Abstract
Purpose of Review
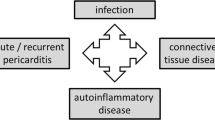
This review article aims to provide a contemporary insight into the pathophysiological mechanisms of and therapeutic targets for pericarditis, drawing distinction between autoinflammatory and autoimmune pericarditis.
Recent Findings
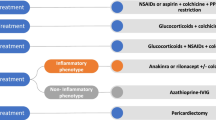
Recent research has focused on the distinction between autoinflammatory and autoimmune pericarditis. In autoinflammatory pericarditis, viruses can activate the sensor molecule of the inflammasome, which results in downstream release of cytokines, such as interleukin-1, that recruit neutrophils and macrophages to the site of injury. Conversely, in autoimmune pericarditis, a type I interferon signature predominates, and pericardial manifestations coincide with the severity of the underlying systemic autoimmune disease. In addition, autoimmune pericarditis can also develop after cardiac injury syndromes. With either type of pericarditis, imaging can help stage the inflammatory state. Prominent pericardial delayed hyperenhancement on magnetic resonance imaging suggests ongoing inflammation whereas calcium on computed tomography suggests a completed inflammatory cascade. In patients with ongoing pericarditis, treatments that converge on the inflammasome, such as colchicine and anakinra, have proved effective in recurrent autoinflammatory pericarditis, though further clinical trials with anakinra are warranted.
Summary
An improved understanding of the pathophysiological mechanisms of pericarditis helps unravel effective therapeutic targets for this condition.




Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Cosyns B, Plein S, Nihoyanopoulos P, Smiseth O, Achenbach S, Andrade MJ, et al. European Association of Cardiovascular Imaging (EACVI) position paper: multimodality imaging in pericardial disease. Eur Heart J Cardiovasc Imaging. 2015;16:12–31. doi:10.1093/ehjci/jeu128.
•• Klein AL, Abbara S, Agler DA, Appleton CP, Asher CR, Hoit B, et al. American society of echocardiography clinical recommendations for multimodality cardiovascular imaging of patients with pericardial disease: endorsed by the society for cardiovascular magnetic resonance and society of cardiovascular computed tomography. J Am Soc Echocardiogr. 2013;26:965–1012.e15. doi:10.1016/j.echo.2013.06.023. An important guideline document on the management of pericardial conditions, with a focus on multimodality cardiovascular imaging assessment
•• Adler Y, Charron P, Imazio M, Badano L, Baron-Esquivias G, Bogaert J, et al. 2015 ESC guidelines for the diagnosis and management of pericardial diseases. Eur Heart J. 2015;36:2921–64. doi:10.1093/eurheartj/ehv318. An important guideline document on pericardial diseases published by the European Society of Cardiology
Imazio M. Idiopathic recurrent pericarditis as an immune-mediated disease: current insights into pathogenesis and emerging treatment options. Expert Rev Clin Immunol. 2014;10:1487–92. doi:10.1586/1744666X.2014.965150.
Cusick MF, Libbey JE, Fujinami RS. Molecular mimicry as a mechanism of autoimmune disease. Clin Rev Allergy Immunol. 2012;42:102–11. doi:10.1007/s12016-011-8294-7.
Imazio M. Pericardial involvement in systemic inflammatory diseases. Heart. 2011;97:1882–92. doi:10.1136/heartjnl-2011-300054.
•• Cremer PC, Kumar A, Kontzias A, Tan CD, Rodriguez ER, Imazio M, et al. Complicated pericarditis understanding risk factors and pathophysiology to inform imaging and treatment. J Am Coll Cardiol. 2016;68:2311–28. doi:10.1016/j.jacc.2016.07.785. An important contemporary review of pericarditis with a focus on multimodality imaging assessment and pathophysiology
Muruve DA, Petrilli V, Zaiss AK, White LR, Clark SA, Ross PJ, et al. The inflammasome recognizes cytosolic microbial and host DNA and triggers an innate immune response. Nature. 2008;452:103–7.
Martinon F, Petrilli V, Mayor A, Tardivel A, Tschopp J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature. 2006;440:237–41.
Duewell P, Kono H, Rayner KJ, Sirois CM, Vladimer G, Bauernfeind FG, et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature. 2010;464:1357–61.
•• Brucato A, Imazio M, Gattorno M, Lazaros G, Maestroni S, Carraro M, et al. Effect of anakinra on recurrent pericarditis among patients with colchicine resistance and corticosteroid dependence. JAMA. 2016;316:1906. doi:10.1001/jama.2016.15826. The first published randomized trial evidence examining the efficacy of anakinra in recurrent pericarditis in a small number patients
van Kempen TS, Wenink MH, Leijten EFA, Radstake TRDJ, Boes M. Perception of self: distinguishing autoimmunity from autoinflammation. Nat Rev Rheumatol. 2015;11:483–92.
Prasad M, Hermann J, Gabriel SE, Weyand CM, Mulvagh S, Mankad R, et al. Cardiorheumatology: cardiac involvement in systemic rheumatic disease. Nat Rev Cardiol. 2015;12:168–76. doi:10.1038/nrcardio.2014.206.Cardiorheumatology.
Voskuyl AE. The heart and cardiovascular manifestations in rheumatoid arthritis. Rheumatology. 2006;45:4–7. doi:10.1093/rheumatology/kel313.
Miner JJ, Kim AHJ. Cardiac manifestations of systemic lupus erythematosus. Rheum Dis Clin. 2014;40:51–60. doi:10.1016/j.rdc.2013.10.003.
Imazio M, Hoit BD. Post-cardiac injury syndromes. An emerging cause of pericardial diseases. Int J Cardiol. 2013;168:648–52. doi:10.1016/j.ijcard.2012.09.052.
Cevik C, Wilborn T, Corona R, Schanzmeyer E, Nugent K. Post-cardiac injury syndrome following transvenous pacemaker insertion: a case report and review of the literature. Hear Lung Circ. 2009;18:379–83. doi:10.1016/j.hlc.2009.04.001.
Alraies MC, Al Jaroudi W, Shabrang C, Yarmohammadi H, Klein AL, Tamarappoo BK. Clinical features associated with adverse events in patients with post-pericardiotomy syndrome following cardiac surgery. Am J Cardiol. 2014;114:1426–30. doi:10.1016/j.amjcard.2014.07.078.
Imazio M, Spodick DH, Brucato A, Trinchero R, Adler Y. Controversial issues in the management of pericardial diseases. Circulation. 2010;121:916–28. doi:10.1161/CIRCULATIONAHA.108.844753.
Khan AH. The postcardiac injury syndromes. Clin Cardiol. 1992;15:67–72. doi:10.1002/clc.4960150203.
Hoffman M, Fried M, Jabareen F, Vardinon N, Turner D, Burke M, et al. Anti-heart antibodies in Postpericardiotomy syndrome: cause or epiphenomenon? Autoimmunity. 2002;35:241–5. doi:10.1080/08916930290028166.
Douglas PS, Garcia MJ, Haines DE, Lai WW, Ragosta M, Ward RP, et al. ACCF/ASE/AHA/ASNC/HFSA/HRS/SCAI/SCCM/SCCT/SCMR 2011 appropriate use criteria for echocardiography. J Am Soc Echocardiogr. 2011;24:229–67. doi:10.1016/j.echo.2010.12.008.
Zurick AO, Bolen MA, Kwon DH, Tan CD, Popovic ZB, Rajeswaran J, et al. Pericardial delayed hyperenhancement with CMR imaging in patients with constrictive pericarditis undergoing surgical pericardiectomy: a case series with histopathological correlation. JACC Cardiovasc Imaging. 2011;4:1180–91. doi:10.1016/j.jcmg.2011.08.011.
Alraies MC, AlJaroudi W, Yarmohammadi H, Yingchoncharoen T, Schuster A, Senapati A, et al. Usefulness of cardiac magnetic resonance-guided management in patients with recurrent pericarditis. Am J Cardiol. 2015;115:542–7. doi:10.1016/j.amjcard.2014.11.041.
Cremer PC, Tariq MU, Karwa A, Alraies MC, Benatti R, Schuster A, et al. Quantitative assessment of pericardial delayed hyperenhancement predicts clinical improvement in patients with constrictive pericarditis treated with anti-inflammatory therapy. Circ Cardiovasc Imaging. 2015;8:1–8. doi:10.1161/CIRCIMAGING.114.003125.
Bogaert J, Francone M. Pericardial disease: value of CT and MR imaging. Radiology. 2013;267:340–56. doi:10.1148/radiol.13121059.
Haley JH, Tajik AJ, Danielson GK, Schaff HV, Mulvagh SL, Oh JK. Transient constrictive pericarditis: causes and natural history. J Am Coll Cardiol. 2004;43:271–5. doi:10.1016/j.jacc.2003.08.032.
Gentry J, Klein AL, Jellis C. Transient constrictive pericarditis: current diagnostic and therapeutic strategies. Curr Cardiol Rep. 2016;18 doi:10.1007/s11886-016-0720-2.
Imazio M, Brucato A, Cemin R, Ferrua S, Maggiolini S, Beqaraj F, et al. A randomized trial of colchicine for acute pericarditis. N Engl J Med. 2013;369:1522–8. doi:10.1056/NEJMoa1208536.
Imazio M, Bobbio M, Cecchi E, Demarie D, Pomari F, Moratti M, et al. Colchicine as first-choice therapy for recurrent pericarditis. Arch Intern Med. 2005;165:1987–91.
Horneffer P, Miller R, Pearson T, Rykiel M, Reitz B, Gardner T. The effective treatment of postpericardiotomy syndrome after cardiac operations. A randomized placebo-controlled trial. J Thorac Cardiovasc Surg. 1990;100:292–6.
Imazio M, Gaita F. Diagnosis and treatment of pericarditis. Heart. 2015;101:1159–68. doi:10.1136/heartjnl-2014-306362.
Imazio M, Brucato A, Maestroni S, Cumetti D, Dominelli A, Natale G, et al. Prevalence of C-reactive protein elevation and time course of normalization in acute pericarditis: implications for the diagnosis, therapy, and prognosis of pericarditis. Circulation. 2011;123:1092–7. doi:10.1161/CIRCULATIONAHA.110.986372.
Imazio M, Brucato A, Cemin R, Ferrua S, Belli R, Maestroni S, et al. Colchicine for Recurrent Pericarditis (CORP) a randomized trial. Ann Intern Med. 2011;155:409–14. doi:10.1111/trf.13134.
Imazio M, Bobbio M, Cecchi E, Demarie D, Demichelis B, Pomari F, et al. Colchicine in addition to conventional therapy for acute pericarditis: results of the COlchicine for acute PEricarditis (COPE) trial. Circulation. 2005;112:2012–6. doi:10.1161/CIRCULATIONAHA.105.542738.
Leung YY, Yao Hui LL, Kraus VB. Colchicine—update on mechanisms of action and therapeutic uses. Semin Arthritis Rheum. 2015;45:341–50. doi:10.1016/j.semarthrit.2015.06.013.
Verma S, Eikelboom JW, Nidorf SM, Al-Omran M, Gupta N, Teoh H, et al. Colchicine in cardiac disease: a systematic review and meta-analysis of randomized controlled trials. BMC Cardiovasc Disord. 2015;15:96. doi:10.1186/s12872-015-0068-3.
Imazio M, Brucato A, Cemin R, Ferrua S, Belli R, Maestroni S, et al. Colchicine for Recurrent Pericarditis (CORP) a randomized trial. Ann Intern Med. 2011:409–14. doi:10.1378/chest.13-0162.
Imazio M, Belli R, Brucato A, Cemin R, Ferrua S, Beqaraj F, et al. Efficacy and safety of colchicine for treatment of multiple recurrences of pericarditis (CORP-2): a multicentre, double-blind, placebo-controlled, randomised trial. Lancet. 2014;383:2232–7. doi:10.1016/S0140-6736(13)62709-9.
Rhen T, Cidlowski JA. Antiinflammatory action of glucocorticoids—new mechanisms for old drugs. N Engl J Med. 2005;353:1711–23. doi:10.1056/NEJMra050541.
Imazio M, Adler Y. Treatment with aspirin, NSAID, corticosteroids, and colchicine in acute and recurrent pericarditis. Heart Fail Rev. 2013;18:355–60. doi:10.1007/s10741-012-9328-9.
Artom G, Koren-Morag N, Spodick DH, Brucato A, Guindo J, Bayes-De-Luna A, et al. Pretreatment with corticosteroids attenuates the efficacy of colchicine in preventing recurrent pericarditis: a multi-centre all-case analysis. Eur Heart J. 2005;26:723–7. doi:10.1093/eurheartj/ehi197.
Imazio M, Brucato A, Cumetti D, Brambilla G, Demichelis B, Ferro S, et al. Corticosteroids for recurrent pericarditis high versus low doses: a nonrandomized observation. Circulation. 2008;118:667–71. doi:10.1161/CIRCULATIONAHA.107.761064.
Imazio M, Brucato A, Trinchero R, Shabetai R, Spodick D, Adler Y. Corticosteroid therapy for pericarditis: a double-edged sword. Nat Clin Pr Cardiovasc Med. 2008;5:118–9.
Elion GB. The purine path to chemotherapy. Science. 1989;244:41–7.
Sandborn W. A review of immune modifier therapy for inflammatory bowel disease: azathioprine, 6-mercaptopurine, cyclosporine, and methotrexate. Am J Gastroenterol. 1996;91:423–33.
Vianello F, Cinetto F, Cavraro M, Battisti A, Castelli M, Imbergamo S, et al. Azathioprine in isolated recurrent pericarditis: a single centre experience. Int J Cardiol. 2011;147:477–8. doi:10.1016/j.ijcard.2011.01.027.
Eftimov F, Winer JB, Vermeulen M, de Haan R, van Schaik IN. Intravenous immunoglobulin for chronic inflammatory demyelinating polyradiculoneuropathy. Cochrane Database Syst Rev. 2013; doi:10.1002/14651858.CD001797.pub3.
Hughes RAC, Swan AV, van Doorn PA. Intravenous immunoglobulin for Guillain-Barré syndrome. Cochrane Database Syst Rev. 2014; doi:10.1002/14651858.CD002063.pub6.
Salib M, Clayden R, Clare R, Wang G, Warkentin TE, Crowther MA, et al. Difficulties in establishing the diagnosis of immune thrombocytopenia: an agreement study. Am J Hematol. 2016;91:E327–9. doi:10.1002/ajh.24404.
Kaneko Y, Nimmerjahn F, Ravetch JV. Anti-inflammatory activity of immunoglobulin G resulting from Fc sialylation. Science. 2006;313:670–3.
Samuelsson A, Towers TL, Ravetch JV. Anti-inflammatory activity of IVIG mediated through the inhibitory Fc receptor. Science. 2001;291:484–6.
Imazio M, Lazaros G, Picardi E, Vasileiou P, Carraro M, Tousoulis D, et al. Intravenous human immunoglobulins for refractory recurrent pericarditis: a systematic review of all published cases. J Cardiovasc Med. 2016;17:263–9.
Moretti M, Buiatti A, Merlo M, Massa L, Fabris E, Pinamonti B, et al. Usefulness of high-dose intravenous human immunoglobulins treatment for refractory recurrent pericarditis. Am J Cardiol. 2013;112:1493–8. doi:10.1016/j.amjcard.2013.06.036.
Dinarello CA, Wolff SM. The role of interleukin-1 in disease. N Engl J Med. 1993;328:106–13. doi:10.1056/NEJM199301143280207.
Dinarello CA, Simon A, van der Meer JWM. Treating inflammation by blocking interleukin-1 in a broad spectrum of diseases. Nat Rev Drug Discov. 2012;11:633–52.
Zeft A, Spalding S. Autoinflammatory syndromes: fever is not always a sign of infection. Cleve Clin J Med. 2012;79:569–81.
Bresnihan B, Alvaro-Gracia JM, Cobby M, Doherty M, Domljan Z, Emery P, et al. Treatment of rheumatoid arthritis with recombinant human interleukin-1 receptor antagonist. Arthritis Rheum. 1998;41:2196–204. doi:10.1002/1529-0131(199812)41:12<2196::AID-ART15>3.0.CO;2-2.
Simon A, Bodar E, van der Hilst JC, van der Meer JW, Fiselier TJ, Cuppen MP, et al. Beneficial response to interleukin 1 receptor antagonist in traps. Am J Med. 2004;117:208–10.
So A, de Smedt T, Revaz S, Tschopp J. Research article a pilot study of IL-1 inhibition by anakinra in acute gout. Arthritis Res Ther. 2007;9:1–6. doi:10.1186/ar2143.
Zeft A, Hollister R, LaFleur B, Sampath P, Soep J, McNally B, et al. Anakinra for systemic juvenile arthritis: the Rocky Mountain experience. JCR J Clin Rheumatol. 2009;15:161–4.
Jain S, Thongprayoon C, Espinosa RE, Hayes SN, Klarich KW, Cooper LT, et al. Effectiveness and safety of anakinra for management of refractory pericarditis. Am J Cardiol. 2015;116:1277–9. doi:10.1016/j.amjcard.2015.07.047.
Lazaros G, Vasileiou P, Koutsianas C, Antonatou K, Stefanadis C, Pectasides D, et al. Anakinra for the management of resistant idiopathic recurrent pericarditis. Initial experience in 10 adult cases. Ann Rheum Dis. 2014; doi:10.1136/annrheumdis-2014-205990.
Khandaker MH, Schaff HV, Greason KL, Anavekar NS, Espinosa RE, Hayes SN, et al. Pericardiectomy vs medical management in patients with relapsing pericarditis. Mayo Clin Proc. 2017;87:1062–70. doi:10.1016/j.mayocp.2012.05.024.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Bo Xu, Serge C. Harb, and Paul C. Cremer declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Pericardial Disease
Rights and permissions
About this article
Cite this article
Xu, B., Harb, S.C. & Cremer, P.C. New Insights into Pericarditis: Mechanisms of Injury and Therapeutic Targets. Curr Cardiol Rep 19, 60 (2017). https://doi.org/10.1007/s11886-017-0866-6
Published:
DOI: https://doi.org/10.1007/s11886-017-0866-6