Abstract
Objective
Acute coronary syndrome (ACS) is an emergency and severe disorder of the cardiovascular system. This paper assessed the expression of plasma HAND2-AS1 in patients with ACS, researched its diagnostic and prognostic significance, and studied its possible mechanism for participating in ACS.
Methods
The concentration of HAND2-AS1 of 101 included patients with ACS was certified by qRT-PCR and its possible diagnostic function was revealed by the receiver operating characteristic (ROC) curve. All patients were followed up for 6 months after percutaneous coronary intervention (PCI) therapy and Kaplan-Meier (K-M) curve and COX regression analysis was performed to estimate the short-term prognostic value of HAND2-AS1 in ACS. The interrelationship between HAND2-AS1 and Gensini score and endothelial injury was identified via Pearson correlation. The function of HAND2-AS1 on the viability, migration, and apoptosis of human umbilical vein endothelial cells (HUVECs) was estimated by the Cell Counting Kit-8 (CCK-8), Transwell chamber, and flow cytometry.
Results
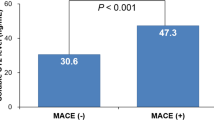
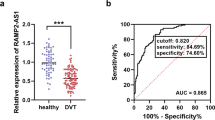
In ACS patients, the expression of serum HAND2-AS1 was prominently decreased and closely correlated with the Gensini score. The decreased HAND2-AS1 expression was of diagnostic significance. Declined plasma HAND2-AS1 was observed in patients with the major adverse cardio-cerebrovascular event (MACCE) and was an independent risk for the poor prognosis of ACS patients. In the cell model, upregulation of HAND2-AS1 improved cell viability and migration and inhibited cell apoptosis.
Conclusion
HAND2-AS1 was an independent biomarker for the diagnosis and prognosis of ACS. HAND2-AS1 might be involved in ACS development by regulating endothelial damage.





Similar content being viewed by others
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Bergmark BA, Mathenge N, Lawrence-Wright MB et al (2022) Acute coronary syndromes. Lancet 399:1347–1358. https://doi.org/10.1016/s0140-6736(21)02391-6
Harskamp RE, Fanaroff AC, Zhen SW et al (2022) Recognising acute coronary syndrome. BMJ 377:e069591. https://doi.org/10.1136/bmj-2021-069591
Shirasaki K, Minai K, Kawai M et al (2022) Unique crosstalk between platelet and leukocyte counts during treatment for acute coronary syndrome: A retrospective observational study. Medicine 101:e32439. https://doi.org/10.1097/md.0000000000032439
Fang Y, Cheng X, Peng W et al (2022) Atypical manifestations of acute coronary syndrome - throat discomfort: A multi-center observational study. Front Med 16:651–658. https://doi.org/10.1007/s11684-021-0859-0
Zhang LS, Liu SQ, Xie XL et al (2021) An improved networked predictive controller for vascular robot using 5G networks. In: 2021 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, p 4674–4678. https://doi.org/10.1109/embc46164.2021.9630311
Wang B, Jinsong L, Hong Y et al (2022) Effect of cardiac rehabilitation therapy combined with WeChat platform education on patients with unstable angina pectoris after PCI. J Healthc Eng 2022:7253631. https://doi.org/10.1155/2022/7253631
Xu H, Zhang J, Jia H et al (2023) Serum histone deacetylase 4 longitudinal change for estimating major adverse cardiovascular events in acute coronary syndrome patients receiving percutaneous coronary intervention. Ir J Med Sci. https://doi.org/10.1007/s11845-023-03326-5
Balaji Srinivasan S, Sehly A, Jaltotage B et al (2022) Short-term DAPT after coronary stenting has similar ischemic and bleeding outcomes as long-term DAPT: A 5-year population-based cohort study. Ir J Med Sci. https://doi.org/10.1007/s11845-022-03171-y
Uchida S, Dimmeler S (2015) Long noncoding RNAs in cardiovascular diseases. Circ Res 116:737–750. https://doi.org/10.1161/circresaha.116.302521
Cao M, Luo H, Li D et al (2022) Research advances on circulating long noncoding RNAs as biomarkers of cardiovascular diseases. Int J Cardiol 353:109–117. https://doi.org/10.1016/j.ijcard.2022.01.070
Chen L, Huang Y (2022) High expression of lncRNA PELATON serves as a risk factor for the incidence and prognosis of acute coronary syndrome. Sci Rep 12:8030. https://doi.org/10.1038/s41598-022-11260-2
Libby P, Pasterkamp G, Crea F, Jang IK (2019) Reassessing the mechanisms of acute coronary syndromes. Circ Res 124:150–160. https://doi.org/10.1161/circresaha.118.311098
Liu X, Li S, Yang Y et al (2021) The lncRNA ANRIL regulates endothelial dysfunction by targeting the let-7b/TGF-βR1 signalling pathway. J Cell Physiol 236:2058–2069. https://doi.org/10.1002/jcp.29993
Ma L, He S, Li H et al (2022) HAND2-AS1 targeting miR-1208/SIRT1 axis alleviates foam cell formation in atherosclerosis. Int J Cardiol 346:53–61. https://doi.org/10.1016/j.ijcard.2021.11.019
Kim M, Kang SH, Kim JR et al (2020) Comparison of shear stress-induced thrombotic and thrombolytic effects among 3 different antithrombotic regimens in patients with acute coronary syndrome. Clin Appl Thromb Hemost 26:1076029620912814. https://doi.org/10.1177/1076029620912814
张新超, 于学忠, 陈凤英, 朱华栋 (2019) 急性冠脉综合征急诊快速诊治指南(2019). 临床急诊杂志:10
Rodrigues AF, Rebelo C, Simões S et al (2023) A polymeric nanoparticle formulation for targeted mRNA delivery to fibroblasts. Adv Sci 10:e2205475. https://doi.org/10.1002/advs.202205475
Atwood J (2022) Management of acute coronary syndrome. Emerg Med Clin North Am 40:693–706. https://doi.org/10.1016/j.emc.2022.06.008
Shin M, Mun S, Park SH et al (2021) Serum biomarker discovery related to pathogenesis in acute coronary syndrome by proteomic approach. Biosci Rep 41. https://doi.org/10.1042/bsr20210344
Wang L, Jin Y (2020) Noncoding RNAs as biomarkers for acute coronary syndrome. Biomed Res Int 2020:3298696. https://doi.org/10.1155/2020/3298696
Batra G, Lindbäck J, Becker RC et al (2022) Biomarker-based prediction of recurrent ischemic events in patients with acute coronary syndromes. J Am Coll Cardiol 80:1735–1747. https://doi.org/10.1016/j.jacc.2022.08.767
Li D, Ma Y, Deng W, Feng J (2022) Construction and analysis of lncRNA-associated ceRNA network in atherosclerotic plaque formation. Biomed Res Int 2022:4895611. https://doi.org/10.1155/2022/4895611
Bampatsias D, Mavroeidis I, Tual-Chalot S et al (2022) Beta-secretase-1 antisense RNA is associated with vascular ageing and atherosclerotic cardiovascular disease. Thromb Haemost 122:1932–1942. https://doi.org/10.1055/a-1914-2094
Wen C, Li B, Nie L et al (2022) Emerging roles of extracellular vesicle-delivered circular RNAs in atherosclerosis. Front Cell Dev Biol 10:804247. https://doi.org/10.3389/fcell.2022.804247
Gao R, Wang L, Bei Y et al (2021) Long noncoding RNA cardiac physiological hypertrophy-associated regulator induces cardiac physiological hypertrophy and promotes functional recovery after myocardial ischemia-reperfusion injury. Circulation 144:303–317. https://doi.org/10.1161/circulationaha.120.050446
Xue Q, Yang L, Wang J et al (2022) lncRNA ROR and miR-125b predict the prognosis in heart failure combined acute renal failure. Dis Markers 2022:6853939. https://doi.org/10.1155/2022/6853939
Yan L, Zhang Y, Zhang W et al (2020) lncRNA-NRF is a potential biomarker of heart failure after acute myocardial infarction. J Cardiovasc Transl Res 13:1008–1015. https://doi.org/10.1007/s12265-020-10029-0
Zheng PF, Chen LZ, Liu P, Pan HW (2022) A novel lncRNA-miRNA-mRNA triple network identifies lncRNA XIST as a biomarker for acute myocardial infarction. Aging 14:4085–4106. https://doi.org/10.18632/aging.204075
Ateş AH, Arslan U, Aksakal A et al (2018) Plasma chemerin levels are increased in ST elevation myocardial infarction patients with high thrombus burden. Cardiol Res Pract 2018:5812704. https://doi.org/10.1155/2018/5812704
Zhang W, Liu B, Wang Y et al (2022) miR-195–3p/BDNF axis regulates hypoxic injury by targeting P-ERK1/2 expression. Medicine 101:e31586. https://doi.org/10.1097/md.0000000000031586
Sanz-Rubio D, Khalyfa A, Qiao Z et al (2021) Cell-selective altered cargo properties of extracellular vesicles following in vitro exposures to intermittent hypoxia. Int J Mol Sci 22. https://doi.org/10.3390/ijms22115604
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
All individuals signed informed consent, and the study was authorized by the ethics committee of Beijing Tongren Hospital, Capital Medical University.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, J., Xu, R., Cao, Q. et al. HAND2-AS1 associates with outcomes of acute coronary syndrome and regulates cell viability of vascular endothelial cells. Ir J Med Sci 193, 131–138 (2024). https://doi.org/10.1007/s11845-023-03466-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-023-03466-8