Opinion Statement
Molecular heterogeneity has confounded attempts to target individual pathways in brain tumors. However, gliomas with BRAF mutations have been identified as being uniquely vulnerable to targeted therapies. Such mutations are predominantly seen in brain tumors of the adolescent and young adult population. Given that accurate and timely identification of such mutations is essential for offering appropriate treatment, treatment centers should offer both immunohistochemical and sequencing methods for detection of these mutations to guide treatment. Additional studies of these tumors at recurrence would also allow identification of breakthrough resistance mechanisms that may also be targetable for treatment. Due to the relative rarity of these tumors, multicenter collaborative studies will be essential in achieving long term control of these tumors.

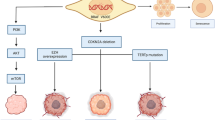
Reproduced from Zaman et al. Cancers 2019, 11(8), 1197, without modification under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/)

Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Davies H, Bignell GR, Cox C, et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417(6892):949–54. https://doi.org/10.1038/nature00766 (published Online First: 18 June 2002).
Chong H, Vikis HG, Guan KL. Mechanisms of regulating the Raf kinase family. Cell Signal. 2003;15(5):463–9. https://doi.org/10.1016/s0898-6568(02)00139-0 (published Online First: 18 March 2003).
El-Osta H, Falchook G, Tsimberidou A, et al. BRAF mutations in advanced cancers: clinical characteristics and outcomes. PLoS ONE. 2011;6(10): e25806. https://doi.org/10.1371/journal.pone.0025806 (published Online First: 1 Nov 2011).
Tiacci E, Trifonov V, Schiavoni G, et al. BRAF mutations in hairy-cell leukemia. N Engl J Med. 2011;364(24):2305–15. https://doi.org/10.1056/NEJMoa1014209 (published Online First: 15 June 2011).
Pritchard CA, Samuels ML, Bosch E, et al. Conditionally oncogenic forms of the A-Raf and B-Raf protein kinases display different biological and biochemical properties in NIH 3T3 cells. Mol Cell Biol. 1995;15(11):6430–42. https://doi.org/10.1128/mcb.15.11.6430 (published Online First: 1 Nov 1995).
Marais R, Light Y, Paterson HF, et al. Differential regulation of Raf-1, A-Raf, and B-Raf by oncogenic ras and tyrosine kinases. J Biol Chem. 1997;272(7):4378–83. https://doi.org/10.1074/jbc.272.7.4378 (published Online First: 14 Feb 1997).
Daum G, Eisenmann-Tappe I, Fries HW, et al. The ins and outs of Raf kinases. Trends Biochem Sci. 1994;19(11):474–80. https://doi.org/10.1016/0968-0004(94)90133-3 (published Online First: 1 Nov 1994).
Pearson G, Robinson F, Beers Gibson T, et al. Mitogen-activated protein (MAP) kinase pathways: regulation and physiological functions. Endocr Rev. 2001;22(2):153–83. https://doi.org/10.1210/edrv.22.2.0428 (published Online First: 11 April 2001).
Tran NH, Wu X, Frost JA. B-Raf and Raf-1 are regulated by distinct autoregulatory mechanisms. J Biol Chem. 2005;280(16):16244–53. https://doi.org/10.1074/jbc.M501185200 (published Online First: 16 Feb 2005).
• Yao Z, Torres NM, Tao A, et al. BRAF mutants evade ERK-dependent feedback by different mechanisms that determine their sensitivity to pharmacologic inhibition. Cancer Cell. 2015;28(3):370–83. https://doi.org/10.1016/j.ccell.2015.08.001 (published Online First: 8 Sep 2015). Identified mechanisms of resistance of BRAF mutant malignant cells to BRAF inhibitors.
Yao Z, Yaeger R, Rodrik-Outmezguine VS, et al. Tumours with class 3 BRAF mutants are sensitive to the inhibition of activated RAS. Nature. 2017;548(7666):234–8. https://doi.org/10.1038/nature23291 (published Online First: 8 Aug 2017).
Cutler RE Jr, Stephens RM, Saracino MR, et al. Autoregulation of the Raf-1 serine/threonine kinase. Proc Natl Acad Sci USA. 1998;95(16):9214–9. https://doi.org/10.1073/pnas.95.16.9214 (published Online First: 5 Aug 1998).
Jones DT, Kocialkowski S, Liu L, et al. Tandem duplication producing a novel oncogenic BRAF fusion gene defines the majority of pilocytic astrocytomas. Cancer Res. 2008;68(21):8673–7. https://doi.org/10.1158/0008-5472.CAN-08-2097 (published Online First: 1 Nov 2008).
Wan PT, Garnett MJ, Roe SM, et al. Mechanism of activation of the RAF-ERK signaling pathway by oncogenic mutations of B-RAF. Cell. 2004;116(6):855–67. https://doi.org/10.1016/s0092-8674(04)00215-6 (published Online First: 24 March 2004).
Ikenoue T, Hikiba Y, Kanai F, et al. Functional analysis of mutations within the kinase activation segment of B-Raf in human colorectal tumors. Cancer Res. 2003;63(23):8132–7 (published Online First: 18 Dec 2003).
Schindler G, Capper D, Meyer J, et al. Analysis of BRAF V600E mutation in 1,320 nervous system tumors reveals high mutation frequencies in pleomorphic xanthoastrocytoma, ganglioglioma and extra-cerebellar pilocytic astrocytoma. Acta Neuropathol. 2011;121(3):397–405. https://doi.org/10.1007/s00401-011-0802-6 (published Online First: 29 Jan 2011).
Behling F, Barrantes-Freer A, Skardelly M, et al. Frequency of BRAF V600E mutations in 969 central nervous system neoplasms. Diagn Pathol. 2016;11(1):55. https://doi.org/10.1186/s13000-016-0506-2 (published Online First: 29 June 2016).
Johnson A, Severson E, Gay L, et al. Comprehensive genomic profiling of 282 pediatric low- and high-grade gliomas reveals genomic drivers, tumor mutational burden, and hypermutation signatures. Oncologist. 2017;22(12):1478–90. https://doi.org/10.1634/theoncologist.2017-0242 (published Online First: 16 Sep 2017).
• Lassaletta A, Zapotocky M, Mistry M, et al. Therapeutic and prognostic implications of BRAF V600E in pediatric low-grade gliomas. J Clin Oncol. 2017;35(25):2934–41. https://doi.org/10.1200/JCO.2016.71.8726 (published Online First: 21 July 2017). Important study that identified the efficacy of BRAF inhibitors against pediatric BRAF mutant gliomas.
Zhang J, Wu G, Miller CP, et al. Whole-genome sequencing identifies genetic alterations in pediatric low-grade gliomas. Nat Genet. 2013;45(6):602–12. https://doi.org/10.1038/ng.2611 (published Online First: 16 April 2013).
Sugiura Y, Nagaishi M. Clinical relevance of BRAF status in glial and glioneuronal tumors: a systematic review. J Clin Neurosci. 2019;66:196–201. https://doi.org/10.1016/j.jocn.2019.05.014 (published Online First: 31 May 2019).
•• Brastianos PK, Taylor-Weiner A, Manley PE, et al. Exome sequencing identifies BRAF mutations in papillary craniopharyngiomas. Nat Genet. 2014;46(2):161–5. https://doi.org/10.1038/ng.2868 (published Online First: 15 Jan 2014). Important paper that reported high frequency of BRAF mutations in papillary craniopharyngiomas which has opened new avenues for treatment of these rare tumors.
La Corte E, Younus I, Pivari F, et al. BRAF V600E mutant papillary craniopharyngiomas: a single-institutional case series. Pituitary. 2018;21(6):571–83. https://doi.org/10.1007/s11102-018-0909-z (published Online First: 7 July 2018).
Cerami E, Gao J, Dogrusoz U, et al. The cBio Cancer Genomics Portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012;2(5):401–4. https://doi.org/10.1158/2159-8290.CD-12-0095 (published Online First: 17 May 2012).
Gao J, Aksoy BA, Dogrusoz U, et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal. 2013;6(269):pl1. https://doi.org/10.1126/scisignal.2004088 (published Online First: 4 April 2013).
Ascierto PA, McArthur GA, Dreno B, et al. Cobimetinib combined with vemurafenib in advanced BRAF(V600)-mutant melanoma (coBRIM): updated efficacy results from a randomised, double-blind, phase 3 trial. Lancet Oncol. 2016;17(9):1248–60. https://doi.org/10.1016/S1470-2045(16)30122-X (published Online First: 3 Aug 2016).
Planchard D, Smit EF, Groen HJM, et al. Dabrafenib plus trametinib in patients with previously untreated BRAF(V600E)-mutant metastatic non-small-cell lung cancer: an open-label, phase 2 trial. Lancet Oncol. 2017;18(10):1307–16. https://doi.org/10.1016/S1470-2045(17)30679-4 (published Online First: 19 Sep 2017).
Diamond EL, Subbiah V, Lockhart AC, et al. Vemurafenib for BRAF V600-mutant Erdheim-Chester disease and Langerhans cell histiocytosis: analysis of data from the histology-independent, phase 2, open-label VE-BASKET study. JAMA Oncol. 2018;4(3):384–8. https://doi.org/10.1001/jamaoncol.2017.5029 (published Online First: 1 Dec 2017).
Bouffet E, Kieran M, Hargrave D, et al. LGG-46: trametinib therapy in pediatric patients with low-grade gliomas (LGG) with BRAF gene fusion: a disease-specific cohort in the first pediatric testing of trametinib. Neuro-Oncology. 2018;20(Suppl 2):i114–214. https://doi.org/10.1093/neuonc/noy059.387 (published Online First: 22 June 2018).
• Kaley T, Touat M, Subbiah V, et al. BRAF inhibition in BRAF(V600)-mutant gliomas: results from the VE-BASKET study. J Clin Oncol. 2018;36(35):3477–84. https://doi.org/10.1200/JCO.2018.78.9990 (published Online First: 24 Oct 2018). Important study that identified the efficacy of BRAF inhibitors against BRAF mutantgliomas.
Wen P, Alexander S, Yung-Jue B, et al. RARE-09: efficacy and safety of dabrafenib + trametinib in patients with recurrent/refractory BRAF V600E-mutated high-grade glioma (HGG). Neuro-Oncology. 2018;20(Suppl 6):vi238–338. https://doi.org/10.1093/neuonc/noy148.986 (published Online First: 5 Nov 2018).
Garnier L, Ducray F, Verlut C, et al. Prolonged response induced by single agent vemurafenib in a BRAF V600E spinal ganglioglioma: a case report and review of the literature. Front Oncol. 2019;9:177. https://doi.org/10.3389/fonc.2019.00177 (published Online First: 16 April 2019).
Kieran MW, Bouffet E, Tabori U, et al. LBA19_PR-CNS tumours: the first study of dabrafenib in pediatric patients with BRAF V600-mutant relapsed or refractory low-grade gliomas. Ann Oncol. 2016;27:vi557. https://doi.org/10.1093/annonc/mdw435.09.
• Fangusaro J, Onar-Thomas A, Young Poussaint T, et al. Selumetinib in paediatric patients with BRAF-aberrant or neurofibromatosis type 1-associated recurrent, refractory, or progressive low-grade glioma: a multicentre, phase 2 trial. Lancet Oncol. 2019;20(7):1011–22. https://doi.org/10.1016/S1470-2045(19)30277-3 (published Online First: 4 June 2019). Demonstrates the efficacy of MEK inhibition in treating BRAF aberrant and NF1-associated low-grade gliomas.
Drobysheva A, Klesse LJ, Bowers DC, et al. Targeted MAPK pathway inhibitors in patients with disseminated pilocytic astrocytomas. J Natl Compr Cancer Netw. 2017;15(8):978–82. https://doi.org/10.6004/jnccn.2017.0139 (published Online First: 9 Aug 2017).
Long GV, Trefzer U, Davies MA, et al. Dabrafenib in patients with Val600Glu or Val600Lys BRAF-mutant melanoma metastatic to the brain (BREAK-MB): a multicentre, open-label, phase 2 trial. Lancet Oncol. 2012;13(11):1087–95. https://doi.org/10.1016/S1470-2045(12)70431-X (published Online First: 12 Oct 2012).
Dummer R, Goldinger SM, Turtschi CP, et al. Vemurafenib in patients with BRAF(V600) mutation-positive melanoma with symptomatic brain metastases: final results of an open-label pilot study. Eur J Cancer. 2014;50(3):611–21. https://doi.org/10.1016/j.ejca.2013.11.002 (published Online First: 4 Dec 2013).
•• Davies MA, Saiag P, Robert C, et al. Dabrafenib plus trametinib in patients with BRAF(V600)-mutant melanoma brain metastases (COMBI-MB): a multicentre, multicohort, open-label, phase 2 trial. Lancet Oncol. 2017;18(7):863–73. https://doi.org/10.1016/S1470-2045(17)30429-1 (published Online First: 9 June 2017). Breakthrough paper that demonstrates the effectiveness of combined BRAF and MEK inhibition in BRAF mutant melanoma brain metastases.
Yamamoto G, Sakakibara-Konishi J, Ikari T, et al. Response of BRAF(V600E)-mutant lung adenocarcinoma with brain metastasis and leptomeningeal dissemination to dabrafenib plus trametinib treatment. J Thorac Oncol. 2019;14(5):e97–9. https://doi.org/10.1016/j.jtho.2018.12.027 (published Online First: 28 April 2019).
Robinson SD, O’Shaughnessy JA, Cowey CL, et al. BRAF V600E-mutated lung adenocarcinoma with metastases to the brain responding to treatment with vemurafenib. Lung Cancer. 2014;85(2):326–30. https://doi.org/10.1016/j.lungcan.2014.05.009 (published Online First: 4 June 2014).
Holderfield M, Deuker MM, McCormick F, et al. Targeting RAF kinases for cancer therapy: BRAF-mutated melanoma and beyond. Nat Rev Cancer. 2014;14(7):455–67. https://doi.org/10.1038/nrc3760 (published Online First: 25 June 2014).
Pratt D, Camelo-Piragua S, McFadden K, et al. BRAF activating mutations involving the beta3-alphaC loop in V600E-negative anaplastic pleomorphic xanthoastrocytoma. Acta Neuropathol Commun. 2018;6(1):24. https://doi.org/10.1186/s40478-018-0525-1 (published Online First: 17 March 2018).
•• Foster SA, Whalen DM, Ozen A, et al. Activation mechanism of oncogenic deletion mutations in BRAF, EGFR, and HER2. Cancer Cell. 2016;29(4):477–93. https://doi.org/10.1016/j.ccell.2016.02.010 (published Online First: 22 March 2016). Identified the major mechanism by which alterations in BRAF activate oncogenesis.
Wang J, Yao Z, Jonsson P, et al. A secondary mutation in BRAF confers resistance to RAF inhibition in a BRAF(V600E)-mutant brain tumor. Cancer Discov. 2018;8(9):1130–41. https://doi.org/10.1158/2159-8290.CD-17-1263 (published Online First: 9 June 2018).
Tse A, Verkhivker GM. Exploring molecular mechanisms of paradoxical activation in the BRAF kinase dimers: atomistic simulations of conformational dynamics and modeling of allosteric communication networks and signaling pathways. PLoS ONE. 2016;11(11): e0166583. https://doi.org/10.1371/journal.pone.0166583 (published Online First: 20 Nov 2016).
Khater F, Langlois S, Cassart P, et al. Recurrent somatic BRAF insertion (p.V504_R506dup): a tumor marker and a potential therapeutic target in pilocytic astrocytoma. Oncogene. 2019;38(16):2994–3002. https://doi.org/10.1038/s41388-018-0623-3 (published Online First: 24 Dec 2018).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Appaji Rayi, Iyad Alnahhas, Shirley Ong and Vinay K. Puduvalli declare that they have no conflict of interest. Pierre Giglio owns stock in Vanguard S&P 500 ETF.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Neuro-oncology
Rights and permissions
About this article
Cite this article
Rayi, A., Alnahhas, I., Ong, S. et al. Targeted Therapy for BRAF Mutant Brain Tumors. Curr. Treat. Options in Oncol. 22, 105 (2021). https://doi.org/10.1007/s11864-021-00901-9
Accepted:
Published:
DOI: https://doi.org/10.1007/s11864-021-00901-9