Opinion statement
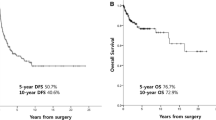
Leiomyosarcoma arises from smooth muscle and represents one of the most common soft tissue sarcomas. Despite aggressive multimodality care, over half of the patients will ultimately develop metastatic and incurable disease with a median survival of 12–18 months. At present, there is no standard system to classify leiomyosarcoma, which itself is a heterogeneous disease. Classification by tumor location is the most simplistic approach and is most frequently utilized in clinical practice. Tumor location impacts diagnosis (recognition pre-operatively versus at the time of surgery) as well as treatment (ability to completely resect with clear margins with minimal morbidity). While tumor location can impact prognosis, for example, extremity tumors would generally be considered as lower risk than inferior vena cava tumors, leiomyosarcoma can exhibit a heterogeneous behavior irrespective of tumor location. Specifically, some patients have rapidly progressing disease despite aggressive chemotherapy, while others display a more indolent course even in the metastatic setting. The pathogenic drivers of the heterogeneity observed in tumor behavior are not well understood. As we learn more about the molecular composition of leiomyosarcoma, various classification groups have been proposed as discussed here. Ultimately, it is unlikely that one variable will be adequate for tumor classification, and a combination of location and molecular composition will be necessary to develop appropriate risk stratification nomograms and treatment strategies.
Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Choi JH, Ro JY. The 2020 WHO classification of tumors of soft tissue: selected changes and new entities. Adv Anat Pathol. 2021;28(1):44–58.
Brennan MF, et al. Lessons learned from the study of 10,000 patients with soft tissue sarcoma. Ann Surg. 2014;260(3):416–21 (discussion 421-2).
Lamm W, et al. Distinctive outcome in patients with non-uterine and uterine leiomyosarcoma. BMC Cancer. 2014;14(1):981.
• George S, et al. Soft tissue and uterine leiomyosarcoma. J Clin Oncol. 2018;36(2):144–50. Contemporary comprehensive review of leiomyosarcoma.
Wang Z, et al. Survival of patients with metastatic leiomyosarcoma: the MD Anderson Clinical Center for targeted therapy experience. Cancer Med. 2016;5(12):3437–44.
Judson I, et al. Doxorubicin alone versus intensified doxorubicin plus ifosfamide for first-line treatment of advanced or metastatic soft-tissue sarcoma: a randomised controlled phase 3 trial. Lancet Oncol. 2014;15(4):415–23.
Seddon B, et al. Gemcitabine and docetaxel versus doxorubicin as first-line treatment in previously untreated advanced unresectable or metastatic soft-tissue sarcomas (GeDDiS): a randomised controlled phase 3 trial. Lancet Oncol. 2017;18(10):1397–410.
Oosten AW, et al. Outcomes of first-line chemotherapy in patients with advanced or metastatic leiomyosarcoma of uterine and non-uterine origin. Sarcoma. 2009;2009:348910.
•• Burns J, Jones RL, Huang PH. Molecular subtypes of leiomyosarcoma: moving toward a consensus. Clin Transl Discov. 2022;2(4):e149. A review of the literature for molecular subtyping and clinical imact.
Neuville A, Chibon F, Coindre J-M. Grading of soft tissue sarcomas: from histological to molecular assessment. Pathology. 2014;46(2):113–20.
Bonvalot S, et al. Preoperative radiotherapy plus surgery versus surgery alone for patients with primary retroperitoneal sarcoma (EORTC-62092: STRASS): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2020;21(10):1366–77.
D’Ambrosio L, et al. Doxorubicin plus dacarbazine, doxorubicin plus ifosfamide, or doxorubicin alone as a first-line treatment for advanced leiomyosarcoma: a propensity score matching analysis from the European Organization for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group. Cancer. 2020;126(11):2637–47.
Pritts EA, et al. The prevalence of occult leiomyosarcoma at surgery for presumed uterine fibroids: a meta-analysis. Gynecol Surg. 2015;12(3):165–77.
Zak K, Zaremba B, Rajkak Ak, Kotarski J, Amant F, Bobinski M. Perioperative differentiation of uterine leiomyoma and leiomyosarcomas: current possibilities and future directions. Cancers. 2022;14(8):1966–91.
Mingoli A, et al. Leiomyosarcoma of the inferior vena cava: analysis and search of world literature on 141 patients and report of three new cases. J Vasc Surg. 1991;14(5):688–99.
Wachtel H, et al. Outcomes after resection of leiomyosarcomas of the inferior vena cava: a pooled data analysis of 377 cases. Surg Oncol. 2015;24(1):21–7.
Hollenbeck ST, et al. Surgical treatment and outcomes of patients with primary inferior vena cava leiomyosarcoma. J Am Coll Surg. 2003;197(4):575–9.
• Beck AH, et al. Discovery of molecular subtypes in leiomyosarcoma through integrative molecular profiling. Oncogene. 2010;29(6):845–54. First discovery of variable molecular subtypes in leiomyosarcoma.
Guo X, et al. Clinically relevant molecular subtypes in leiomyosarcoma. Clin Cancer Res. 2015;21(15):3501–11.
Barlin JN, et al. Molecular subtypes of uterine leiomyosarcoma and correlation with clinical outcome. Neoplasia. 2015;17(2):183–9.
Cancer Genome Atlas Research Network. Comprehensive and integrated genomic characterization of adult soft tissue sarcomas. Cell. 2017;171(4):950–65.
Chudasama P, et al. Integrative genomic and transcriptomic analysis of leiomyosarcoma. Nat Commun. 2018;9(1):144.
Anderson ND, et al. Lineage-defined leiomyosarcoma subtypes emerge years before diagnosis and determine patient survival. Nat Commun. 2021;12(1):4496.
Hemming ML, et al. Oncogenic gene-expression programs in leiomyosarcoma and characterization of conventional, inflammatory, and uterogenic subtypes. Mol Cancer Res. 2020;18(9):1302–14.
Guo X, Forgó E, van de Rijn M. Molecular subtyping of leiomyosarcoma with 3’ end RNA sequencing. Genomics data. 2015;5:366–7.
Oza J, et al. Homologous recombination repair deficiency as a therapeutic target in sarcoma. Semin Oncol. 2020;47(6):380–9.
Yang J, et al. Therapeutic perspectives for adult soft tissue sarcoma-updates from the 2022 ASCO annual meeting. Cancer Biol Med. 2022;19(10):1496–502.
• Schaefer IM, et al. Relationships between highly recurrent tumor suppressor alterations in 489 leiomyosarcomas. Cancer. 2021;127(15):2666–73. Molecular analysis of larges leiomyosarcoma cohort to date.
Pautier P, et al. Doxorubicin alone versus doxorubicin with trabectedin followed by trabectedin alone as first-line therapy for metastatic or unresectable leiomyosarcoma (LMS-04): a randomised, multicentre, open-label phase 3 trial. Lancet Oncol. 2022;23(8):1044–54.
Hemming ML, et al. Preclinical modeling of leiomyosarcoma identifies susceptibility to transcriptional CDK inhibitors through antagonism of E2F-driven oncogenic gene expression. Clin Cancer Res. 2022;28(11):2397–408.
Kim CG, et al. Phase II clinical trial of eribulin-gemcitabine combination therapy in previously treated patients with advanced liposarcoma or leiomyosarcoma. Clin Cancer Res. 2022;28(15):3225–34.
Chen TW, et al. A single-arm phase Ib/II study of lenvatinib plus eribulin in advanced liposarcoma and leiomyosarcoma. Clin Cancer Res. 2022;28(23):5058–65.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors do not have any potential conflicts of interest to disclose.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Sarcoma
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hickman, A., Siontis, B.L. Not All Leiomyosarcomas Are the Same: How to Best Classify LMS. Curr. Treat. Options in Oncol. 24, 327–337 (2023). https://doi.org/10.1007/s11864-023-01067-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11864-023-01067-2