Abstract
Particulate matter (PM) is the major air pollution problem with health impacts in Estonia. The prevailing sources of particles are traffic and local heating. In this study, we quantified the health effects of PM in neighbourhoods of five main cities with a health impact assessment (HIA) approach that uses information on exposure, baseline mortality/morbidity and exposure–response relationships from previous epidemiological studies. The exposure was defined as modelled PM2.5 annual levels and daily averages of PM10 (monitoring data in Tallinn and Kohtla-Järve and modelled levels in Tartu, Narva and Pärnu). The modelled results were validated with data from monitoring stations and additional measuring programmes. The annual average concentration of PM2.5 in the neighbourhoods studied varied from 7.6 to 23.6 μg m−3. The analysis indicated that the exposure above natural background corresponds to 462 [95% confidence interval (CI) 120–815] premature deaths, resulting in 6,034 (95% CI 1,583–10,309) years of life lost per year. The average decrease in life-expectancy at birth per resident of Tallinn was estimated to be 0.63 (95% CI 0.16–1.08) years. In the polluted city centres, this average decrease may reach >1 year and in Pärnu, it may reach 0.95 year. However, in the least polluted neighbourhood, the decrease of life expectancy was only 0.17 years. In addition, 231 (95% CI 145–306) respiratory and 338 (95% CI 205–454) cardiovascular hospitalisations per year could be expected. The majority of the external costs are related to the long-term effects on mortality and amount to €270 (95% CI 190–350) million annually. In comparison, the costs of hospitalisations contribute just €1.1 (95% CI 0.6–1.6) million. The main differences in health impacts were mostly driven by differences in the pollution sources, the magnitude of such sources and distribution patterns in the atmosphere. The smallest health effects, with the exception of the green residential areas, were observed in the industrial cities Kohtla-Järve and Narva (due to the small share contributed by local residential heating and relatively little car traffic). However, it is questionable whether the mass of fine particles is the best indicator of air pollution risk in such areas.
Similar content being viewed by others
Introduction
Air pollution is one of the most salient issues in environmental health. The relationship between air pollution and human health is being intensively studied throughout the world; however, much remains to be understood. Even though air pollution levels have dropped in recent decades, air pollution remains a significant problem. One of the most important triggers of air pollution-related health effects is particulate matter (PM). PM consists of tiny particles of solid or liquid suspended in the air. Permitted values in PM10 (particles <10 μm in diameter) and PM2.5 (<2.5 μm) are frequently exceeded in Europe (EEA 2009). Additionally, epidemiological studies have shown that health effects may even appear at lower levels than the current limit values (ERS, ISEE, ISEA 2006), and available data have not established firm threshold values (Chen and Kan 2008; Anderson 2009).
PM has been widely studied and is associated with numerous health outcomes either with short-term or long-term exposure. Many short-term exposure studies have reported on the relationship between PM and mortality and morbidity, and there have also been several meta-analyses of these studies, some of which have provided pooled effect estimates (Anderson et al. 2005). The relationships have also been found in multi-city studies (Katsouyanni et al. 2003; Dominici et al. 2005; Wong et al. 2008).
Long-term exposure to PM (years or decades) leads to chronic health problems, such as cardiovascular disease, cardiopulmonary disease and lung cancer. The association between particles and increased mortality has been shown in cohort studies conducted in the USA (Dockery et al. 1993; Pope et al. 1995; Abbey et al. 1999; McDonnell et al. 2000; Pope et al. 2002; Jerrett et al. 2005; Laden et al. 2006) and Europe (Hoek et al. 2002; Filleul et al. 2005; Gehring et al. 2006; Naess et al. 2007). However, the results of these studies are often dissimilar, most likely due to the use of different exposure metrics and slightly diverse endpoints. The main difference between U.S. and European studies is that North American studies tend to compare mortality or morbidity between communities rather than within communities (Brunekreef 2007).
It is believed that the negative effects of particles are mostly related to chronic cardiopulmonary diseases (Pope and Dockery 2006; Schwarze et al. 2006). PM together with its components [e.g. polycyclic aromatic hydrocarbons (PAH), volatile organic compounds (VOC)] are able to induce inflammatory processes and cause activation of redox mechanisms and oxidative stress (Nel 2005; Pope and Dockery 2006; Brook 2008).
The components and fractions of particulate pollutants related to adverse health effects are still not fully understood; therefore, particles are largely used as an indicator of toxic air pollutants in epidemiological studies. Still, many studies have shown that the smaller the PM size, the higher the toxicity through mechanisms related to oxidative stress and inflammation (Valavanidis et al. 2008). While these toxicological data are supported by the epidemiological findings (Kappos et al. 2004), a review of epidemiological studies has shown that there seems to be ambiguous health effects also resulting from the coarser particle mode, namely, PM2.5–10 (Brunekreef and Forsberg 2005). According to these authors, acute exposure to coarse PM may be significantly associated with mortality and morbidity (especially respiratory hospital admissions). People with chronic diseases, such as asthma, chronic obstructive pulmonary disease, pneumonia, or other respiratory diseases, as well as patients with cardiovascular diseases, and diabetes are especially affected and can be defined as risk groups.
Different sources of particles
Estonia is an example of a transitional country, having regained its independence from the former Soviet Union only in 1991. The car fleet and main air pollution sources are currently relatively similar to those of other European countries, and especially similar to those of the Nordic countries (Orru et al. 2008). Traffic exhaust, the use of studded tires, and the intensive use of local heating by a large proportion of the population are considered to be the most problematic air pollution sources. In terms of pollutants, the relatively high levels of PM is of major concern (Urb et al. 2005; Kirso et al. 2006), similar to the situation in other European countries (Danielis 2006).
The health relevance of traffic-induced air pollution has been demonstrated in numerous studies on the associations between PM and acute respiratory illness, lung cancer, chronic respiratory, and cardiovascular diseases and cardiopulmonary mortality (WHO 2005b). While fine particles (PM2.5) and ultrafine particles (UFP) are mainly generated by combustion from exhaust, non-exhaust particles (mainly PM2.5–10) typically arise from abrasive sources, such as brake wear, tire wear and abrasion of the road surface (Thorpe and Harrison 2008). However, recent studies show that the use of studded tires contributes not only to PM10, but also to PM2.5 and likely also to PM1 (Gustafsson et al. 2008).
Residential wood combustion is widely used for local heating in Estonia (Kaasik et al. 2007) and has been found to be a significant source of particulate pollutants in many European countries (McDonald et al. 2000; Hellén et al. 2008; Frey et al. 2009). Moreover, combustion emits PAH and monosaccharide anhydrides (levoglucosan and mannosan), which have been found to be higher in urban areas than in background sites (Glasius et al. 2008).
Industrial sources of air pollution include boiler houses, power plants, smelters and incinerators. In Eastern Europe (especially Russia), these have a very high public health relevance (Cara et al. 2007; Jedrychowski et al. 2007). In the Estonian setting, district heating is an important point source that influences the urban air quality in major cities. However, the health risks from boiler houses are much smaller than those from other particle sources (Orru et al. 2009b).
Risk assessments of air pollution
Risk assessment is an important tool used by researchers to estimate the increased risk of health problems in people exposed to different levels of particles. This approach combines the results of human exposure, baseline mortality or morbidity in the population and exposure-–response (E–R) relationships from epidemiological studies. Health impact assessment (HIA) is a risk analysis tool by which a policy, programme or project may be evaluated based on its potential effects on the health and distribution of those effects in a population (Wismar et al. 2007). As such, PM effect estimates provides valuable information that can be used by policy-makers, for example to improve regulatory standards, reduce human exposure to toxic air pollutants, and decrease the risk of the public experiencing health problems.
According to the World Health Organisation (WHO; 2002), air pollution is responsible for 1.4% of all premature deaths and 0.8% of disability-adjusted life years globally. Exposure to outdoor air pollution accounts for approximately 2% of the global cardiopulmonary and 1% of the total respiratory disease burden (Cohen et al. 2004). Although the magnitude of the estimated increased risk does not appear to be extensive, the numbers of people affected are very large when extrapolated to the entire population that has been exposed (Chen et al. 2008). Moreover, public health effects can be substantial and costly, even in areas with relatively low levels of air pollution (Furberg et al. 2005).
Several HIAs focusing on particle effect in Europe have been published. One of the first studies (Künzli et al. 2000) estimated the impact of particulate pollutants in Austria, France and Switzerland. The authors found that PM causes 40,000 premature deaths, 25,000 new cases of chronic bronchitis and hundreds of thousands of chronic bronchitis episodes and asthma attacks each year. Other early HIAs have found that the lifespan of men in the Netherlands can be decreased by 1.5 years due to air pollution (Brunekreef 1997).
A recent large HIA included 23 European cities in the Apheis project (Boldo et al. 2006). This study showed that by reducing annual mean PM2.5 concentrations to 15 μg m−3, nearly 17,000 premature deaths could be avoided in the participating cities. Additionally, the average life expectancy at birth would increase by up to 2 years in more polluted cities (Boldo et al. 2006). The most recent large assessment from the USA found that a decrease in PM2.5 annual mean concentrations by 10 µg m−3 corresponded with an estimated increase in mean life expectancy by 0.61 years (Pope et al. 2009).
If the PM2.5 annual mean concentration remains <10 μg m−3 (WHO guidelines), the premature death rate would be reduced by 41 individuals per 100,000 population in the cities participating in the Apheis project (Ballester et al. 2008). Furthermore, if the agreed policies related to PM reductions were to be fulfilled, the average life expectancy in Europe would increase by 2.3 months by 2020 (WHO 2005a). Fine particles have been reported to have decrease the average life expectancy at birth by 8.6 months among all European Union (EU) citizens and to have been the cause of annual premature death amongst 348,000 people in Europe in 2000 (COMM 2005). Worldwide, the annual number of premature deaths due to outdoor PM2.5 has been estimated at >800,000 (Cohen et al. 2004). A more recent study places this estimate at 3.5 million premature deaths from cardiopulmonary disease and 220,000 mortalities from lung cancer (Anenberg et al. 2010).
To a large extent, the adverse health effects stem from air masses originating in more polluted areas. Air pollution in Russia causes 87,000 deaths annually and comprises approximately 4% of Russia’s total mortality (Golub and Strukova 2008). Estimates suggest that if the annual PM2.5 level were to be lowered to 12 μg m−3 in Tokyo, Japan, the total mortality rate would decrease by 8% and 6,700 premature deaths would be prevented (Yorifuji et al. 2005). Forsberg et al. (2005) assessed that PM matter accounted for 4,700 premature deaths in Swedish cities and nearly 600 deaths in the countryside.
The negative effects of air pollution of public health have both direct and indirect costs to society. In the EU, the external annual costs of air pollution are estimated at between €50 and161 billion due to premature mortality and €29 billion from morbidity. This corresponds to more than 1% of the gross domestic product (GDP) of the EU (WHO 2005a). It is also important to note that the majority of the morbidity-related external costs from air pollution are related to the public health sector (years of life lost, YLL) and not to the health care sector (hospitalisations and medical care) (ExternE 2005). In the relatively more polluted countries of the world, such as China, health care costs associated with air pollution account for as much as 3.3% of the GDP (World Bank 2007). In Russia, these values are even higher (Golub and Strukova 2008).
An air pollution HIA has already been published on Tallinn (Orru et al. 2009c), but in the study reported here, we have expanded the analysis by adding four other towns with diverse characteristics. We also discuss the possible reasons for these differences among the cities and probable weaknesses of the HIA procedure.
Material and methods
Study sites
The study presented in this paper was performed in five major cities in Estonia: Tallinn, Tartu, Kohtla-Järve, Narva and Pärnu.
Tallinn, the largest city in Estonia and also its capital, is situated on the country’s northern coast on the Gulf of Finland. The sources of air pollution in Tallinn are quite complex; however, traffic and local heating make significant contributions. Other contributors to increased emissions include the large number of cars in Tallinn that are more than 10 years old, with higher exhaust emission coefficients than newer vehicles, and the extensive use of wood stoves with low stacks for heating.
Tartu is the second largest city in Estonia and situated inland in a river valley. Similar to Tallinn, Tartu’s street network capacity remains limited as the volume of traffic steadily increases; consequently, congestion is common during rush hours. Furthermore, because of domestic heating in the large number of small apartment houses, the proportion of emissions related to local heating in Tartu is even higher than that in Tallinn.
Kohtla-Järve and Narva are industrial cities, situated in the eastern part of Estonia. The main industries are related to oil shale, electricity generation and the production of fertilisers. The traffic flows are much less dense than those in Tallinn and Tartu, and the proportion of local heating is very low.
Pärnu is the fifth largest city in Estonia and is situated on the western coast on Pärnu Bay; the Pärnu River flows through the city.
Thus, all five cities differ not only in their pollution patterns, but also by their different distribution patterns of pollution, with two cities situated on relatively windy coasts and three cities situated inland to varying extents. In the summer, Pärnu is a popular vacation resort, and the traffic flows increase extensively during holiday seasons. Moreover, local heating is especially common in the central part of the city.
Methodological overview of health impact assessment
Our aim was to estimate the health impacts of particle emissions in Tallinn, Tartu, Kohtla-Järve, Narva and Pärnu. The population data for Tallinn were obtained from the Estonian Population Register in 2006 and was divided according to address and registration into the following age groups: 0–6, 7–17, 18–27, 28–37, 38–47, 48–57, 58–67, 68+ years. The population data for the other four cities are based on population count data in 2000 (age groups: 0–5, 6–10, 11–15, 16–20, 21–30, 31–40, 41–50, 51–64, 65+ years), which were available in 500 × 500-m grids in the cities. The citizens’ residences were divided into neighbourhoods (regions with similar geographical and socio-economic patterns, among others, used in city planning and management) to identify site-specific exposure to air pollution and identify those areas with greatest risk. The age-structure of the population in these areas was also studied. Baseline total mortality data (A00–Y98), cardio-pulmonary mortality data for the sensitivity analysis (I10–I69, J00–J99) and data on hospitalisation due to cardiovascular (I00–I99) and respiratory cause (J00–J99) were retrieved from statistics for Estonia and from the Estonian Health Insurance Fund (EHIF) in 2006 and 2007.
The annual levels of PM2.5 were modelled using AirViro, Web based environmental GIS tool, with a grid resolution of 200 × 200 m. AirViro, developed by The Swedish Meteorological and Hydrological Institute, uses data on emissions, measured levels from air pollution monitoring stations and meteorological variables from meteorological stations. These data are used to perform air pollution dispersion modelling and mapping. The average concentration of grid cells in a neighbourhood was assigned as the typical long-term exposure to all residents of that area. Short-term effects of air pollution were calculated using daily average concentrations of PM10 recorded by the monitoring stations in 2006 in Tallinn and in 2008 in Kohtla-Järve (measured by beta-attenuation analyzers; model FH-62; Thermo Andersen, Smyrna, GA). In Tartu, Narva and Pärnu, the daily PM10 concentrations were modelled for 2008, and the modelling results were validated with monitoring station data recorded in Tallinn and Kohtla-Järve (stations operated during the whole study period) and in Tartu and Narva (stations opened summer and fall 2008). For additional model validation, measuring campaigns were conducted in Tartu, Narva and Pärnu using FH 62-I-R β-radiation absorption equipment.
From the exposure assessment for the HIA calculation, we subtracted the natural background, as many experts believe that there are likely no effects below these levels. Moreover, we can only diminish the anthropogenic part of the particulate pollution, not the natural sources. As fine particles are not measured in Estonian rural areas, the annual concentration (approx. 5 μg−3) in nearby areas of Helsinki was used as the natural background in Tartu, Kohtla-Järve, Narva and Pärnu (YTV 2008). However, as earlier modelling in Tallinn was based only on local emission, the effects in Tallinn were calculated according to these modelled annual levels that were smaller by a natural background rather than from actual measured concentrations (Orru et al. 2009c).
For the mortality analysis, we used the following E–R relationship from previous studies: a total mortality increase of 6.2% [95% confidence interval (CI) 1.6–11%] per 10 μg m−3 increase of annual mean PM2.5 concentration (Pope et al. 2002). For the sensitivity analysis, we used a cardio-pulmonary mortality increase of 13% (95% CI 10–13%) per 10 μg m−3 increase of annual mean PM2.5 concentration. We used an E–R relationship of 1.0114 per 10 μg m–3 increase of PM10 to calculate hospitalisations due to respiratory ailments (Atkinson et al. 2005). For cardiovascular hospitalisations, we used an E–R of 0.73% (95% CI 0.47–0.93%) as a weighted average based on the occurrence of cardiac and cerebrovascular admission with E–Rs from COMEAP meta-analysis. The cases of premature deaths were calculated in city districts (bigger city administrative region) in Tallinn and in neighbourhoods in other cities. For these calculations, the following equation was used:
where Y 0 is the baseline rate; pop is the number of exposed persons; β is the exposure–response relationship (relative risk); X is the estimated excess exposure.
The number of YLL and decrease in life expectancy were assessed using the WHO software programme AirQ 2.2.3 in all 142 neighbourhoods. The number of hospitalisations was determined with AirQ at different exposure intervals (10–19.9 μg m−3, 20–29.9 μg m−3, …) in Tallinn, Tartu, Kohtla-Järve, Narva, and Pärnu. For the calculation of short-term exposure effects, no effect was assumed below 10 μg m–3.
The direct expenses related to morbidity were calculated using the costs of hospitalisation, while time on sick leave (data provided by EHIF) was used to calculate loss of labour input.
To determine the cost of premature deaths, the value of a life year was calculated based on the decrease of life expectancy (due to PM exposure) retrieved from the statistical value of life (equal to 120-fold the GDP per capita in a country). The methodology has been described in more detail by Orru et al. (2009c).
Results
Population and baseline mortality and morbidity
Altogether it was possible to define 650,225 air pollution-affected inhabitants: 388,964 in Tallinn, 101,192 in Tartu, 46,187 in Kohtla-Järve, 68,445 in Narva and 45,437 in Pärnu. All 142 neighbourhoods were different in terms of area, number of inhabitants, population density, main pollution sources and pollution concentrations. The main factors affecting the HIA results were exposure to particles, baseline mortality/morbidity and, to some extent, population age structure in the cities.
The mortality rates differed among the cities in different age groups. On average, the rates per 100,000 citizens per year were 1,136 cases in Tallinn, 1,037 in Tartu, 1,515 in Kohtla-Järve, 1,346 in Narva and 1,251 in Pärnu. The baseline annual hospitalisation rates were determined separately for cardiovascular and respiratory admissions using the same principles. The analysis showed that, on average, there were annually 6,204 and 2,765 hospital admissions for cardiovascular and respiratory health-related problems per 100,000 people in these cities.
Exposure to PM
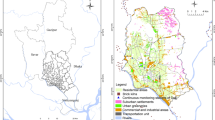
In Tallinn and Tartu, the city centre and neighbourhoods with local heating could be clearly distinguished as areas with bigger exposure to fine particles (Fig. 1). Higher concentrations also appeared in residential areas close to busy streets. In Pärnu, all of the areas in the central part of city and close to the river valley appeared to have high values (neighbourhood average of up to 23.6 μg m–3). The lower levels in Pärnu were notable in Raeküla and the southern part of Kesklinn, where blockhouses with central heating dominate. The lowest levels in these three cities appeared in residential areas located at the edges of the cities, where population density is relatively low (e.g. neighbourhood average in Ihaste was 7.6 μg m−3; Fig. 1).
Of the five cities studied, the smallest levels were distinguished in Narva and Kohtla-Järve. Even though these are large industrial cities, the outdoor concentrations of particles in urban areas were not very high there. Somewhat higher levels appeared in the Ahtme mining area and the region near the Narva River; however these levels remained lower than those in the other three cities. Moreover, these cities have less traffic and local heating is very rare.
The daily averages of PM10 differed considerably among days and cities. The highest values were seen in Tallinn, where they reached up to 136 μg m−3. The lowest concentrations—just few micrograms per cubic metre—were found on various days in different towns. However, the majority of the PM10 daily levels stayed between 10 and 30 μg m−3.
The modelling results were validated with monitoring station data from Tallinn and Kohtla-Järve (stations were in operation during the whole study period), Tartu and Narva (stations opened in the summer and fall of 2008) and Pärnu (mobile station in 2008).The average difference for all monitoring stations above the modelled PM levels over the measurement time was 22% (Table 1). In Tartu and Pärnu, the modelled levels were mostly higher than the measured levels, revealed that the emission databases for local heating are probably overestimated. In Kohtla-Järve and Narva, the measured values were higher than the modelled values, and we can assume that the emission databases are underestimated for these cities. The model validation in Tallinn has been discussed previously by Orru et al. (2009c). Even the agreements between the measured and modelled PM levels were imperfect; the model should satisfactorily represent the particle levels at different receptor points in the city as well.
Health effects and socioeconomic impacts
It was established that particulate pollutants cause on average of 462 premature deaths per year, corresponding to 6,034 YLL (Table 2). The greatest total loss (205–650 YLL) was expected in neighbourhoods with a large number of residents (>20,000), such as Mustamäe, Lilleküla, and Laagna in Tallinn, Annelinn in Tartu and Kesklinn in Pärnu (Fig. 2). This is likely to be reflected in the reduction of life expectancy by an average of 0.64 years in Tallinn, 0.68 years in Tartu and 0.95 years in Pärnu. In the city centres and in regions with extensive local heating, the life expectancy may be decreased by up to 1.2 years, whereas in the least polluted neighbourhoods, the decrease of life expectancy remains around 0.3 years (Fig. 3). If the average number of YLL is divided by the number of premature deaths, the loss will be approximately 13 years among these individuals, indicating greater health impacts among risk groups. The risk groups include individuals with chronic respiratory and cardiovascular disease, among other diseases, and immunosuppressed persons who could live several years less due to outdoor air pollution exposure. Additionally, 231 (95% CI 145–306) respiratory and 338 (95% CI 205–454) cardiovascular hospitalisations could be expected annually as a consequence of short-term exposure to PM10 (Table 2).
Most of the external costs of air pollution are related to the long-term effects on mortality and years of life lost from premature deaths. Annually, this will add up to €270 (95% CI 190–350) million. Compared to losses from premature mortality, the costs of short-term exposure are small, €1.1 (95% CI 0.6–1.6) million, with the majority (>55%) being directly related to hospitalisations, and the rest to lost input to the national economy due to time spent on sick leave.
Discussion
The size of the health effect
The results of this analysis indicate that particulate air pollution is a significant public health issue in Estonia. In five main cities, the estimated annual number of premature deaths due to outdoor particles was 462, resulting in 6,034 YLL among 650,225 residents; this poses a considerable health concern. The decrease in life expectancy in the city centre of Tallinn can be as high as 1.17 years. Moreover, the effects of local heating can result in a decrease in life expectancy of approximately 1 year in neighbourhoods with dense small apartment buildings (Tartu, Pärnu). The daily average limit values of PM10 have been exceeded occasionally, resulting in an increased number of hospitalisations. When comparing our HIA results with those of other studies, our estimated average decrease of life expectancy of 0.63 years (7.6 months) is slightly lower than the 8.6 months arrived at for Europe (COMM 2005). Even if the assessment methods are somewhat different, from a PM standpoint, major Estonian cities are, on average, nearly as polluted as European urban areas. Calculated YLL from outdoor air pollution could contribute up to 5% of the burden of premature mortality in Estonia (Lai et al. 2009).
Air pollution induces illnesses, hospitalisations, and years of life lost, all of which have negative implications on a state’s economy. The financial costs of health impairment are a tangible negative outcome that is acceptable to transitional societies where the socio-economic security is often prioritised at the expense of what has been called more “elusive” health concerns.
The societal costs of air pollution are relatively high. The estimates of the economic cost associated with health care because of air particulate pollution in Tallinn, Tartu, Kohtla-Järve, Narva, and Pärnu are, on average, €270 million. This corresponds to an estimate of approximately 3% of the cities GDP. Compared to the economic effect assessments for other European regions, the estimations in Estonia are relatively high. For example, in the EU assessment, the external costs of air pollution were estimated to correspond to slightly more than 1% of the GDP of EU (WHO 2005a). However, when the proportion is calculated from the cost estimation’s upper confidence interval, the proportion is much higher (CI €79–190 billion). The assessment from more polluted regions, such as China, give estimations as high as 3.3% of the GDP (World Bank 2007). In extreme cases, such as Beijing, there have been even higher estimated costs, 6.6% of the GDP (Zhang et al. 2007). In Russia, it is as high as 6.5% of the GDP (Golub and Strukova 2008).
As mentioned above, the majority of the external costs from air pollution are related to premature deaths and not to hospitalisations and health care (ExternE 2005). However, there are many suggested effects typically not included in the calculations, such as the induction of asthma and effects on children. The willingness to pay concept has been used in various calculations; these indicate the maximum amount a person would be willing to pay for an extra life year. It is a hypothetical variable, as health and well-being have no real market value. Moreover, respondents may consider it unethical to put a price tag on someone’s death or illness (de Hollander and Melse 2006). Willingness-to-pay estimations are likely overstated. Therefore, it has been suggested that researchers should consider the external costs of the effect of air pollution on health as crude estimates, thereby allowing comparisons between different risks rather than regarding the expenses in real monetary terms (de Hollander and Melse 2006). However, we should not underestimate the value of external expenditure calculations as an instrument for analysing the costs and benefits of various environmental health policy options in specific socio-economic contexts.
Susceptible risk groups
Environmental health effects vary among different societal groups, and special attention should be paid to susceptible groups. There is a wide range of susceptibility within any large population, and certain individuals are at risk even at the lowest observed concentration levels (WHO 2006). This includes people with underlying health conditions, such as cardiovascular and lung diseases, asthmatics, socially deprived people, children, and the elderly. Due to a lack of data, some of the susceptible groups, such as children, are not very well described in impact calculations.
Our assessment in Tallinn revealed that even though the average loss of life expectancy in all citizens is 0.63 years, the loss per premature case is greater, with an estimation of approximately 13 years. This indicates that there are much larger effects among risk groups. As life expectancy and healthy-lived years are lower in Estonia than in the EU15 countries, the susceptible groups may be younger. However, the decrease in life expectancy in risk groups reported in our study is similar to Swedish results (Forsberg et al. 2005). When compared to Swedish cases, people in Estonia tend to die on average 6 years earlier, and the detrimental effects appear most in the younger age groups.
Modelled results
Dispersion models have not been widely used in studies on air pollution health effects. However, using modelled results, we can attain a more detailed picture of the exposure gradients in different parts of the city. A station represents a nearby neighbourhood or areas with similar characteristics. During the study period, air pollution was monitored at three sites in Tallinn, one site in Kohtla-Järve and at sites for short periods in Tartu, Narva and Pärnu. Air pollution dispersion modelling can also be used to construct scenarios of future trends and predict the extent of the health impact. The potential health impacts of air quality management programmes have been estimated prospectively (Mindell and Joffe 2004; Tonne et al. 2008; Tiwary et al. 2009).
However, there are several limitations to the disperson modelling methodology, the major one being the poor quality of the emissions data. To reduce the effect of this drawback, a database of local heating emissions was developed based on the results of a survey on fuel consumption (Kaasik et al. 2007). As the questionnaire was conducted several years ago in Tartu, and the results were then adjusted to reflect the conditions in four other towns, certain differences in results may exist (e.g. prevailing sources of pollution). The more frequent use of electric heaters and heat pumps may decrease the emissions from local heating, which may be the main reasons for the overestimation of emissions from local heating in Tartu and Pärnu. The considerable difference (on average 22%) between all monitoring stations and modelled PM levels is an important concern in terms of the reliability of the results. Thus, we should be careful in applying the results to other scenarios. However, as the concentrations have in some places been overestimated and in other places underestimated, the inaccuracy may not be crucial.
Estimates of the emission factors for traffic pollution dispersion modelling were determined using CORINAIR (EAA 2007). As there are negligible differences in car usage and climate, the differences are predictable. The modelled PM concentrations in Tallinn showed a relatively good agreement with measured values in the residential area and areas close to busy streets; however there was a considerable underestimation in the industrial area (Orru et al. 2009c). Even though the data on pollutant emissions and meteorology are representative, more advanced models also take into account land use, topography, buildings (especially street canyon models) and atmospheric chemistry.
Exposure resolution for the home address improves as the resolution of the air pollution modelling grid increases. On the other hand, the results become more sensitive to small errors in position of the source and receptor (i.e. home). In addition, as the dispersion modelling resolution is increased, the need for more precise emission data also rises. However, in real life, people move and spend time in different environments (e.g. indoors), and particles are usually measured outdoors. Thus, the personal exposure assessment has sometimes been implemented. Since this assessment method is relatively expensive, usually only 30–130 people are monitored (Nerriere et al. 2005; Williams et al. 2008; Folino et al. 2009; Scapellato et al. 2009). Another conflict arises in methodology, as we have used more detailed modelled exposure data, whereas in original the ACS study (from where E–R relationships were implemented), the data from nearby monitoring stations had been applied (Pope and Dockery 2006). The spatial analysis methods have recently been implemented as well (Krewski et al. 2009).
Selection of E–R relationships
One of the crucial issues in the HIA process is the selection of the E–R relationship, as has also been discussed by Hubbell et al. (2009). The main problem lies in the lack of consistent long-term PM exposure effect studies for PM in Europe. One future possible solution could be the use of estimates from the ESCAPE (European Study of Cohorts for Air Pollution Effects) study (Brunekreef 2008).
In our HIA, we used the relationships reported from the American Cancer Society (ACS) study. The sensitivity analysis was also made with an E–R relationship from the same study, but a different baseline mortality indicator was used. One question arose regarding whether we can carry over the E–R relationships from the U.S. to the Estonian context. The pollution sources in Estonia are different; for example, there is a larger proportion of local heating. Additionally, the measuring sites might be dissimilar since primarily urban background levels are controlled and monitored in USA relative to only urban areas in Europe. Considering that the ACS study is the biggest air pollution cohort study to date and that it includes over 100 metropolitan areas, the results should show an average E–R relationship for all kinds of areas with multiple sources (Pope et al. 2002). Support for this point of view is found in the study by Jerrett et al. (2005), which is an analysis of ACS participants from California, where traffic-induced particles explain a bigger proportion of gradients in the PM pollution and where E–R relationships are nearly threefold higher. Krewski et al. (2009) extended the analyses, showing various E–R relationships in different parts of the country. A recent study in Tartu, Estonia indicated that the E–R relationship could actually be much higher for traffic-induced particles (Orru et al. 2009a). Thus, when conducting health impact assessments, prevailing pollution sources should be considered.
The question related to higher toxicity and effects per PM mass may have arisen in the Kohtla-Järve industrial area, where specific toxic pollutants, such as phenols, are very common. Our HIA showed smaller effects there as the outdoor annual mass concentration of fine particles was used as a pollution indicator. Therefore, largely used PM2.5 mass concentrations may not have been the best indicator in this case. Finding the E–R relationship for special areas and special sources remains one of the main research challenges in future studies. Moreover, the model underestimated the concentration of fine particles in these towns.
Conclusions
There is a substantial exposure to PM in Tallinn, Tartu, Kohtla-Järve, Narva and Pärnu that cause considerable health effects in the form of cardiopulmonary diseases. Biomass combustion (local heating) and traffic remain the most important contributing factors to air quality degradation and the resulting adverse health effects. However, the assessment of the importance of the problem is not a straightforward task. The sources and effects are manifold and vary among different societal groups.
Characterising the effects among neighbourhoods could provide the public with a much better perception of the effects in different parts of the city. However, increasing the resolution also evokes many questions on methodology, particularly as the main pollution sources in cities were different, yet the same E–R relationship was used, possibly resulting in some bias. These factors indicate that the results of the HIA should be considered carefully as much expectancy has been assumed, and the assessed local condition may be somewhat different from the study sites where the original estimations were drawn from.
References
Abbey DE, Nishino N, McDonnell WF, Burchette RJ, Knutsen SF, Lawrence Beeson W, Yang JX (1999) Long-term inhalable particles and other air pollutants related to mortality in nonsmokers. Am J Respir Crit Care Med 159:373–382
Anderson HR (2009) Air pollution and mortality: A history. Atmos Environ 43:142–152
Anderson HR, Atkinson RW, Peacock JL, Sweeting MJ, Marston L (2005) Ambient particulate matter and health effects—publication bias in studies of short-term associations. Epidemiology 16:155–163
Anenberg SC, Horowitz LW, Tong DQ, West JJ (2010) An estimate of the global burden of anthropogenic ozone and fine particulate matter on premature human mortality using atmospheric modeling. Environ Health Perspect. doi:10.1289/ehp.0901220
Atkinson RW, Anderson HR, Medina S, Iñiguez C, Forsberg B, Segerstedt B, Artazcoz L, Paldy A, Zorrilla B, Lefranc A, Michelozzi P (2005) Analysis of all-age respiratory hospital admissions and particulate air pollution within the APHEIS programme. In: Medina S, Boldo E, Saklad M, Niciu EM, Krzyzanowski M, Frank F, Cambra K, Mücke HG, Zorilla B, Atkinson R, Le Tertre A, Forsberg B et al (eds) APHEIS Health Impact Assessment of Air Pollution and Communication Strategy. Third year report. Institut de Veille Sanitaire, Saint-Maurice
Ballester F, Medina S, Boldo E, Goodman P, Neuberger M, Iniguez C, Künzli N, network obotA, (2008) Reducing ambient levels of fine particulates could substantially improve health: a mortality impact assessment for 26 European cities. J Epidemiol Community Health 62:98–105
Boldo E, Medina S, LeTertre A, Hurley F, Mucke H, Ballester F, Aguilera I, Eilstein D (2006) Apheis: Health impact assessment of long-term exposure to PM2.5 in 23 European cities. Eur J Epidemiol 21:449–458
Brook RD (2008) Cardiovascular effects of air pollution. Clin Sci 115:175–187
Brunekreef B (1997) Air pollution and life expectancy: is there a relation? Occup Environ Med 54:781–784
Brunekreef B (2007) Health effects of air pollution observed in cohort studies in Europe. J Expo Sci Environ Epidemiol 17:S61–65
Brunekreef B (2008) Design of amMulti-cohort study of air pollution effects in Europe: The ESCAPE Study. In: 20th Annu Conf Int Soc-Environmental Epidemiology. Lippincott Williams & Wilkins, Pasadena, pp S32–S32
Brunekreef B, Forsberg B (2005) Epidemiological evidence of effects of coarse airborne particles on health. Eur Respir J 26:309–318
Cara AC, Buntinx F, Van den Akker M, Dinant GJ, Manolovici C (2007) Industrial air pollution and children’s respiratory health: a natural experiment in Calarasi. Eur J Gen Pract 13:135–143
Chen B, Kan H (2008) Air pollution and population health: a global challenge. Environ Health Prevent Med 13:94–101
Chen Y, Craig L, Krewski D (2008) Air quality risk assessment and management. J Toxicol Environ Health A 71:24–39
Cohen A, Anderson H, Ostro B, Pandey K, Krzyzanowski M, Künzli N, Gutschmidt K, Pope CI, Romieu I, Samet J, Smith K (2004) Urban air pollution. In: Ezzati M, Lopez A, Rodgers A, Murray C (eds) Comparative quantification of health risks. World Health Organization, Geneva
COMM (2005) Commission Staff Working Paper. Annex to: The communication on thematic strategy on air pollution and the directive on “ambient air quality and cleaner air for Europe”. Impact Assessment Commission of the European Communities, Brussels
Danielis R (2006) Air pollution and road transport in Europe. A cluster and a regression analysis among countries and cities. Working Paper no. 105. Trieste University, Trieste
de Hollander AEM, Melse JM (2006) Valuing the health impact of air pollution: deaths, DALYs or dollars? In: Ayres JG, Maynard RL, Richards R (eds) Air pollution and health. Imperial College Press, London
Dockery DW, Pope CA 3rd, Xu X, Spengler JD, Ware JH, Fay ME, Ferris BG Jr, Speizer FE (1993) An association between air pollution and mortality in six U.S. cities. N Engl J Med 329:1753–1759
Dominici F, McDermott A, Daniels M, Zeger SL, Samet JM (2005) Revised analyses of the National Morbidity, Mortality, and Air Pollution Study: Mortality among residents of 90 cities. Journal of Toxicology and Environmental Health-Part a-Current Issues 68:1071–1092
EAA (2007) EMEP/CORINAIR Emission Inventory Guidebook—2007. European Environment Agency, Copenhagen
EEA (2009) Air quality, health and vegetation impacts of PM10 and ozone in Europe, year 2005. European Environment Agency, Copenhagen
ERS, ISEE, ISEA (2006) Declaration on need for stricter European regulation of air pollution. Munich and Paris
ExternE (2005) Externalities of energy: methodology update. European Communities, Luxembourg
Filleul L, Rondeau V, Vandentorren S, Le Moual N, Cantagrel A, Annesi-Maesano I, Charpin D, Declercq C, Neukirch F, Paris C, Vervloet D, Brochard P, Tessier J-F, Kauffmann F, Baldi I (2005) Twenty five year mortality and air pollution: results from the French PAARC survey. Occup Environ Med 62:453–460
Folino AF, Scapellato ML, Canova C, Maestrelli P, Bertorelli G, Simonato L, Iliceto S, Lotti M (2009) Individual exposure to particulate matter and the short-term arrhythmic and autonomic profiles in patients with myocardial infarction. Eur Heart J 30:1614–1620
Forsberg B, Hansson H, Johansson C, Areskoug H, Persson K, Jarvholm B (2005) Comparative health impact assessment of local and regional particulate air pollutants in Scandinavia. AMBIO 34:11–19
Frey AK, Tissari J, Saarnio KM, Timonen HJ, Tolonen-Kivimäki O, Aurela MA, Saarikoski SK, Makkonen U, Hytönen K, Jokiniemi J, Salonen RO, Hillamo REJ (2009) Chemical composition and mass size distribution of fine particulate matter emitted by a small masonry heater. Boreal Environ Res 14:255–271
Furberg M, Preston K, Sawyer D, Brauer M, Hanvelt R (2005) Health and air quality 2005—phase 2: valuation of health impacts from air quality in the lower Fraser valley airshed. Final report. BC Lung Association, Vancouver
Gehring U, Heinrich J, Kramer U, Grote V, Hochadel M, Sugiri D, Kraft M, Rauchfuss K, Eberwein HG, Wichmann HE (2006) Long-term exposure to ambient air pollution and cardiopulmonary mortality in women. Epidemiology 17:545–551
Glasius M, Ketzel M, Wåhlin P, Bossi R, Stubkjær J, Hertel O, Palmgren F (2008) Characterization of particles from residential wood combustion and modelling of spatial variation in a low-strength emission area. Atmos Environ 42:8686–8697
Golub A, Strukova E (2008) Evaluation and identification of priority air pollutants for environmental management on the basis of risk analysis in Russia. J Toxicol Environ Health A 71:86–91
Gustafsson M, Blomqvist G, Gudmundsson A, Dahl A, Swietlicki E, Bohgard M, Lindbom J, Ljungman A (2008) Properties and toxicological effects of particles from the interaction between tyres, road pavement and winter traction material. Sci Total Environ 393:226–240
Hellén H, Hakola H, Haaparanta S, Pietarila H, Kauhaniemi M (2008) Influence of residential wood combustion on local air quality. Sci Total Environ 393:283–290
Hoek G, Brunekreef B, Goldbohm S, Fischer P, van den Brandt PA (2002) Association between mortality and indicators of traffic-related air pollution in the Netherlands: a cohort study. Lancet 360:1203–1209
Hubbell B, Fann N, Levy JI (2009) Methodological considerations in developing local-scale health impact assessments: balancing national, regional, and local data. Air Quality Atmos Health 2:99–110
Jedrychowski W, Masters E, Choi H, Sochacka E, Flak E, Mroz E, Pac A, Jacek R, Kaim I, Skolicki Z, Spengler JD, Perera F (2007) Pre-pregnancy dietary vitamin A intake may alleviate the adverse birth outcomes associated with prenatal pollutant exposure: epidemiologic cohort study in Poland. Int J Occup Environ Health 13:175–180
Jerrett M, Burnett R, Ma R, Pope I, Krewski D, Newbold K, Thurston G, Shi Y, Finkelstein N, Calle E, Thun M (2005) Spatial analysis of air pollution and mortality in Los Angeles. Epidemiology 16:1–10
Kaasik M, Orru H, Tekkel E, Vals P (2007) Situation and tendencies in air quality in a north European medium-sized town. In: Sokhi R, Neophytou M (eds) Abstr 6th Int Conf Urban Air Quality. Larnaca, p 212
Kappos AD, Bruckmann P, Eikmann T, Englert N, Heinrich U, Hoppe P, Koch E, Krause GH, Kreyling WG, Rauchfuss K, Rombout P, Schulz-Klemp V, Thiel WR, Wichmann HE (2004) Health effects of particles in ambient air. Int J Hyg Environ Health 207:399–407
Katsouyanni K, Touloumi G, Samolu E, Petasakis Y, Analitis A, Le Tertre A, Rossi G, Zmirou D, Ballester F, Boumghar A, Anderson HR, Wojtyniak B, Paldy A, Braustein R, Pekkanen J, Schindler C, Schwartz J (2003) Sensitivity analysis of various models of short-term effects of ambient particles on total mortality in 29 cities in APHEA2 Revised Analyses of Time-Series of Air Pollution and Health. Special Report. Health Effects Institute, Boston, pp 157–164
Kirso U, Steinnes E, Urb G, Teinemaa E, Kettrup A, Gebefügi I, Adamson J, Adamson K (2006) Levels of priority pollutants in ambient air of Estonia. In: 7th Eur Meet Environmental Chemistry. Brno
Krewski D, Jerrett M, Burnett RT, Ma R, Hughes E, Shi Y (2009) Extended follow-up and spatial analysis of the American Cancer Society study linking particulate air pollution and mortality. Health Effects Institute, Cambridge
Künzli N, Kaiser R, Medina S, Studnicka M, Chanel O, Filliger P, Herry M, Horak F, Puybonnieux-Texier V, Quenel P, Schneider J, Seethaler R, Vergnaud J, Sommer H (2000) Public-health impact of outdoor and traffic-related air pollution: a European assessment. Lancet 356:795–801
Laden F, Schwartz J, Speizer FE, Dockery DW (2006) Reduction in fine particulate air pollution and mortality: Extended follow-up of the Harvard Six Cities study. Am J Respir Crit Care Med 173:667–672
Lai T, Habicht J, Kiivet R-A (2009) Measuring burden of disease in Estonia to support public health policy. Eur J Public Health 19:541–547
McDonald JD, Zielinska B, Fujita EM, Sagebiel JC, Chow JC, Watson JG (2000) Fine particle and gaseous emission rates from residential wood combustion. Environ Sci Technol 34:2080–2091
McDonnell WF, Nishino-Ishikawa N, Petersen FF, Chen LH, Abbey DE (2000) Relationships of mortality with the fine and coarse fractions of long-term ambient PM10 concentrations in nonsmokers. J Expos Anal Environ Epidemiol 10:427–436
Mindell J, Joffe M (2004) Predicted health impacts of urban air quality management. J Epidemiol Community Health 58:103–113
Naess O, Nafstad P, Aamodt G, Claussen B, Rosland P (2007) Relation between concentration of air pollution and cause-specific mortality: four-year exposures to nitrogen dioxide and particulate matter pollutants in 470 neighborhoods in Oslo, Norway. Am J Epidemiol 165:435–443
Nel A (2005) Air pollution-related illness: effects of particles. Science 308:804–806
Nerriere É, Zmirou-Navier D, Blanchard O, Momas I, Ladner J, Le Moullec Y, Personnaz M-B, Lameloise P, Delmas V, Target A, Desqueyroux H (2005) Can we use fixed ambient air monitors to estimate population long-term exposure to air pollutants? The case of spatial variability in the Genotox ER study. Environ Res 97:32–42
Orru H, Jõgi R, Kaasik M, Forsberg B (2009a) Chronic traffic-induced PM exposure and self-reported respiratory and cardiovascular health in the RHINE Tartu Cohort. Int J Environ Res Public Health 6:2740–2751
Orru H, Kaasik M, Antov D, Forsberg B (2008) Evolution of traffic flows and traffic-induced air pollution due to structural changes and development during 1993–2006 in Tartu (Estonia). Baltic J Road Bridge Eng 3:206–212
Orru H, Kaasik M, Merisalu E, Forsberg B (2009b) Health impact assessment in case of biofuel peat—co-use of environmental scenarios and exposure-response functions. Biomass Bioenergy 33:1080–1086
Orru H, Teinemaa E, Lai T, Tamm T, Kaasik M, Kimmel V, Kangur K, Merisalu E, Forsberg B (2009c) Health impact assessment of particulate pollution in Tallinn using fine spatial resolution and modeling techniques. Environ Health 8:7
Pope C, Burnett R, Thun M, Calle E, Krewski D, Ito K, Thurston G (2002) Lung cancer, cardiopulmonary mortality, and long-term exposure to fine particulate air pollution. JAMA 287:1132–1141
Pope C, Dockery D (2006) Health effects of fine particulate air pollution: lines that connect. J Air Waste Manage Assoc 56:709–742
Pope CA 3rd, Thun MJ, Namboodiri MM, Dockery DW, Evans JS, Speizer FE, Heath CW Jr (1995) Particulate air pollution as a predictor of mortality in a prospective study of U.S. adults. Am J Respir Crit Care Med 151:669–674
Pope CA III, Ezzati M, Dockery DW (2009) Fine-Particulate Air Pollution and Life Expectancy in the United States. N Engl J Med 360:376–386
Scapellato ML, Canova C, de Simone A, Carrieri M, Maestrelli P, Simonato L, Bartolucci GB (2009) Personal PM10 exposure in asthmatic adults in Padova, Italy: seasonal variability and factors affecting individual concentrations of particulate matter. Int J Hygiene Environ Health 212:626–636
Schwarze PE, Ovrevik J, Lag M, Refsnes M, Nafstad P, Hetland RB, Dybing E (2006) Particulate matter properties and health effects: consistency of epidemiological and toxicological studies. Human Exp Toxicol 25:559–579
Zhang M, Song Y, Cai X (2007) A health-based assessment of particulate air pollution in urban areas of Beijing in 2000–2004. Sci Total Environ 376:100–108
Thorpe A, Harrison RM (2008) Sources and properties of non-exhaust particulate matter from road traffic: A review. Scie Total Environ 400:270–282
Tiwary A, Sinnett D, Peachey C, Chalabi Z, Vardoulakis S, Fletcher T, Leonardi G, Grundy C, Azapagic A, Hutchings TR (2009) An integrated tool to assess the role of new planting in PM10 capture and the human health benefits: a case study in London. Environ Poll 157:2645–2653
Tonne C, Beevers S, Armstrong B, Kelly F, Wilkinson P (2008) Air pollution and mortality benefits of the London Congestion Charge: spatial and socioeconomic inequalities. Occup Environ Med 65:620–627
Urb G, Teinemaa E, Kettrup A, Gebefügi I, Laja M, Reinik J, Tamm E, Kirso U (2005) Atmospheric pollution in Tallinn, levels of priority pollutants. Proc Estonian Acad Sci Chem 54:123–133
Valavanidis A, Fiotakis K, Vlachogianni T (2008) Airborne particulate matter and human health: toxicological assessment and importance of size and composition of particles for oxidative damage and carcinogenic mechanisms. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev 26:339–362
WHO (2002) World Health Report 2002—reducing risks, promoting healthy life. World Health Organization, Geneva
WHO (2005a) European Union can save up to E161 billion a year by reducing air-pollution deaths
WHO (2005b) Health effects of transport-related air pollution. WHO/Euro, Bonn
WHO (2006) Health risks of particulate matter from long-range transboundary air pollution. WHO/Euro, Copenhagen
Williams R, Rea A, Vette A, Croghan C, Whitaker D, Stevens C, McDow S, Fortmann R, Sheldon L, Wilson H, Thornburg J, Phillips M, Lawless P, Rodes C, Daughtrey H (2008) The design and field implementation of the Detroit Exposure and Aerosol Research Study. J Expo Sci Environ Epidemiol 22:22
Wismar M, Blau J, Ernst K, Figueras J (eds) (2007) The effectiveness of health impact assessment. Scope and limitations of supporting decision-making in Europe. WHO, Copenhagen
Wong CM, Vichit-Vadakan N, Kan HD, Qian ZM, Teams PP (2008) Public Health and Air Pollution in Asia (PAPA): A multicity study of short-term effects of air pollution on mortality. Environ Health Perspect 116:1195–1202
World Bank (2007) Cost of Pollution in China. World Bank, Washington D.C.
Yorifuji T, Yamamoto E, Tsuda T, Kawakami N (2005) Health impact assessment of particulate matter in Tokyo, Japan. Int Arch Occup Environ Health 60:179–185
YTV (2008) Ilmanlaatu pääkaupunkiseudulla vuonna 2007. Helsinki Metropolital Area Counsil, Helsinki
Acknowledgements
The studies were funded by the Estonian Ministry of Environment. We would like to thank the local governments of these cities for supportive co-operation. The air quality assessment methods were partially supported by Estonian Ministry of Education and Research, research themes SF0180060s09, SF0180038s08 and SF1090050s07.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Orru, H., Maasikmets, M., Lai, T. et al. Health impacts of particulate matter in five major Estonian towns: main sources of exposure and local differences. Air Qual Atmos Health 4, 247–258 (2011). https://doi.org/10.1007/s11869-010-0075-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11869-010-0075-6