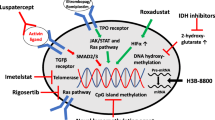
Opinion statement
Lower risk myelodysplastic syndromes are typically characterized by an indolent disease course with a relatively low risk of transformation into acute myeloid leukemia. These patients are classically identified using the revised International Prognostic Scoring System and most likely its molecular version in the near future which may change the paradigm of treatment. The overall goals of care are symptomatic control to reduce transfusion requirements and improve quality of life. Symptomatic anemia is the most common indication to initiate disease-specific therapies after the optimization of supportive measures. Currently, erythropoiesis-stimulating agents remain the standard upfront therapy for anemia, and patients with del(5q) cytogenetic changes can benefit from lenalidomide monotherapy. Other therapeutic options after failure of upfront treatment include luspatercept, hypomethylating agents, and immunosuppressive therapies after taking into account of individualized disease features. Allogeneic hematopoietic stem cell transplant is the only potentially curative option and is usually reserved for medically fit patients with severe symptomatic cytopenias who failed all standard options and/or the disease is progressing toward higher risk categories. Fortunately, novel investigational therapies are rapidly emerging by targeting different biological processes contributing to MDS pathogenesis, and eligible patients should be managed in clinical trials if available.

Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Ogawa S. Genetics of MDS. Blood. 2019;133(10):1049–59.
Carraway HE, Saygin C. Therapy for lower-risk MDS. Hematology Am Soc Hematol Educ Program. 2020;2020(1):426–33.
Stahl M, Abdel-Wahab O, Wei AH, Savona MR, Xu ML, Xie Z, et al. An agenda to advance research in MDS: a TOP 10 Priority List from the first international workshop in MDS (iwMDS). Blood Adv. 2022. https://doi.org/10.1182/bloodadvances.2022008747
Greenberg PL, Tuechler H, Schanz J, Sanz G, Garcia-Manero G, Solé F, et al. Revised international prognostic scoring system for myelodysplastic syndromes. Blood. 2012;120(12):2454–65.
Benton CB, Khan M, Sallman D, Nazha A, Nogueras González GM, Piao J, et al. Prognosis of patients with intermediate risk IPSS-R myelodysplastic syndrome indicates variable outcomes and need for models beyond IPSS-R. Am J Hematol. 2018;93(10):1245–53.
Zeidan AM, Sekeres MA, Garcia-Manero G, Steensma DP, Zell K, Barnard J, et al. Comparison of risk stratification tools in predicting outcomes of patients with higher-risk myelodysplastic syndromes treated with azanucleosides. Leukemia. 2016;30(3):649–57.
•• Bernard E, Tuechler H, Greenberg PL, Hasserjian RP, Arango OJE, Nannya Y, et al. Molecular International Prognostic Scoring System for Myelodysplastic Syndromes. NEJM Evidence. 2022;1(7):EVIDoa2200008 (This study reports the molecular version of international prognostic scoring system, which will reshape the therapeutic algorithm of MDS in the near future given refined risk stratification by incorporating mutational information.).
Zimmerman Savill KM, Gajra A, Price K, Kish JK, Brown-Bickerstaff C, Falkenstein A, et al. Lower-risk myelodysplastic syndromes: erythropoiesis-stimulating agent treatment approaches and outcomes in the United States. Blood. 2021;138(Supplement 1):4657.
•• Platzbecker U, Fenaux P, Adès L, Giagounidis A, Santini V, van de Loosdrecht AA, et al. Proposals for revised IWG 2018 hematological response criteria in patients with MDS included in clinical trials. Blood. 2019 Mar 7;133(10):1020–30. In this report, an expert panel on behalf of the International Working Group proposed a revised version of the hematological response criteria of MDS, particularly the erythroid response assessment, hoping to provide more accurate and clinically meaningful treatment response assessments in MDS patients.
Platzbecker U, Symeonidis A, Oliva EN, Goede JS, Delforge M, Mayer J, et al. A phase 3 randomized placebo-controlled trial of darbepoetin alfa in patients with anemia and lower-risk myelodysplastic syndromes. Leukemia. 2017;31(9):1944–50.
Fenaux P, Santini V, Spiriti MAA, Giagounidis A, Schlag R, Radinoff A, et al. A phase 3 randomized, placebo-controlled study assessing the efficacy and safety of epoetin-α in anemic patients with low-risk MDS. Leukemia. 2018;32(12):2648–58.
Garelius HKG, Johnston WT, Smith AG, Park S, de Swart L, Fenaux P, et al. Erythropoiesis-stimulating agents significantly delay the onset of a regular transfusion need in nontransfused patients with lower-risk myelodysplastic syndrome. J Intern Med. 2017;281(3):284–99.
Park S, Greenberg P, Yucel A, Farmer C, O’Neill F, De Oliveira BC, et al. Clinical effectiveness and safety of erythropoietin-stimulating agents for the treatment of low- and intermediate-1-risk myelodysplastic syndrome: a systematic literature review. Br J Haematol. 2019;184(2):134–60.
Hellström-Lindberg E, Gulbrandsen N, Lindberg G, Ahlgren T, Dahl IMS, Dybedal I, et al. A validated decision model for treating the anaemia of myelodysplastic syndromes with erythropoietin + granulocyte colony-stimulating factor: significant effects on quality of life. Br J Haematol. 2003;120(6):1037–46.
Malcovati L, Della Porta MG, Strupp C, Ambaglio I, Kuendgen A, Nachtkamp K, et al. Impact of the degree of anemia on the outcome of patients with myelodysplastic syndrome and its integration into the WHO classification-based Prognostic Scoring System (WPSS). Haematologica. 2011;96(10):1433–40.
Hellström-Lindberg E, Ahlgren T, Beguin Y, Carlsson M, Carneskog J, Dahl IM, et al. Treatment of anemia in myelodysplastic syndromes with granulocyte colony-stimulating factor plus erythropoietin: results from a randomized phase II study and long-term follow-up of 71 patients. Blood. 1998;92(1):68–75.
Giagounidis A, Mufti GJ, Fenaux P, Sekeres MA, Szer J, Platzbecker U, et al. Results of a randomized, double-blind study of romiplostim versus placebo in patients with low/intermediate-1-risk myelodysplastic syndrome and thrombocytopenia. Cancer. 2014;120(12):1838–46.
Kantarjian HM, Fenaux P, Sekeres MA, Szer J, Platzbecker U, Kuendgen A, et al. Long-term follow-up for up to 5 years on the risk of leukaemic progression in thrombocytopenic patients with lower-risk myelodysplastic syndromes treated with romiplostim or placebo in a randomised double-blind trial. Lancet Haematol. 2018;5(3):e117–26.
Oliva EN, Alati C, Santini V, Poloni A, Molteni A, Niscola P, et al. Eltrombopag versus placebo for low-risk myelodysplastic syndromes with thrombocytopenia (EQoL-MDS): phase 1 results of a single-blind, randomised, controlled, phase 2 superiority trial. Lancet Haematol. 2017;4(3):e127–36.
Vicente A, Patel BA, Gutierrez-Rodrigues F, Groarke E, Giudice V, Lotter J, et al. Eltrombopag monotherapy can improve hematopoiesis in patients with low to intermediate risk-1 myelodysplastic syndrome. Haematologica. 2020;105(12):2785–94.
Hutzschenreuter F, Monsef I, Kreuzer KA, Engert A, Skoetz N. Granulocyte and granulocyte-macrophage colony stimulating factors for newly diagnosed patients with myelodysplastic syndromes. Cochrane Database Syst Rev. 2016;16(2):CD009310.
Komrokji R, Al Ali N, Chan O, Sweet K, Kuykendall A, Lancet J, et al. IDH mutations are enriched in myelodysplastic syndrome patients with severe neutropenia and can be a potential for targeted therapy. Haematologica. 2022. https://doi.org/10.3324/haematol.2022.281607
Fenaux P, Kiladjian JJ, Platzbecker U. Luspatercept for the treatment of anemia in myelodysplastic syndromes and primary myelofibrosis. Blood. 2019;133(8):790–4.
Sherman ML, Borgstein NG, Mook L, Wilson D, Yang Y, Chen N, et al. Multiple-dose, safety, pharmacokinetic, and pharmacodynamic study of sotatercept (ActRIIA-IgG1), a novel erythropoietic agent, in healthy postmenopausal women. J Clin Pharmacol. 2013;53(11):1121–30.
Abdulkadyrov KM, Salogub GN, Khuazheva NK, Sherman ML, Laadem A, Barger R, et al. Sotatercept in patients with osteolytic lesions of multiple myeloma. Br J Haematol. 2014;165(6):814–23.
Komrokji R, Garcia-Manero G, Ades L, Prebet T, Steensma DP, Jurcic JG, et al. Sotatercept with long-term extension for the treatment of anaemia in patients with lower-risk myelodysplastic syndromes: a phase 2, dose-ranging trial. Lancet Haematol. 2018;5(2):e63-72.
Platzbecker U, Germing U, Götze KS, Kiewe P, Mayer K, Chromik J, et al. Luspatercept for the treatment of anaemia in patients with lower-risk myelodysplastic syndromes (PACE-MDS): a multicentre, open-label phase 2 dose-finding study with long-term extension study. Lancet Oncol. 2017;18(10):1338–47.
•• Fenaux P, Platzbecker U, Mufti GJ, Garcia-Manero G, Buckstein R, Santini V, et al. Luspatercept in patients with lower-risk myelodysplastic syndromes. N Engl J Med. 2020 Jan 9;382(2):140–51. This phase 3 study demonstrated the efficacy of luspatercept to reduce the severity of anemia in transfusion-dependent lower risk MDS patients with ring sideroblasts, who were either refractory to or unlikely to respond to erythropoiesis-stimulating agents.
Bristol Myers Squibb Announces Positive Topline Results of Phase 3 COMMANDS Trial. Available from: https://news.bms.com/news/details/2022/Bristol-Myers-Squibb-Announces-Positive-Topline-Results-of-Phase-3-COMMANDS-Trial/default.aspx. [Cited 2023 Jan 6].
List A, Ebert BL, Fenaux P. A decade of progress in myelodysplastic syndrome with chromosome 5q deletion. Leukemia. 2018;32(7):1493–9.
List A, Kurtin S, Roe DJ, Buresh A, Mahadevan D, Fuchs D, et al. Efficacy of lenalidomide in myelodysplastic syndromes. N Engl J Med. 2005;352(6):549–57.
• List A, Dewald G, Bennett J, Giagounidis A, Raza A, Feldman E, et al. Lenalidomide in the myelodysplastic syndrome with chromosome 5q deletion. N Engl J Med. 2006 Oct 5;355(14):1456–65. This single arm study showed that lenalidomide could reduce transfusion requirements and reverse cytogenetic abnormalities in MDS patients with 5q31 deletion.
Fenaux P, Giagounidis A, Selleslag D, Beyne-Rauzy O, Mufti G, Mittelman M, et al. A randomized phase 3 study of lenalidomide versus placebo in RBC transfusion-dependent patients with Low-/Intermediate-1-risk myelodysplastic syndromes with del5q. Blood. 2011;118(14):3765–76.
Kuendgen A, Lauseker M, List AF, Fenaux P, Giagounidis AA, Brandenburg NA, et al. Lenalidomide does not increase AML progression risk in RBC transfusion-dependent patients with Low- or Intermediate-1-risk MDS with del(5q): a comparative analysis. Leukemia. 2013;27(5):1072–9.
Mallo M, Cervera J, Schanz J, Such E, García-Manero G, Luño E, et al. Impact of adjunct cytogenetic abnormalities for prognostic stratification in patients with myelodysplastic syndrome and deletion 5q. Leukemia. 2011;25(1):110–20.
Heuser M, Meggendorfer M, Cruz MMA, Fabisch J, Klesse S, Köhler L, et al. Frequency and prognostic impact of casein kinase 1A1 mutations in MDS patients with deletion of chromosome 5q. Leukemia. 2015;29(9):1942–5.
Jädersten M, Saft L, Smith A, Kulasekararaj A, Pomplun S, Göhring G, et al. TP53 mutations in low-risk myelodysplastic syndromes with del(5q) predict disease progression. J Clin Oncol. 2011;29(15):1971–9.
Mossner M, Jann JC, Nowak D, Platzbecker U, Giagounidis A, Götze K, et al. Prevalence, clonal dynamics and clinical impact of TP53 mutations in patients with myelodysplastic syndrome with isolated deletion (5q) treated with lenalidomide: results from a prospective multicenter study of the german MDS study group (GMDS). Leukemia. 2016;30(9):1956–9.
Lodé L, Ménard A, Flet L, Richebourg S, Loirat M, Eveillard M, et al. Emergence and evolution of TP53 mutations are key features of disease progression in myelodysplastic patients with lower-risk del(5q) treated with lenalidomide. Haematologica. 2018;103(4):e143–6.
•• Sperling AS, Guerra VA, Kennedy JA, Yan Y, Hsu JI, Wang F, et al. Lenalidomide promotes the development of TP53-mutated therapy-related myeloid neoplasms. Blood. 2022 Oct 20;140(16):1753–63. This study showed that lenalidomide exposure provides a selective advantage to TP53-mutated hematopoietic stem/progenitor cells and is associated with the development of therapy-related myeloid neoplasms.
López Cadenas F, Lumbreras E, Xicoy B, Sánchez J, Coll R, Slama B, et al. Phase 3 study of lenalidomide (LEN) vs placebo in non-transfusion dependent (TD) low risk Del(5q) MDS patients - interim analysis of the European Sintra-REV Trial. Blood. 2020;136(Supplement 1):28–9.
Toribio Castelló SM, Lopez-Cadenas F, Preudhomme C, Villaverde Ramiro A, Fenwarth L, Lumbreras E, et al. Long-term evolution of somatic mutations in patients with Del(5q) MDS early treated with lenalidomide in the Sintra-Rev clinical trial: Safe and Effecitive Approach? Blood. 2022;140(Supplement 1):9740–3.
Raza A, Reeves JA, Feldman EJ, Dewald GW, Bennett JM, Deeg HJ, et al. Phase 2 study of lenalidomide in transfusion-dependent, low-risk, and intermediate-1 risk myelodysplastic syndromes with karyotypes other than deletion 5q. Blood. 2008;111(1):86–93.
Santini V, Almeida A, Giagounidis A, Gröpper S, Jonasova A, Vey N, et al. Randomized phase III study of lenalidomide versus placebo in RBC transfusion-dependent patients with lower-risk non-del(5q) myelodysplastic syndromes and ineligible for or refractory to erythropoiesis-stimulating agents. J Clin Oncol. 2016;34(25):2988–96.
Toma A, Kosmider O, Chevret S, Delaunay J, Stamatoullas A, Rose C, et al. Lenalidomide with or without erythropoietin in transfusion-dependent erythropoiesis-stimulating agent-refractory lower-risk MDS without 5q deletion. Leukemia. 2016;30(4):897–905.
List AF, Sun Z, Verma A, Bennett JM, Komrokji RS, McGraw K, Maciejewski J, Altman JK, Cheema PS, Claxton DF, Luger SM, Mattison RJ, Wassenaar TR, Artz AS, Schiffer CA, Litzow MR, Tallman MS. Lenalidomide-epoetin alfa versus lenalidomide monotherapy in myelodysplastic syndromes refractory to recombinant erythropoietin. J Clin Oncol. 2021;39(9):1001–9. https://doi.org/10.1200/JCO.20.01691.
Wang C, McGraw KL, McLemore AF, Komrokji R, Basiorka AA, Al Ali N, et al. Dual pyroptotic biomarkers predict erythroid response in lower-risk non-del(5q) myelodysplastic syndromes treated with lenalidomide and recombinant erythropoietin. Haematologica. 2022;107(3):737–9.
Garcia-Manero G, Jabbour E, Borthakur G, Faderl S, Estrov Z, Yang H, et al. Randomized open-label phase II study of decitabine in patients with low- or intermediate-risk myelodysplastic syndromes. J Clin Oncol. 2013;31(20):2548–53.
Filì C, Malagola M, Follo MY, Finelli C, Iacobucci I, Martinelli G, et al. Prospective phase II Study on 5-days azacitidine for treatment of symptomatic and/or erythropoietin unresponsive patients with low/INT-1-risk myelodysplastic syndromes. Clin Cancer Res. 2013;19(12):3297–308.
Tobiasson M, Dybedahl I, Holm MS, Karimi M, Brandefors L, Garelius H, et al. Limited clinical efficacy of azacitidine in transfusion-dependent, growth factor-resistant, low- and Int-1-risk MDS: results from the Nordic NMDSG08A phase II trial. Blood Cancer J. 2014;7(4):e189.
Thépot S, Ben Abdelali R, Chevret S, Renneville A, Beyne-Rauzy O, Prébet T, et al. A randomized phase II trial of azacitidine +/- epoetin-β in lower-risk myelodysplastic syndromes resistant to erythropoietic stimulating agents. Haematologica. 2016;101(8):918–25.
• Garcia-Manero G, Santini V, Almeida A, Platzbecker U, Jonasova A, Silverman LR, et al. Phase III, randomized, placebo-controlled trial of CC-486 (oral azacitidine) in patients with lower-risk myelodysplastic syndromes. J Clin Oncol. 2021 May 1;39(13):1426–36. This phase 3 study evaluated CC-486 (oral azacitidine) in lower risk MDS patients, which not only improved the transfusion-independent rate but induced bilineage hematologic responses. However, more early deaths were observed in the CC-486 arm, most attributed to infections in those with significant neutropenia. Potentially, a shorter schedule of CC-486 could provide a better efficacy and safety balance, which needs to be evaluated in clinical trials.
Azacitidine and Cedazuridine (ASTX030) Oral DNMT inhibitor (Hematological Malignancies) – Astex. Available from: https://astx.com/azacitidine-and-cedazuridine-astx030-oral-dnmt-inhibitor-hematological-malignancies/. [Cited 2023 Jan 7].
Garcia-Manero G, McCloskey JK, Griffiths EA, Yee K, Zeidan AM, Al-Kali A, et al. Oral decitabine/cedazuridine in patients with lower risk myelodysplastic syndrome: a longer-term follow-up of from the ascertain study. Blood. 2021;138(Supplement 1):66.
Passweg JR, Giagounidis AAN, Simcock M, Aul C, Dobbelstein C, Stadler M, et al. Immunosuppressive therapy for patients with myelodysplastic syndrome: a prospective randomized multicenter phase III trial comparing antithymocyte globulin plus cyclosporine with best supportive care–SAKK 33/99. J Clin Oncol. 2011;29(3):303–9.
Saunthararajah Y, Nakamura R, Nam JM, Robyn J, Loberiza F, Maciejewski JP, et al. HLA-DR15 (DR2) is overrepresented in myelodysplastic syndrome and aplastic anemia and predicts a response to immunosuppression in myelodysplastic syndrome. Blood. 2002;100(5):1570–4.
Sloand EM, Wu CO, Greenberg P, Young N, Barrett J. Factors affecting response and survival in patients with myelodysplasia treated with immunosuppressive therapy. J Clin Oncol. 2008;26(15):2505–11.
Stahl M, DeVeaux M, de Witte T, Neukirchen J, Sekeres MA, Brunner AM, et al. The use of immunosuppressive therapy in MDS: clinical outcomes and their predictors in a large international patient cohort. Blood Adv. 2018;2(14):1765–72.
de Witte T, Bowen D, Robin M, Malcovati L, Niederwieser D, Yakoub-Agha I, et al. Allogeneic hematopoietic stem cell transplantation for MDS and CMML: recommendations from an international expert panel. Blood. 2017;129(13):1753–62.
Cutler CS, Lee SJ, Greenberg P, Deeg HJ, Pérez WS, Anasetti C, et al. A decision analysis of allogeneic bone marrow transplantation for the myelodysplastic syndromes: delayed transplantation for low-risk myelodysplasia is associated with improved outcome. Blood. 2004;104(2):579–85.
Alessandrino EP, Porta MGD, Malcovati L, Jackson CH, Pascutto C, Bacigalupo A, et al. Optimal timing of allogeneic hematopoietic stem cell transplantation in patients with myelodysplastic syndrome. Am J Hematol. 2013;88(7):581–8.
Briatore F, Barrera G, Pizzimenti S, Toaldo C, Casa CD, Laurora S, et al. Increase of telomerase activity and hTERT expression in myelodysplastic syndromes. Cancer Biol Ther. 2009;8(10):883–9.
• Steensma DP, Fenaux P, Van Eygen K, Raza A, Santini V, Germing U, et al. Imetelstat Achieves meaningful and durable transfusion independence in high transfusion-burden patients with lower-risk myelodysplastic syndromes in a phase II study. J Clin Oncol. 2021 Jan 1;39(1):48–56. This is the phase 2 part of the IMerge (MDS3001) study, which showed that imetelstat resulted in a durable transfusion independent rate in transfusion-dependent lower risk MDS patients who were ineligible for or relapsed/refractory to erythropoiesis-stimulating agents.
Geron announces positive top-line results from IMerge phase 3 trial of imetelstat in lower risk MDS. Available from: https://ir.geron.com/investors/press-releases/press-release-details/2023/Geron-Announces-Positive-Top-Line-Results-from-IMerge-Phase-3-Trial-of-Imetelstat-in-Lower-Risk-MDS/default.aspx. [Cited 2023 Jan 7].
Sebert M, Cluzeau T, Beyne Rauzy O, Stamatoulas Bastard A, Dimicoli-Salazar S, Thepot S, et al. Ivosidenib monotherapy is effective in patients with IDH1 mutated myelodysplastic syndrome (MDS): the Idiome phase 2 study By the GFM Group. Blood. 2021;138(Supplement 1):62.
Ades L, Dimicoli-Salazar S, Sebert M, Cluzeau T, Stamatoulas Bastard A, Laribi K, et al. Enasidenib (ENA) is effective in patients with IDH2 mutated myelodysplastic syndrome (MDS): the Ideal phase 2 study by the GFM Group. Blood. 2021;138(Supplement 1):63.
Komrokji RS, Al Ali N, Chan O, Kuykendall AT, Sweet K, Lancet JE, et al. IDH mutations are enriched in myelodysplastic syndromes patients with severe neutropenia: a potential targeted therapy. Blood. 2021;138(Supplement 1):1526.
Patnaik MM, Santini V. Targeting ineffective hematopoiesis in myelodysplastic syndromes. Am J Hematol. 2022;97(2):171–3.
Chen N, Hao C, Liu BC, Lin H, Wang C, Xing C, et al. Roxadustat treatment for anemia in patients undergoing long-term dialysis. N Engl J Med. 2019;381(11):1011–22.
Chen N, Hao C, Peng X, Lin H, Yin A, Hao L, et al. Roxadustat for anemia in patients with kidney disease not receiving dialysis. N Engl J Med. 2019;381(11):1001–10.
Henry DH, Glaspy J, Harrup R, Mittelman M, Zhou A, Carraway HE, et al. Roxadustat for the treatment of anemia in patients with lower-risk myelodysplastic syndrome: open-label, dose-selection, lead-in stage of a phase 3 study. Am J Hematol. 2022;97(2):174–84.
Basiorka AA, McGraw KL, Eksioglu EA, Chen X, Johnson J, Zhang L, et al. The NLRP3 inflammasome functions as a driver of the myelodysplastic syndrome phenotype. Blood. 2016;128(25):2960–75.
McLemore AF, Hou HA, Meyer BS, Lam NB, Ward GA, Aldrich AL, et al. Somatic gene mutations expose cytoplasmic DNA to co-opt the cGAS/STING/NLRP3 axis in myelodysplastic syndromes. JCI Insight. 2022;7(15):e159430.
Sallman DA, List A. The central role of inflammatory signaling in the pathogenesis of myelodysplastic syndromes. Blood. 2019;133(10):1039–48.
Basiorka AA, McGraw KL, Abbas-Aghababazadeh F, McLemore AF, Vincelette ND, Ward GA, et al. Assessment of ASC specks as a putative biomarker of pyroptosis in myelodysplastic syndromes: an observational cohort study. Lancet Haematol. 2018;5(9):e393-402.
Garcia-Manero G, Jabbour EJ, Konopleva MY, Daver NG, Borthakur G, DiNardo CD, et al. A clinical study of tomaralimab (OPN-305), a toll-like receptor 2 (TLR-2) antibody, in heavily pre-treated transfusion dependent patients with lower risk myelodysplastic syndromes (MDS) that have received and failed on prior hypomethylating agent (HMA) therapy. Blood. 2018;132(Supplement 1):798.
Seiler M, Yoshimi A, Darman R, Chan B, Keaney G, Thomas M, et al. H3B–8800, an orally available small-molecule splicing modulator, induces lethality in spliceosome-mutant cancers. Nat Med. 2018;24(4):497–504.
Steensma DP, Wermke M, Klimek VM, Greenberg PL, Font P, Komrokji RS, et al. Phase I first-in-human dose escalation study of the oral SF3B1 modulator H3B–8800 in myeloid neoplasms. Leukemia. 2021;35(12):3542–50.
Fong JY, Pignata L, Goy PA, Kawabata KC, Lee SCW, Koh CM, et al. Therapeutic targeting of RNA splicing catalysis through inhibition of protein arginine methylation. Cancer Cell. 2019;36(2):194-209.e9.
Schwartz LY, Choudhary GS, Ramachandra N, Sahu S, Gordon S, Ruggeri B, et al. Preclinical activity of the clinical stage protein arginine methyltransferase 5 (PRMT5) inhibitor PRT543 in splicing mutant myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML). Blood. 2021;23(138):2597.
Watts JM, Bradley TJ, Thomassen A, Brunner AM, Minden MD, Papadantonakis N, et al. A phase I/II study to investigate the safety and clinical activity of the protein arginine methyltransferase 5 inhibitor GSK3326595 in subjects with myelodysplastic syndrome and acute myeloid leukemia. Blood. 2019;134(Supplement_1):2656.
Nguyen HD, Leong WY, Li W, Reddy PNG, Sullivan JD, Walter MJ, et al. Spliceosome mutations induce R loop-associated sensitivity to ATR inhibition in myelodysplastic syndromes. Cancer Res. 2018;78(18):5363–74.
Brunner A, Liu Y, Mendez L, Garcia J, Amrein P, Neuberg D, et al. P758: INHIBITION OF ATR WITH CERALASERTIB (AZD6738) FOR THE TREATMENT OF PROGRESSIVE OR RELAPSED MYELODYSPLASTIC SYNDROMES AND CHRONIC MYELOMONOCYTIC LEUKEMIA: A PHASE IB/II STUDY. HemaSphere. 2022;6:653.
Ambaglio I, Malcovati L, Papaemmanuil E, Laarakkers CM, Della Porta MG, Gallì A, et al. Inappropriately low hepcidin levels in patients with myelodysplastic syndrome carrying a somatic mutation of SF3B1. Haematologica. 2013;98(3):420–3.
Bondu S, Alary AS, Lefèvre C, Houy A, Jung G, Lefebvre T, et al. A variant erythroferrone disrupts iron homeostasis in SF3B1-mutated myelodysplastic syndrome. Sci Transl Med. 2019;11(500):eaav5467.
Malcovati L, Porta MGD, Pascutto C, Invernizzi R, Boni M, Travaglino E, et al. Prognostic factors and life expectancy in myelodysplastic syndromes classified according to WHO criteria: a basis for clinical decision making. J Clin Oncol. 2005;23(30):7594–603.
Malcovati L, Della Porta MG, Cazzola M. Predicting survival and leukemic evolution in patients with myelodysplastic syndrome. Haematologica. 2006;91(12):1588–90.
de Swart L, Reiniers C, Bagguley T, van Marrewijk C, Bowen D, Hellström-Lindberg E, et al. Labile plasma iron levels predict survival in patients with lower-risk myelodysplastic syndromes. Haematologica. 2018;103(1):69–79.
Pascal L, Beyne-Rauzy O, Brechignac S, Marechaux S, Vassilieff D, Ernst O, et al. Cardiac iron overload assessed by T2* magnetic resonance imaging and cardiac function in regularly transfused myelodysplastic syndrome patients. Br J Haematol. 2013;162(3):413–5.
Ho PJ, Hiwase D, Ramakrishna R, Viiala N, Solterbeck A, Traficante R, et al. Cardiac and hepatic siderosis in myelodysplastic syndrome, thalassemia and diverse causes of transfusion-dependent anemia: the TIMES study. Hemasphere. 2019;3(3):e224.
• Angelucci E, Li J, Greenberg P, Wu D, Hou M, Montano Figueroa EH, et al. Iron chelation in transfusion-dependent patients with low- to intermediate-1-risk myelodysplastic syndromes: a randomized trial. Ann Intern Med. 2020 Apr 21;172(8):513–22. This phase 2 study supported the use of iron chelation therapy in lower risk MDS patients with iron overload, given it led to a longer event-free survival compared to placebo with a manageable safety profile.
Vinchi F, Hell S, Platzbecker U. Controversies on the consequences of iron overload and chelation in MDS. Hemasphere. 2020;4(3):e357.
Brunner AM, Blonquist TM, Hobbs GS, Amrein PC, Neuberg DS, Steensma DP, et al. Risk and timing of cardiovascular death among patients with myelodysplastic syndromes. Blood Adv. 2017;1(23):2032–40.
Adrianzen Herrera D, Pradhan K, Snyder R, Karanth S, Janakiram M, Mantzaris I, et al. Myelodysplastic syndromes and the risk of cardiovascular disease in older adults: a SEER-medicare analysis. Leukemia. 2020;34(6):1689–93.
Dayyani F, Conley AP, Strom SS, Stevenson W, Cortes JE, Borthakur G, et al. Cause of death in patients with lower-risk myelodysplastic syndrome. Cancer. 2010;116(9):2174–9.
Fuster JJ, MacLauchlan S, Zuriaga MA, Polackal MN, Ostriker AC, Chakraborty R, et al. Clonal hematopoiesis associated with TET2 deficiency accelerates atherosclerosis development in mice. Science. 2017;355(6327):842–7.
Jaiswal S, Natarajan P, Silver AJ, Gibson CJ, Bick AG, Shvartz E, et al. Clonal hematopoiesis and risk of atherosclerotic cardiovascular disease. N Engl J Med. 2017;377(2):111–21.
Jaiswal S, Fontanillas P, Flannick J, Manning A, Grauman PV, Mar BG, et al. Age-related clonal hematopoiesis associated with adverse outcomes. N Engl J Med. 2014;371(26):2488–98.
Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med. 2017;377(12):1119–31.
Svensson EC, Madar A, Campbell CD, He Y, Sultan M, Healey ML, et al. TET2-driven clonal hematopoiesis and response to canakinumab: an exploratory analysis of the CANTOS randomized clinical trial. JAMA Cardiol. 2022;7(5):521–8.
Hochman MJ, DeZern AE. Myelodysplastic syndrome and autoimmune disorders: two sides of the same coin? Lancet Haematol. 2022;9(7):e523–34.
• Beck DB, Ferrada MA, Sikora KA, Ombrello AK, Collins JC, Pei W, et al. Somatic mutations in UBA1 and severe adult-onset autoinflammatory disease. N Engl J Med. 2020 Dec 31;383(27):2628–38. This study identified an adult-onset severe autoinflammatory disorder associated with MDS, i.e., VEXAS syndrome, due to somatic UBA1 mutations in myeloid cells.
Comont T, Heiblig M, Rivière E, Terriou L, Rossignol J, Bouscary D, et al. Azacitidine for patients with Vacuoles, E1 Enzyme, X-linked, Autoinflammatory, Somatic syndrome (VEXAS) and myelodysplastic syndrome: data from the French VEXAS registry. Br J Haematol. 2022;196(4):969–74.
Diarra A, Duployez N, Fournier E, Preudhomme C, Coiteux V, Magro L, et al. Successful allogeneic hematopoietic stem cell transplantation in patients with VEXAS syndrome: a 2-center experience. Blood Adv. 2022;6(3):998–1003.
Heiblig M, Ferrada MA, Koster MJ, Barba T, Gerfaud-Valentin M, Mékinian A, et al. Ruxolitinib is more effective than other JAK inhibitors to treat VEXAS syndrome: a retrospective multicenter study. Blood. 2022;140(8):927–31.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
David A. Sallman: Shattuck Labs: Membership on an entity’s Board of Directors or advisory committees; Syndax: Membership on an entity’s Board of Directors or advisory committees; Magenta: Consultancy; Takeda: Consultancy; Kite: Membership on an entity’s Board of Directors or advisory committees; Bristol-Myers Squibb: Membership on an entity’s Board of Directors or advisory committees, Speakers Bureau; Incyte: Speakers Bureau; Novartis: Consultancy, Membership on an entity’s Board of Directors or advisory committees; Intellia: Membership on an entity’s Board of Directors or advisory committees; AbbVie: Membership on an entity’s Board of Directors or advisory committees; Agios: Membership on an entity’s Board of Directors or advisory committees; Aprea: Membership on an entity’s Board of Directors or advisory committees, Research Funding.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Leukemia
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, C., Sallman, D.A. Current Therapeutic Landscape in Lower Risk Myelodysplastic Syndromes. Curr. Treat. Options in Oncol. 24, 387–408 (2023). https://doi.org/10.1007/s11864-023-01062-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11864-023-01062-7