Abstract
Purpose of Review
Quinolones are a group of synthetic antibiotics widely use as first-line treatment for many infections. There has been an increase in the incidence of hypersensitivity reactions to quinolones in recent years, likely due to increased prescription. The purpose of this review is to summarize the clinical pictures, the methods used for diagnosing and the management of allergic reactions to quinolones.
Recent Findings
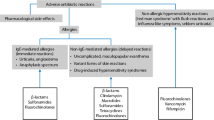
Allergic reactions to quinolones can be immediate or delayed, being anaphylaxis and maculopapular exanthema respectively the most frequent clinical entities. A precise diagnosis is particularly difficult since clinical history is often unreliable, skin tests can induce false-positive results, and commercial in vitro test are not well validated. Therefore, drug provocation testing is considered the gold standard to establish diagnosis, which is not a risk-free procedure. Cross-reactivity between quinolones is difficult to predict due to the small number of patients included in the few published studies. Moreover, hypersensitivity to quinolones has also been associated with beta-lactam and neuromuscular blocking agent allergies, although further studies are needed to understand the underlying mechanisms. Avoidance of the culprit quinolone is indicated in patients with a diagnosis of hypersensitivity to these drugs. When quinolone treatment is the only therapeutic option available, desensitization is necessary.
Summary
This review summarizes the complex diagnostic approach and management of allergic reactions to quinolones.



Similar content being viewed by others
Abbreviations
- AGEP:
-
Acute generalized exanthematous pustulosis
- BAT:
-
Basophil activation test
- DR:
-
Delayed reactions
- DPT:
-
Drug provocation test
- ELISA:
-
Enzyme-linked immunosorbent assay
- FDE:
-
Fixed drug eruption
- IR:
-
Immediate reactions
- IDT:
-
Intradermal test
- MPE:
-
Maculopapular exanthema
- NMBA:
-
Neuromuscular blocking agent
- RAST:
-
Radioallergosorbent test
- SPT:
-
Skin prick test
- SJS:
-
Stevens-Johnson syndrome
- TEN:
-
Toxic epidermal necrolysis
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Hooper DC. Mechanisms of action of antimicrobials: focus on fluoroquinolones. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America. 2001;32(Suppl 1):S9–S15.
Corcoy M, Dursteler C, Arbones E, Comps O, Escolano F. Intraoperative anaphylactic reaction after ciprofloxacin administration. Revista espanola de anestesiologia y reanimacion. 1999;46(9):419–20.
Solensky R, Earl HS, Gruchalla RS. Clinical approach to penicillin-allergic patients: a survey. Annals of allergy, asthma & immunology: official publication of the American College of Allergy, Asthma, & Immunology. 2000;84(3):329–33.
Dona I, Blanca-Lopez N, Torres MJ, et al. Drug hypersensitivity reactions: response patterns, drug involved, and temporal variations in a large series of patients. J Investig Allergol Clin Immunol. 2012;22(5):363–71.
Chang B, Knowles SR, Weber E. Immediate hypersensitivity to moxifloxacin with tolerance to ciprofloxacin: report of three cases and review of the literature. Ann Pharmacother. 2010;44(4):740–5.
Ball P. Quinolone generations: natural history or natural selection? J Antimicrob Chemother. 2000;46(Suppl T1):17–24.
Blanca-Lopez N, Andreu I, Torres Jaen MJ. Hypersensitivity reactions to quinolones. Curr Opin Allergy Clin Immunol. 2011;11(4):285–91.
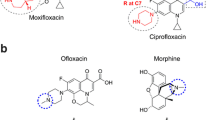
• Lobera T, Audicana MT, Alarcon E, Longo N, Navarro B, Munoz D. Allergy to quinolones: low cross-reactivity to levofloxacin. J Investig Allergol Clin Immunol. 2010;20(7):607–11. In this study, authors showed the low cross-reactivity between levofloxacin and ciprofloxacin in patients with immediate hypersensitivity reactions to quinolones, hightlighting the importance of the drug provocation test for finding safe quinolone alternatives.
González-Gregori R, Dolores Hernández Fernandez De Rojas M, López-Salgueiro R, Díaz-Palacios M, García AN. Allergy alerts in electronic health records for hospitalized patients. Annals of allergy, asthma & immunology: official publication of the American College of Allergy, Asthma & Immunology. 2012;109(2):137–40.
•• Blanca-Lopez N, Ariza A, Dona I, et al. Hypersensitivity reactions to fluoroquinolones: analysis of the factors involved. Clinical and experimental allergy: journal of the British Society for Allergy and Clinical Immunology. 2013;43(5):560–7. This study showed that fluoroquinolone allergy is more often confirmed in patients with immediate reactions, when moxifloxacin is the quinolone involved and inpatients with hypersensitivity to betalactams.
• Sachs B, Riegel S, Seebeck J, et al. Fluoroquinolone-associated anaphylaxis in spontaneous adverse drug reaction reports in Germany: differences in reporting rates between individual fluoroquinolones and occurrence after first-ever use. Drug Saf. 2006;29(11):1087–100. This article concluded that drug-incuced anaphylaxis seems to be associated with the fluoroquinolone administration mainly with moxifloxacin
•• Aranda A, Mayorga C, Ariza A, et al. In vitro evaluation of IgE-mediated hypersensitivity reactions to quinolones. Allergy. 2011;66(2):247–54. In this study, the authors analyzed the value of basophil activation test in the evaluation of IgE mediated reactions to quinolones. They found that the basophil activation test is sensitive and useful for diagnosis.
Renaudin JM, Beaudouin E, Ponvert C, Demoly P, Moneret-Vautrin DA. Severe drug-induced anaphylaxis: analysis of 333 cases recorded by the allergy vigilance network from 2002 to 2010. Allergy. 2013;68(7):929–37.
Salvo F, Polimeni G, Cutroneo PM, et al. Allergic reactions to oral drugs: a case/non-case study from an Italian spontaneous reporting database (GIF). Pharmacol Res. 2008;58(3–4):202–7.
Lapi F, Tuccori M, Motola D, et al. Safety profile of the fluoroquinolones: analysis of adverse drug reactions in relation to prescription data using four regional pharmacovigilance databases in Italy. Drug Saf. 2010;33(9):789–99.
Fernandez-Rivas M. Fixed drug eruption (FDE) caused by norfloxacin. Allergy. 1997;52(4):477–8.
Garcia Rodriguez R, Galindo Bonilla PA, Feo Brito FJ, et al. Chronic desensitization to quinolones in fixed drug eruption. J Investig Allergol Clin Immunol. 2011;21(1):76–7.
• Schmid DA, Depta JP, Pichler WJ. T cell-mediated hypersensitivity to quinolones: mechanisms and cross-reactivity. Clinical and experimental allergy: journal of the British Society for Allergy and Clinical Immunology. 2006;36(1):59–69. In this study patterns of recognition of different quinolones by T cell were analyzed in patients with delayed reaction to ciprofloxacin. The results demonstrated that cross-reactivity among the different quinolones is frequent
Davila G, Ruiz-Hornillos J, Rojas P, De Castro F, Zubeldia JM. Toxic epidermal necrolysis induced by levofloxacin. Annals of allergy, asthma & immunology: official publication of the American College of Allergy, Asthma, & Immunology. 2009;102(5):441–2.
Islam AF, Rahman MS. Levofloxacin-induced fatal toxic epidermal necrolysis. Ann Pharmacother. 2005;39(6):1136–7.
Yoon SY, Bae YJ, Cho YS, Moon HB, Kim TB. Toxic epidermal necrolysis induced by ofloxacin. Acta Derm Venereol. 2010;90(5):550–1.
Rodriguez-Morales A, Llamazares AA, Benito RP, Cocera CM. Fixed drug eruption from quinolones with a positive lesional patch test to ciprofloxacin. Contact Dermatitis. 2001;44(4):255.
Roujeau JC, Kelly JP, Naldi L, et al. Medication use and the risk of Stevens-Johnson syndrome or toxic epidermal necrolysis. N Engl J Med. 1995;333(24):1600–7.
Neuman MG, Cohen LB, Nanau RM. Quinolones-induced hypersensitivity reactions. Clin Biochem. 2015;48(10–11):716–39.
Sullivan TJOR, Gillian LK. Studies of the multiple drug allergy syndrome. J Allergy Clin Immunol. 1989;83:270.
Mockenhaupt M, Viboud C, Dunant A, et al. Stevens-Johnson syndrome and toxic epidermal necrolysis: assessment of medication risks with emphasis on recently marketed drugs. The EuroSCAR-study. The Journal of investigative dermatology. 2008;128(1):35–44.
Scherer K, Bircher AJ. Hypersensitivity reactions to fluoroquinolones. Curr Allergy Asthma Rep. 2005;5(1):15–21.
Davila I, Diez ML, Quirce S, Fraj J, De La Hoz B, Lazaro M. Cross-reactivity between quinolones. Report of three cases. Allergy. 1993;48(5):388–90.
Ben Said B, Berard F, Bienvenu J, Nicolas JF, Rozieres A. Usefulness of basophil activation tests for the diagnosis of IgE-mediated allergy to quinolones. Allergy. 2010;65(4):535–6.
Gonzalez-Mancebo E, Fernandez-Rivas M. Immediate hypersensitivity to levofloxacin diagnosed through skin prick test. Ann Pharmacother. 2004;38(2):354.
Sanchez-Morillas L, Rojas Perez-Ezquerra P, Reano-Martos M, Laguna-Martinez JJ, Gomez-Tembleque P. Systemic anaphylaxis caused by moxifloxacin. Allergol Immunopathol. 2010;38(4):226–7.
Gonzalez I, Lobera T, Blasco A, del Pozo MD. Immediate hypersensitivity to quinolones: moxifloxacin cross-reactivity. J Investig Allergol Clin Immunol. 2005;15(2):146–9.
Alonso MD, Martin JA, Quirce S, Davila I, Lezaun A, Sanchez CM. Fixed eruption caused by ciprofloxacin with cross-sensitivity to norfloxacin. Allergy. 1993;48(4):296–7.
Seitz CS, Brocker EB, Trautmann A. Diagnostic testing in suspected fluoroquinolone hypersensitivity. Clinical and experimental allergy: journal of the British Society for Allergy and Clinical Immunology. 2009;39(11):1738–45.
Venturini Diaz M, Lobera Labairu T, del Pozo Gil MD, Blasco Sarramian A, Gonzalez MI. In vivo diagnostic tests in adverse reactions to quinolones. J Investig Allergol Clin Immunol. 2007;17(6):393–8.
•• Manfredi M, Severino M, Testi S, et al. Detection of specific IgE to quinolones. J Allergy Clin Immunol. 2004;113(1):155–60. This is the first study in demonstrating the presence of specific IgE in patients with quinolone hypersensitivity by radioimmunoassay with sepharose. This study also demonstratedfrequent cross-reactivity within different quinolones.
Sanchez-Morillas L, Rojas Perez-Ezquerra P, Gonzalez Morales ML, Mayorga C, Gonzalez-Mendiola R, Laguna Martinez JJ. Fixed drug eruption due to norfloxacin and cross-reactivity with other quinolones. Allergol Immunopathol. 2013;41(1):60–1.
Ponce Guevara LV, Yges EL, Gracia Bara MT, Gonzalez Ruiz AM, Rodilla EM. Fixed drug eruption due to amoxicillin and quinolones. Annals of allergy, asthma & immunology: official publication of the American College of Allergy, Asthma & Immunology. 2013;110(1):61–2.
Mayorga C, Torres MJ, Fernandez J, Canto G, Blanca M. Cutaneous symptoms in drug allergy: what have we learnt? Curr Opin Allergy Clin Immunol. 2009;9(5):431–6.
Tokura Y. Quinolone photoallergy: photosensitivity dermatitis induced by systemic administration of photohaptenic drugs. J Dermatol Sci. 1998;18(1):1–10.
Hausermann P, Scherer K, Weber M, Bircher AJ. Ciprofloxacin-induced acute generalized exanthematous pustulosis mimicking bullous drug eruption confirmed by a positive patch test. Dermatology. 2005;211(3):277–80.
Howard-Thompson A, Cartmell B, Suda KJ. Toxic epidermal necrolysis reaction associated with the use of moxifloxacin. Int J Antimicrob Agents. 2014;44(2):178–9.
Demoly P, Kropf R, Bircher A, Pichler WJ. Drug hypersensitivity: questionnaire. EAACI interest group on drug hypersensitivity. Allergy. 1999;54(9):999–1003.
Empedrad R, Darter AL, Earl HS, Gruchalla RS. Nonirritating intradermal skin test concentrations for commonly prescribed antibiotics. J Allergy Clin Immunol. 2003;112(3):629–30.
Uyttebroek AP, Sabato V, Bridts CH, De Clerck LS, Ebo DG. Moxifloxacin hypersensitivity: uselessness of skin testing. J Allergy Clin Immunol Pract. 2015;3(3):443–5.
Barbaud A, Trechot P, Reichert-Penetrat S, Commun N, Schmutz JL. Relevance of skin tests with drugs in investigating cutaneous adverse drug reactions. Contact Dermatitis. 2001;45(5):265–8.
Broz P, Harr T, Hecking C, et al. Nonirritant intradermal skin test concentrations of ciprofloxacin, clarithromycin, and rifampicin. Allergy. 2012;67(5):647–52.
Brockow K, Romano A, Blanca M, Ring J, Pichler W, Demoly P. General considerations for skin test procedures in the diagnosis of drug hypersensitivity. Allergy. 2002;57(1):45–51.
Brajon D, Menetre S, Waton J, Poreaux C, Barbaud A. Non-irritant concentrations and amounts of active ingredient in drug patch tests. Contact Dermatitis. 2014;71(3):170–5.
Holzle E, Neumann N, Hausen B, et al. Photopatch testing: the 5-year experience of the German, Austrian, and Swiss Photopatch test group. J Am Acad Dermatol. 1991;25(1 Pt 1):59–68.
Fernandez TD, Torres MJ, Blanca-Lopez N, et al. Negativization rates of IgE radioimmunoassay and basophil activation test in immediate reactions to penicillins. Allergy. 2009;64(2):242–8.
Gomez E, Blanca-Lopez N, Torres MJ, et al. Immunoglobulin E-mediated immediate allergic reactions to dipyrone: value of basophil activation test in the identification of patients. Clinical and experimental allergy: journal of the British Society for Allergy and Clinical Immunology. 2009;39(8):1217–24.
Kvedariene V, Kamey S, Ryckwaert Y, et al. Diagnosis of neuromuscular blocking agent hypersensitivity reactions using cytofluorimetric analysis of basophils. Allergy. 2006;61(3):311–5.
Rouzaire P, Nosbaum A, Denis L, et al. Negativity of the basophil activation test in quinolone hypersensitivity: a breakthrough for provocation test decision-making. Int Arch Allergy Immunol. 2012;157(3):299–302.
Fernandez TD, Ariza A, Palomares F, et al. Hypersensitivity to fluoroquinolones: the expression of basophil activation markers depends on the clinical entity and the culprit fluoroquinolone. Medicine. 2016;95(23):e3679.
Decuyper II, Mangodt EA, Van Gasse AL, et al. In vitro diagnosis of immediate drug hypersensitivity anno 2017: potentials and limitations. Drugs in R&D. 2017;17(2):265–78.
Demir S GA, Akdeniz N, Aktas-Cetin E, et al.. Usefulness of in vivo and in vitro diagnostic tests in the diagnosis of hypersensitivity reactions to quinolones and in the evaluation of cross-reactivity: a comprehensive study including the latest quinolone gemifloxacin. Allergy Asthma Immunol Res. 2017;9(4):347–59.
Mayorga C, Andreu I, Aranda A, et al. Fluoroquinolone photodegradation influences specific basophil activation. Int Arch Allergy Immunol. 2013;160(4):377–82.
Campi P, Pichler WJ. Quinolone hypersensitivity. Curr Opin Allergy Clin Immunol. 2003;3(4):275–81.
Tokura Y, Seo N, Ohshima A, Yagi H, Furukawa F, Takigawa M. Lymphocyte stimulation test with drug-photomodified cells in patients with quinolone photosensitivity. J Dermatol Sci. 1999;21(1):34–41.
Tokura Y, Seo N, Yagi H, Furukawa F, Takigawa M. Cross-reactivity in murine fluoroquinolone photoallergy: exclusive usage of TCR Vbeta13 by immune T cells that recognize fluoroquinolone-photomodified cells. J Immunol. 1998;160(8):3719–28.
Lucena MI, Andrade RJ, Sanchez-Martinez H, Perez-Serrano JM, Gomez-Outes A. Norfloxacin-induced cholestatic jaundice. Am J Gastroenterol. 1998;93(11):2309–11.
Aberer W, Bircher A, Romano A, et al. Drug provocation testing in the diagnosis of drug hypersensitivity reactions: general considerations. Allergy. 2003;58(9):854–63.
Nishijima S, Nakagawa M. Fixed drug eruption caused by tosufloxacin tosilate. The Journal of international medical research. 1997;25(6):359–63.
Lozano Ayllon M, Gomez Martinez M, Mosquera MR, Laguna Martinez JJ, Orta Martiartu M. Fernandez de Miguel C. Fixed eruption caused by ciprofloxacin without cross-sensitivity to norfloxacin. Allergy. 1995;50(7):598–9.
Kameswari PD, Selvaraj N, Adhimoolam M. Fixed drug eruptions caused by cross-reactive quinolones. Journal of basic and clinical pharmacy. 2014;5(2):54–5.
Ball P, Mandell L, Patou G, Dankner W, Tillotson G. A new respiratory fluoroquinolone, oral gemifloxacin: a safety profile in context. Int J Antimicrob Agents. 2004;23(5):421–9.
Depta JP, Altznauer F, Gamerdinger K, Burkhart C, Weltzien HU, Pichler WJ. Drug interaction with T-cell receptors: T-cell receptor density determines degree of cross-reactivity. J Allergy Clin Immunol. 2004;113(3):519–27.
Sullivan TJOR, Gillian LK. Studies of the multiple drug allergy syndrome. J Allergy Clin Immunol. 1989;83:270.
•• Rouzaire P, Nosbaum A, Mullet C, et al. Immediate allergic hypersensitivity to quinolones associates with neuromuscular blocking agent sensitization. J Allergy Clin Immunol Pract. 2013;1(3):273–9 e1. In this study authors found a high prevalence of quaternary ammonium sensitization in patients with immediate hypersensitivity to quinolone, however the clinical relevance of this finding should be further analyzed. This could explain that in some cases quinolone hypersensitivity happens after the first drug exposition.
Erdem G, Staat MA, Connelly BL, Assa'ad A. Anaphylactic reaction to ciprofloxacin in a toddler: successful desensitization. Pediatr Infect Dis J. 1999;18(6):563–4.
Gea-Banacloche JC, Metcalfe DD. Ciprofloxacin desensitization. J Allergy Clin Immunol. 1996;97(6):1426–7.
Lantner RR. Ciprofloxacin desensitization in a patient with cystic fibrosis. J Allergy Clin Immunol. 1995;96(6 Pt 1):1001–2.
Bircher AJ, Rutishauser M. Oral "desensitization" of maculopapular exanthema from ciprofloxacin. Allergy. 1997;52(12):1246–8.
Acknowledgements and Authors’ Contributions
We thank James R. Perkins for his help with the English language version of this manuscript. MJT and ID compiled the entire manuscript; MJT contributed to the introduction, management, and desensitization and conclusion sections; IA contributed to the classification and chemical structure and in vitro tests sections; EM contributed to the clinical reactions immediate and delayed reactions, clinical history and skin tests sections; ID contributed to the epidemiology and risk factors; drug provocation test and cross-reactivity sections. All authors read and approved the final manuscript.
The present study has been supported by Institute of Health “Carlos III” of the Ministry of Economy and Competitiveness (grants cofunded by European Regional Development Fund (ERDF): RETIC ARADYAL RD16/0006/0001. I Doña holds a Juan Rodes research contract (JR15/00036) from the Carlos III National Health Institute, Spanish Ministry of Economy and Competitiveness (grants cofounded by European Social Fund, ESF).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
None of the authors have any conflict of interest, nor have they received any money for this study. Research is part of their daily activities. All authors had full access to all data and take responsibility for the integrity and accuracy of the data analysis.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Anaphylaxis and Drug Allergy
Rights and permissions
About this article
Cite this article
Doña, I., Moreno, E., Pérez-Sánchez, N. et al. Update on Quinolone Allergy. Curr Allergy Asthma Rep 17, 56 (2017). https://doi.org/10.1007/s11882-017-0725-y
Published:
DOI: https://doi.org/10.1007/s11882-017-0725-y