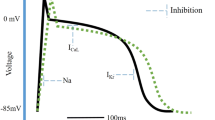
Abstract
This review on antiarrhythmic drugs traces the evolution of the fundamental mechanisms of action of drugs that have been used to control disorders of cardiac rhythm. It describes the very earliest data from experimental studies that dealt with the effects of acute and chronic administration of drugs in whole animals combined with the measurements of the action potential duration and the effective refractory period in isolated tissues. Antiarrhythmic drugs were found to have properties consistent with the block of fast sodium channel conduction, adrenergic blockade, repolarization block, and the block of slow-channel mediated conduction especially in the atrioventricular node. Over the past 15 years, the attention has focused on atrial tissue with atrial fibrillation emerging as the most common arrhythmia in clinical practice. Druginduced increases in refractoriness as a function rate and in wavelength (product of refractoriness and conduction velocity), and a reduction in numbers of wavelets have been found to be critical in the conversion of atrial fibrillation and maintenance of sinus rhythm. The continued development of newer pharmacologic agents is likely to lead to the resolution of the controversy regarding rhythm versus rate control in various clinical subsets of the arrhythmia by controlled clinical trials.
Similar content being viewed by others
References and Recommended Reading
Singh BN: Current antiarrhythmic drugs: an overview of mechanisms of action and potential clinical utility. J Cardiovasc Electrophysiol 1999, 10:283–301.
Feld GK: Evolution of diagnostic and interventional cardiac electrophysiology: a brief historical review. Am J Cardiol 1999, 84:115R-124R.
Marchlinski FE: Adenosine as an antiarrhythmic agent. Am J Cardiol 1997, 12A:30–37.
Ho DS, Zecchin RP, Richards DA, et al.: Double-blind trial of lignocaine versus sotalol for acute termination of spontaneous sustained ventricular tachycardia. Lancet 1994, 344:18–23. This randomized, double-blind study comparing the efficacy of intravenous sotalol and intravenous lidocaine is the only one of its kind for terminating sustained VT. The message is decisive despite the small sample size; there was a highly statistically significant difference in the efficacy of lidocaine in terminating (18%) VT compared with intravenous sotalol (69%).
Singh BN: Antiarrhythmic action of dl-sotalol in ventricular and supraventricular arrhythmias. J Cardiovasc Pharmacol 1992, 2:590–604.
Movsowitz C, Marchlinski FE: Interactions between implantable cardioverter-defibrillators and class III antiarrhythmic drugs. Am J Cardiol 1998, 82:411.
The Cardiac Arrhythmia Suppression Trial II Investigators: The Cardiac Arrhythmia Suppression Trial (CAST) Investigators. Effect of encainide and flecainide on mortality in a randomized trial of arrhythmia suppression after myocardial infarction. N Engl J Med 1989, 321:406–412, 227–233.
Guidelines 2000 for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care: An international consensus on science. 6. Advanced cardiovascular life support. 5. Pharmacology 1: Agents for arrhythmias. Circulation 2000, 102(Suppl 1):112–128.
Singh BN, Vaughan Williams EM: A third class of anti-arrhythmic action. Effects on atrial and ventricular intracellular potentials, and other pharmacological actions on cardiac muscle, of MJ dy1999 and AH 3474. Br J Pharmacol 1970, 39:675–687. The first experimental study, along with Singh and Vaugh Williams [10], that raised the possibility that prolongation of repolarization per se is a distinct antiarrhythmic and antifibrillatory mechanism. The two compounds have formed the basis for synthesis and development of pure class III agents.
Singh BN, Vaughan Williams EM: The effect of amiodarone, a new antianginal drug, cardiac muscle. Br J Pharmacol 1970, 39:657–667.
Singh BN: Pharmacological Actions of Certain Cardiac Drugs and Hormones: Focus on Antiarrhythmic Mechanisms. D. Phil. Thesis. Oxford: Hertford College & University of Oxford; 1971, and Mt. Kisco, NY: Futura Publishing; 1991:1–98.
Vaughan Williams EM: Classification of Antiarrhythmic Drugs. Edited by Sandoe E, Flenstedt-Johnson E, Olesen KH. Symposium on Cardiac Arrhythmias. Sodertalje, Sweden: AB Astra; 1970:440–469.
Singh BN, Vaughan Williams EM: A fourth class of antidysrhythmic action? Effect of verapamil on ouabain toxicity, on atrial and ventricular intracellular potentials, and on other features of cardiac function. Cardiovasc Res 1972, 6:109–119.
Singh BN, Hauswirth O: Comparative mechanisms of action of antiarrhythmic drugs. Am Heart J 1974, 87:367–382.
Singh BN, Williams EM: Effect of altering potassium concentration on the action of lidocaine and diphenylhydantoin on rabbit atrial and ventricular muscle. Circ Res 1971, 29:286–295.
Nattel S, Singh BN: Evolution, mechanisms, and classification of antiarrhythmic drugs: focus on Class III actions. Am J Cardiol 1999, 84(9A):11–19.
Hauswirth O, Singh BN: Ionic mechanisms in heart muscle in relationship to the genesis and the pharmacological control of cardiac arrhythmias. Pharmacol Rev 1979, 30:5–63.
Hoffman BF, Bigger JTJr.: Antiarrhythmic drugs. In Drill’s Pharmacology in Medicine, edn 4. Edited by DiPalma JR. New York: McGraw-Hill Book; 1971:824–852.
Harrison DC: Is there a rational basis for the modified classification of antiarrhythmic drugs ? In Cardiac Arrhythmias: New Therapeutic Drugs and Devices. Edited by Morganroth J, Moore EN. Boston: Martinus Nijhoff; 1985:36–48.
Campbell TJ: Kinetics of onset of rate-dependent effects of class I antiarrhythmic drugs are important in determining their effects on refractoriness in guinea-pig ventricle, and provide a theoretical basis for their subclassification. Cardiovasc Res 1983, 17:344–352.
The Task Force of the Working Group on Arrhythmias of the European Society of Cardiology: The Sicilian gambit. A new approach to the classification of antiarrhythmic drugs based on their actions on arrhythmogenic mechanisms. Circulation 1991, 84:1831–1851.
Singh BN: The coming of age of class III antiarrhythmic principle: retrospective and future trends. Am J Cardiol 1996, 78(Suppl 4a):17–27.
Lazzara R: From first class to third class: Recent upheaval in antiarrhythmic therapy-lessons from clinical trials. Am J Cardiol 1996, 78:28–33.
Singh BN: Expanding indications for the use of class III antiarrhythmic agents in patients at high risk for sudden death. J Cardiovasc Electrophysiol 1995, 6:887–900.
Singh BN, Vaghaiwalla Mody F, Lopez B, Sarma JSM: Antiarrhythmic agents for atrial fibrillation: focus on prolong atrial repolarization. Am J Cardiol 1999, 84:161R-173R.
Jais P, Haissaguerre M, Shah DC: A focal source of atrial fibrillation treated by discrete radiofrequency ablation. Circulation 1997, 95:572–576.
Haissaguerre M, Jais P, Shah DC, et al.: Spontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins. N Engl J Med 1998, 339:659–666.
Moe GK, Abildskov JA: Atrial fibrillation as a self-sustaining arrhythmia independent of focal discharge. Am Heart J 1959, 58:59–70. This, along with the paper by Moe [29] and Moe et al. [30], is a critical report of the best exposition of the mechanism of AF, an understanding of which has led to the rational basis of pharmacologic therapy of AF.
Moe GK: On the multiple wavelet hypothesis of atrial fibrillation. Arch Int Pharmacodyn Ther 1962, 140:183–188.
Moe GK, Rheinboldt WC, Abildskov JA: A computer model of atrial fibrillation. Am Heart J 1964, 67:200–220.
Allessie MA, Lammers WJEP, Bonke FIM, Hollen J: Experimental evaluation of Moe’s multiple wavelet hypothesis of atrial fibrillation. In Cardiac Electrophysiology and Arrhythmias. Edited by Zipes DP, Jalife J. Orlando: Grune & Stratton; 1985:265–276.
Jung F, DiMarco JP: Treatment strategies for atrial fibrillation. Am J Med 1998, 104:272–286.
Ortiz J, Niwano S, Abe H, et al.: Mapping the conversion of atrial flutter to atrial fibrillation and atrial fibrillation to atrial flutter-insights into mechanism. Circ Res 1994, 74:882–894.
Prystowsky EN, Benson D, Woodrow M, et al.: Management of patients with atrial fibrillation: a statement for healthcare professionals from the Subcommittee on Electrocardiography and Electrophysiology, American Heart Association. Circulation 1996, 93:1262–1277.
Le Heuzy J-Y, Boutjdir M, Gagey S, et al.: Cellular aspects of atrial vulnerability. In The Atrium in Health and Disease. Edited by Attuel P, Coumel P, Janse MJ. Mt Kisco, NY: Futura Publishing; 1989:81–94. Although not a recent publication, this still remains one of the most compelling for the series of observations in experimental animals and tissues from the human atria in the study of factors that play the crucial in the genesis of AF in humans. It provides an excellent background to rational pharmacologic control of AF and atrial flutter.
Hondeghem LM, Snyders DJ: Class III anti-arrhythmic agents have a lot of potential but a long way to go: reduced effectiveness and dangers of reverse use dependence. Circulation 1990, 81:686–694.
Wang Z, Pelletier LC, Talajic M, Nattel S: Effects of flecainide and quinidine on human atrial potentials: role of rate dependence and comparison with guinea pig, rabbit and dog tissues. Circulation 1990, 82:274–283. An elegant and compelling study that clearly demonstrated that class IC agents and especially flecainide possessed an unusual electrophysiologic effects in mammalian tissues as a function of rate. The APD duration and refractoriness increased as the of frequency of stimulation was increased. This was in sharp contrast to the effects of the usual class III agents the effects of which dwindle as the rate is increase. These observations paved the way for the search for atrialspecific antifibrillatory compounds.
Camm AJ, Karam R, Pratt CM: The azimilide post-infarct survival evaluation (ALIVE) trial. Am Cardiol 1998, 81:35D-39D.
Pritchett E, Page, R, Connolly S, Marcello S: Azimilide treatment of atrial fibrillation. Circulation 1998, 98:633.
Maisel WH, Rawn JD, Stevenson WG: Atrial fibrillation after cardiac surgery. Ann Inern Med 2001, 135:1061–1073. An outstanding current review of the risk factors predisposing to the development of AF in setting of postoperative cardiac surgery, its incidence, elective and prophylactic control, and impact on hospitalization. The report is a compelling one for mounting an important prophylactic clinical trial.
Sun W, Sarma JSM, Singh BN: Electrophysiologic effects of dronedarone (SR33589), o non-diodinate benzfuran derivative, in the rabbit heart. Comparison with amiodarone. Circulation 1999, 100:2276–2283. The initial description of the basic electrophysiologic properties of a promising deiodinated congener of amiodarone. Its structure suggests it is not likely to have the complexity of side effects of the parent compound. Preliminary data indicate that it is effective in maintaining sinus rhythm in patients with AF.
Sun W, Sarma JSM, Singh BN: Chronic and acute effects of dronedarone in the action potential duration of rabbit atrial muscle preparations: comparison with amiodarone. J Cardiovasc Pharmacol 2002, 39:677–684.
Hohnloser, SH, Singh BN: Proarrhythmia with class III antiarrhythmic drugs. Definition, electrophysiologic mechanisms, incidence, predisposing factors, and clinical implications. J Cardiovasc Electrophysiol 1995, 6:920–936.
Singh BN: Antiarrhythmic actions of amiodarone: a profile of a paradoxical agent. Am J Cardiol 1996, 78:41–53.
Sicouri S, Moro S, Litovsky S, et al.: Chronic amiodarone reduces transmural dispersion of repolarization in the canine heart. J Cardiovasc Electrophysiol 1997, 8:1269–1279.
Papp J Gy, Nemeth M, Krassoi I, et al.: Differential electrophysiologic effects of chronically administered amiodarone on canine Purkinje fibers versus ventricular muscle. J Pharmacol Exp Therap 1996, 1:187–196.
Cui G, Sen L, Sager PT, et al.: Effects of amiodarone, sematilide and sotalol on QT dispersion. Am J Cardiol 1995, 75:465–469.
Kodama I, Kamiya K, Toyama J: Cellular electropharmacology of amiodarone. Cardiovasc Res 1997, 35:13–29.
Singh BN: What niche will newer class III antiarrhythmic drugs occupy? Curr Cardiol Reports 2001, 3:314–323.
Roy D, Talajic M, Dorian P, et al., for the Canadian Trial of Atrial Fibrillation Investigators: Amiodarone to prevent recurrence of atrial fibrillation. N Engl J Med 2000, 342:913–918.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Srivatsa, U., Wadhani, N. & Singh, B.N. Mechanisms of antiarrhythmic drug actions and their clinical relevance for controlling disorders of cardiac rhythm. Curr Cardiol Rep 4, 401–410 (2002). https://doi.org/10.1007/s11886-002-0040-6
Issue Date:
DOI: https://doi.org/10.1007/s11886-002-0040-6