sequencing in atrial fibrillation. PLoS Genet. 1016;12:e1006284. 108. PMID 27589061. https://doi.org/10.1371/journal.pgen.1006284.
Ackerman MJ, Priori SG, Willems S, et al. HRS/EHRA expert consensus statement on the state of genetic testing for the channelopathies and cardiomyopathies this document was developed as a partnership between the Heart Rhythm Society (HRS) and the European Heart Rhythm Association (EHRA). Heart Rhythm. 2011;8:1308–39. https://doi.org/10.1016/j.hrthm.2011.05.020.
Wojcik GL, Graff M, Nishimura KK, et al. The PAGE study: how genetic diversity improves our understanding of the architecture of complex traits. 2017. bioRxiv 188094. https://doi.org/10.1101/188094.
Martin AR, Kanai M, Kamatani Y, Okada Y, Neale BM, Daly MJ. Clinical use of current polygenic risk scores may exacerbate health disparities. Nat Genet. 2019;51:584–91. https://doi.org/10.1038/s41588-019-0379-x.
Low S-K, Takahashi A, Ebana Y, et al. Identification of six new genetic loci associated with atrial fibrillation in the Japanese population. Nat Genet. 2017;49:953–8. https://doi.org/10.1038/ng.3842.
Lee J-Y, Kim T-H, Yang P-S, et al. Korean atrial fibrillation network genome-wide association study for early-onset atrial fibrillation identifies novel susceptibility loci. Eur Heart J. 2017;38:2586–94. https://doi.org/10.1093/eurheartj/ehx213.
Mostafavi H, Harpak A, Agarwal I, Conley D, Pritchard JK, Przeworski M. Variable prediction accuracy of polygenic scores within an ancestry group. Elife. 2020;9:1–52. https://doi.org/10.7554/eLife.48376.
Hu Z, Zou D. Genotype-phenotype associations in atrial fibrillation: meta-analysis. J Interv Card Electrophysiol. 2019;54:283–8. https://doi.org/10.1007/s10840-018-0484-2.
Burridge PW, Matsa E, Shukla P, et al. Chemically defined generation of human cardiomyocytes. Nat Methods. 2014;11:855–60. https://doi.org/10.1038/nmeth.2999.
Devalla HD, Schwach V, Ford JW, et al. Atrial-like cardiomyocytes from human pluripotent stem cells are a robust preclinical model for assessing atrial-selective pharmacology. EMBO Mol Med. 2015;7:394–410. https://doi.org/10.15252/emmm.201404757.
Cerignoli F, Charlot D, Whittaker R, et al. High throughput measurement of Ca2+ dynamics for drug risk assessment in human stem cell-derived cardiomyocytes by kinetic image cytometry. J Pharmacol Toxicol Methods. 2012;66:246–56. https://doi.org/10.1016/j.vascn.2012.08.167.
McKeithan WL, Savchenko A, Yu MS, Cerignoli F, Bruyneel AAN, Price JH, Colas AR, Miller EW, Cashman JR, Mercola M. An automated platform for assessment of congenital and drug-induced arrhythmia with hiPSC-derived cardiomyocytes. Front Physiol. 2017;8:766. https://doi.org/10.3389/fphys.2017.00766.
Gee KR, Brown KA, Chen WN, Bishop-Stewart J, Gray D, Johnson I. Chemical and physiological characterization of fluo-4 Ca(2+)-indicator dyes. Cell Calcium. 2000;27:97–106. https://doi.org/10.1054/ceca.1999.0095.
Liu P, Miller EW. Electrophysiology, unplugged: imaging membrane potential with fluorescent indicators. Acc Chem Res. 2020;53:11–9. https://doi.org/10.1021/acs.accounts.9b00514.
McKeithan WL, Feyen DAM, Bruyneel AAN, et al. Reengineering an antiarrhythmic drug using patient hiPSC cardiomyocytes to improve therapeutic potential and reduce toxicity. Cell Stem Cell. 2020;27:813-821.e6. https://doi.org/10.1016/j.stem.2020.08.003.
Cashman JR, Ryan D, McKeithan WL, et al. Antiarrhythmic hit to lead refinement in a dish using patient-derived iPSC cardiomyocytes. J Med Chem. 2021. https://doi.org/10.1021/acs.jmedchem.0c01545.
Bodmer R. The gene tinman is required for specification of the heart and visceral muscles in Drosophila. Development. 1993;118:719–29.
Azpiazu N, Frasch M. tinman and bagpipe: two homeo box genes that determine cell fates in the dorsal mesoderm of Drosophila. Genes Dev. 1993;7:1325–40. https://doi.org/10.1101/gad.7.7b.1325.
Cripps RM, Olson EN. Control of cardiac development by an evolutionarily conserved transcriptional network. Dev Biol. 2002;246:14–28. https://doi.org/10.1006/dbio.2002.0666.
Bodmer R, Frasch M. Chapter 1.2 - Development and Aging of the Drosophila Heart. In: Rosenthal N, Harvey RP, editors. Heart Development and Regeneration. Academic Press; 2010. p. 47–86. ISBN 9780123813329. https://doi.org/10.1016/B978-0-12-381332-9.00002-5. https://www.sciencedirect.com/science/article/pii/B9780123813329000025.
Cammarato A, Ahrens CH, Alayari NN, et al. A mighty small heart: the cardiac proteome of adult Drosophila melanogaster. PLoS One. 2011;6:e18497. PMID 21541028. https://doi.org/10.1371/journal.pone.0018497.
Ocorr K, Reeves NL, Wessells RJ, et al. KCNQ potassium channel mutations cause cardiac arrhythmias in Drosophila that mimic the effects of aging. Proc Natl Acad Sci U S A. 2007;104:3943–8. https://doi.org/10.1073/pnas.0609278104.
Ocorr K, Zambon A, Nudell Y, Pineda S, Diop S, Tang M, et al. Age-dependent electrical and morphological remodeling of the Drosophila heart caused by hERG/seizure mutations. PLoS Genet. 2017;13:e1006786. PMID 17125816. https://doi.org/10.1371/journal.pgen.1006786.
Pineda S, Nikolova-Krstevski V, Leimena C, et al. Conserved role of the large conductance calcium-activated potassium channel, KCa1.1, in Sinus node function and arrhythmia risk. Circ Genomic Precis Med. 2021;14:e003144. PMID 33629867. https://doi.org/10.1161/CIRCGEN.120.003144.
Ocorr K, Akasaka T, Bodmer R. Age-related cardiac disease model of Drosophila. Mech Ageing Dev. 2007;128:112–6. https://doi.org/10.1016/j.mad.2006.11.023.
Nishimura M, Ocorr K, Bodmer R, Cartry J. Drosophila as a model to study cardiac aging. Exp Gerontol. 2011;46:326–30. https://doi.org/10.1016/j.exger.2010.11.035.
Ocorr K, Vogler G, Bodmer R. Methods to assess Drosophila heart development, function and aging. Methods. 2014;68:265–72. https://doi.org/10.1016/j.ymeth.2014.03.031.
Na J, Musselman LP, Pendse J, Baranski TJ, Bodmer R, Ocorr K, et al. A Drosophila model of high sugar diet-induced cardiomyopathy. PLoS Genet. 2013;9:e1003175. PMID 23326243. https://doi.org/10.1371/journal.pgen.1003175.
Birse RT, Choi J, Reardon K, Rodriguez J, Graham S, Diop S, Ocorr K, Bodmer R, Oldham S. High-fat-diet-induced obesity and heart dysfunction are regulated by the TOR pathway in Drosophila. Cell Metab. 2010;12:533–44. https://doi.org/10.1016/j.cmet.2010.09.014.
Diop SB, Bisharat-Kernizan J, Birse RT, Oldham S, Ocorr K, Bodmer R. PGC-1/spargel counteracts high-fat-diet-induced obesity and cardiac lipotoxicity downstream of TOR and brummer ATGL lipase. Cell Rep. 2015;10:1572–84. https://doi.org/10.1016/j.celrep.2015.02.022.
Fink M, Callol-Massot C, Chu A, Ruiz-Lozano P, Izpisua Belmonte JC, Giles W, Bodmer R, Ocorr K. A new method for detection and quantification of heartbeat parameters in Drosophila, zebrafish, and embryonic mouse hearts. Biotechniques. 2009;46:101–13. https://doi.org/10.2144/000113078.
Vogler G. FlyHearts-tdtK-Rscripts: first release of the R tdtK script. 2021. https://doi.org/10.5281/zenodo.4749935.
Theis JL, Vogler G, Missinato MA, et al. Patient-specific genomics and cross-species functional analysis implicate LRP2 in hypoplastic left heart syndrome. Elife. 2020. https://doi.org/10.7554/eLife.59554.
Hall AW, Chaffin M, Roselli C, et al. Epigenetic analyses of human left atrial tissue identifies gene networks underlying atrial fibrillation. Circ Genomic Precis Med. 2020;588–98. PMID 33155827. https://doi.org/10.1161/CIRCGEN.120.003085.
Shiffman D, Perez MV, Bare LA, Louie JZ, Arellano AR, Devlin JJ. Genetic risk for atrial fibrillation could motivate patient adherence to warfarin therapy: a cost effectiveness analysis. BMC Cardiovasc Disord. 2015;15:104. https://doi.org/10.1186/s12872-015-0100-7.
Lubitz SA, Parsons OE, Anderson CD, et al. Atrial fibrillation genetic risk and ischemic stroke mechanisms. Stroke. 2017;48:1451–6. https://doi.org/10.1161/STROKEAHA.116.016198.
Pulit SL, Weng LC, McArdle PF, et al. Atrial fibrillation genetic risk differentiates cardioembolic stroke from other stroke subtypes. Neurol Genet. 2018;4:1–8. https://doi.org/10.1212/NXG.0000000000000293.
Christopoulos G, Graff-Radford J, Lopez CL, et al. Artificial intelligence-electrocardiography to predict incident atrial fibrillation: a population-based study. Circ Arrhythm Electrophysiol. 2020;13:e009355. PMID 33185118. https://doi.org/10.1161/CIRCEP.120.009355.
Acknowledgments
EDM is supported by UL1TR002550 from NCATS/NIH to The Scripps Research Institute. Dr. Ocorr has a patent US 9,186,093 B2 for the Semi-automatic Optical Heartbeat Analysis, licensed to None, available for free to researchers at www.sohasoftware.com.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Evan D. Muse is a co-founder of GeneXwell. The other authors declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any data with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Cardiovascular Genomics
Rights and permissions
About this article
Cite this article
Yoo, D.H., Bodmer, R., Ocorr, K. et al. Atrial Fibrillation Genomics: Discovery and Translation. Curr Cardiol Rep 23, 164 (2021). https://doi.org/10.1007/s11886-021-01597-x
Accepted:
Published:
DOI: https://doi.org/10.1007/s11886-021-01597-x