Abstract
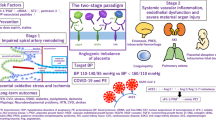
Preeclampsia is the major cause of maternal and fetal morbidity and mortality, involving 15% to 20% of pregnancies in developed countries and even more in less developed parts of the world. Superficial placentation driven by immune maladaptation, with subsequently reduced concentrations of angiogenic growth factors and increased placental debris in the maternal circulation, are likely responsible. Recent advances suggest that antiangiogenic factors (soluble fms-like tyrosine receptor kinase and soluble endoglin), altered relaxin-mediated mechanisms leading to impaired nitric oxide production through asymmetrical dimethylarginine production, and activating antibodies directed at the angiotensin II type 1 receptor may be responsible. The field of preeclampsia research is enjoying a well-deserved blossoming of novel ideas and approaches. We hope the activity will lead to much earlier diagnostic capacities and novel prophylactic treatments. The prize will go to the affected women and their afflicted children. For the investigators in the area, such a prize would be welcome.
Similar content being viewed by others
References and Recommended Reading
Sibai B, Dekker G, Kupferminc M: Pre-eclampsia. Lancet 2005, 365:785–799.
Poston L, Briley AL, Seed PT, et al.: Vitamin C and vitamin E in pregnant women at risk for pre-eclampsia (VIP trial): randomised placebo-controlled trial. Vitamins in Pre-eclampsia (VIP) Trial Consortium. Lancet 2006, 367:1145–1154.
Askie LM, Duley L, Henderson-Smart DJ, Stewart LA: Antiplatelet agents for prevention of pre-eclampsia: a meta-analysis of individual patient data. PARIS Collaborative Group. Lancet 2007, 369:1791–1798.
Altman D, Carroli G, Duley L, et al.: Do women with pre-eclampsia, and their babies, benefit from magnesium sulphate? The Magpie Trial: a randomised placebo-controlled trial. Magpie Trial Collaboration Group. Lancet 2002, 359:1877–1890.
Maynard SE, Min JY, Merchan J, et al.: Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J Clin Invest 2003, 111:649–658.
Levine RJ, Maynard SE, Qian C, et al.: Circulating angiogenic factors and the risk of preeclampsia. N Engl J Med 2004, 350:672–683.
Levine RJ, Thadhani R, Qian C, et al.: Urinary placental growth factor and risk of preeclampsia. JAMA 2005, 293:77–85.
Venkatesha S, Toporsian M, Lam C, et al.: Soluble endoglin contributes to the pathogenesis of preeclampsia. Nat Med 2006, 12:642–649.
Levine RJ, Lam C, Qian C, et al.: Soluble endoglin and other circulating antiangiogenic factors in preeclampsia. CPEP Study Group. N Engl J Med 2006, 355:992–1005.
Verheul HM, Pinedo HM: Possible molecular mechanisms involved in the toxicity of angiogenesis inhibition. Nat Rev Cancer 2007, 7:475–485.
Eremina V, Jefferson JA, Kowalewska J, et al.: VEGF inhibition and renal thrombotic microangiopathy. N Engl J Med 2008, 358:1129–1136.
Chapman AB, Abraham WT, Zamudio S, et al.: Temporal relationships between hormonal and hemodynamic changes in early human pregnancy. Kidney Int 1998, 54:2056–2063.
Groenendijk R, Trimbos JB, Wallenburg HC: Hemodynamic measurements in preeclampsia: preliminary observations. Am J Obstet Gynecol 1984, 150:232–236.
Ma S, Gundlach AL: Relaxin-family peptide and receptor systems in brain: insights from recent anatomical and functional studies. Adv Exp Med Biol 2007, 612:119–137.
Jeyabalan A, Shroff SG, Novak J, Conrad KP: The vascular actions of relaxin. Adv Exp Med Biol 2007, 612:65–87.
Novak J, Danielson LA, Kerchner LJ, et al.: Relaxin is essential for renal vasodilation during pregnancy in conscious rats. J Clin Invest 2001, 107:1469–1475.
Jeyabalan A, Novak J, Danielson LA, et al.: Essential role for vascular gelatinase activity in relaxin-induced renal vasodilation, hyperfiltration, and reduced myogenic reactivity of small arteries. Circ Res 2003, 93:1249–1257.
Carbillon L, Uzan M, Uzan S: Pregnancy, vascular tone, and maternal hemodynamics: a crucial adaptation. Obstet Gynecol Surv 2000, 55:574–581.
Savvidou MD, Hingorani AD, Tsikas D, et al.: Endothelial dysfunction and raised plasma concentrations of asymmetric dimethylarginine in pregnant women who subsequently develop pre-eclampsia. Lancet 2003, 361:1511–1517.
Wallukat G, Homuth V, Fischer T, et al.: Patients with preeclampsia develop agonistic autoantibodies against the angiotensin AT1 receptor. J Clin Invest 1999, 103:945–952.
Fu ML, Herlitz H, Schulze W, et al.: Autoantibodies against the angiotensin receptor (AT1) in patients with hypertension. J Hypertens 2000, 18:945–953.
Dechend R, Viedt C, Muller DN, et al.: AT1 receptor agonistic antibodies from preeclamptic patients stimulate NADPH oxidase. Circulation 2003, 107:1632–1639.
Bohlender J, Ganten D, Luft FC: Rats transgenic for human renin and human angiotensinogen as a model for gestational hypertension. J Am Soc Nephrol 2000, 11:2056–2061.
Dechend R, Gratze P, Wallukat G, et al.: Agonistic autoantibodies to the AT1 receptor in a transgenic rat model of preeclampsia. Hypertension 2005, 45:742–746.
Xia Y, Wen H, Bobst S, et al.: Maternal autoantibodies from preeclamptic patients activate angiotensin receptors on human trophoblast cells. J Soc Gynecol Investig 2003, 10:82–93.
Thway TM, Shlykov SG, Day MC, et al.: Antibodies from preeclamptic patients stimulate increased intracellular Ca2+ mobilization through angiotensin receptor activation. Circulation 2004, 110:1612–1619.
Bobst SM, Day MC, Gilstrap LC 3rd, et al.: Maternal autoantibodies from preeclamptic patients activate angiotensin receptors on human mesangial cells and induce interleukin-6 and plasminogen activator inhibitor-1 secretion. Am J Hypertens 2005, 18:330–336.
Zhou CC, Ahmad S, Mi T, et al.: Angiotensin II induces soluble fms-like tyrosine kinase-1 release via calcineurin signaling pathway in pregnancy. Circ Res 2007, 100:88–95.
Zhou CC, Ahmad S, Mi T, et al.: Autoantibody from women with preeclampsia induces soluble Fms-like tyrosine kinase-1 production via angiotensin type 1 receptor and calcineurin/nuclear factor of activated T-cells signaling. Hypertension 2008, 51:991–992.
Hubel CA, Wallukat G, Wolf M, et al.: Agonistic angiotensin II type 1 receptor autoantibodies in postpartum women with a history of preeclampsia. Hypertension 2007, 49:612–617.
Herse F, Dechend R, Harsem NK, et al.: Dysregulation of the circulating and tissue-based renin-angiotensin system in preeclampsia. Hypertension 2007, 49:604–611.
Walther T, Wallukat G, Jank A, et al.: Angiotensin II type 1 receptor agonistic antibodies reflect fundamental alterations in the uteroplacental vasculature. Hypertension 2005, 46:1275–1279.
Stepan H, Faber R, Wessel N, et al.: Relation between circulating angiotensin II type 1 receptor agonistic autoantibodies and soluble fms-like tyrosine kinase 1 in the pathogenesis of preeclampsia. J Clin Endocrinol Metab 2006, 91:2424–2427.
Stepan H, Wallukat G, Schultheiss HP, et al.: Is parvovirus B19 the cause for autoimmunity against the angiotensin II type receptor? J Reprod Immunol 2007, 73:130–134.
Dragun D, Muller DN, Brasen JH, et al.: Angiotensin II type 1-receptor activating antibodies in renal-allograft rejection. N Engl J Med 2005, 352:558–569.
Zhou CC, Zhang Y, Irani RA, et al.: Angiotensin receptor agonistic autoantibodies induce pre-eclampsia in pregnant mice. Nat Med 2008, 14:855–862.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dechend, R., Luft, F.C. Are we getting closer to a Nobel Prize for unraveling preeclampsia?. Curr Cardiol Rep 10, 440–447 (2008). https://doi.org/10.1007/s11886-008-0070-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11886-008-0070-9