Abstract
Background
Long noncoding RNA HULC (lnc-HULC) and its target microRNA-128-3p (miR-128-3p) regulate endothelial cell function, blood lipid level, and inflammatory cytokine production, which are involved in the pathogenesis of coronary heart disease (CHD). Based on the above information, this study intended to further investigate the correlation between lnc-HULC and miR-128-3p, as well as their clinical values for CHD management.
Methods
Totally, 141 CHD patients and 70 controls were enrolled. Lnc-HULC and miR-128-3p in peripheral blood mononuclear cells were detected by reverse transcription quantitative polymerase chain reaction (RT-qPCR). Serum inflammatory cytokines and cell adhesion molecules were further determined by enzyme-linked immunosorbent assay (ELISA) in CHD patients.
Results
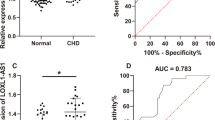
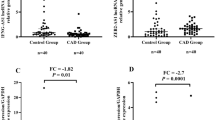
Lnc-HULC was upregulated, while miR-128-3p was downregulated in CHD patients than in controls (both P < 0.001). The ROC curve further displayed that lnc-HULC (AUC: 0.906, 95% CI: 0.867–0.945) and miR-128-3p (AUC: 0.814, 95% CI: 0.756–0.873) had the potential of discriminating CHD patients from controls. Regarding the correlation between lnc-HULC and miR-128-3p, lnc-HULC was negatively associated with miR-128-3p in CHD patients (rs = − 0.307, P < 0.001), but this association was not observed in controls (rs = − 0.155, P = 0.199). Furthermore, it was discovered that upregulated lnc-HULC was associated with elevated blood lipid levels (TG, LDL-C), inflammatory cytokines (interleukin (IL)-1β, IL-17A), cell adhesion molecules (VCAM-1), and Gensini score (all P < 0.05) in CHD patients. Meanwhile, miR-128-3p was negatively associated with blood lipid level (LDL-C), inflammatory cytokines (TNF-α, IL-1β, IL-6), cell adhesion molecules (VCAM-1, ICAM-1), and Gensini score (all P < 0.05) in CHD patients.
Conclusion
Lnc-HULC and its target miR-128-3p relate to lipid level, stenosis degree, inflammatory cytokines, and cell adhesion molecules in CHD patients.




Similar content being viewed by others
References
Makki N, Brennan TM, Girotra S (2015) Acute coronary syndrome. J Intensive Care Med 30(4):186–200. https://doi.org/10.1177/0885066613503294
Severino P, D'Amato A, Pucci M et al (2020) Ischemic heart disease pathophysiology paradigms overview: from plaque activation to microvascular dysfunction. Int J Mol Sci 21 (21). https://doi.org/10.3390/ijms21218118
Ma LY, Chen WW, Gao RL et al (2020) China cardiovascular diseases report 2018: an updated summary. J Geriatr Cardiol 17(1):1–8. https://doi.org/10.11909/j.issn.1671-5411.2020.01.001
Smith JN, Negrelli JM, Manek MB et al (2015) Diagnosis and management of acute coronary syndrome: an evidence-based update. J Am Board Fam Med 28(2):283–293. https://doi.org/10.3122/jabfm.2015.02.140189
Jia S, Liu Y, Yuan J (2020) Evidence in guidelines for treatment of coronary artery disease. Adv Exp Med Biol 1177:37–73. https://doi.org/10.1007/978-981-15-2517-9_2
Andreou I, Stone PH, Ikonomidis I et al (2020) Recurrent atherosclerosis complications as a mechanism for stent failure. Hellenic J Cardiol 61(1):9–14. https://doi.org/10.1016/j.hjc.2019.04.007
Patel MR, Norgaard BL, Fairbairn TA et al (2020) 1-year impact on medical practice and clinical outcomes of FFRCT: the ADVANCE Registry. JACC Cardiovasc Imaging 13(1 Pt 1):97–105. https://doi.org/10.1016/j.jcmg.2019.03.003
Kosmas N, Manolis AS, Dagres N et al (2020) Myocardial infarction or acute coronary syndrome with non-obstructive coronary arteries and sudden cardiac death: a missing connection. Europace 22(9):1303–1310. https://doi.org/10.1093/europace/euaa156
Pouralijan Amiri M, Khoshkam M, Salek RM et al (2019) Metabolomics in early detection and prognosis of acute coronary syndrome. Clin Chim Acta 495:43–53. https://doi.org/10.1016/j.cca.2019.03.1632
Chen X, Song D (2020) LPS promotes the progression of sepsis by activation of lncRNA HULC/miR-204-5p/TRPM7 network in HUVECs. Biosci Rep 40 (6). https://doi.org/10.1042/BSR20200740
Liang H, Li F, Li H et al (2021) Overexpression of lncRNA HULC attenuates myocardial ischemia/reperfusion injury in rat models and apoptosis of hypoxia/reoxygenation cardiomyocytes via targeting miR-377-5p through NLRP3/Caspase1/IL1beta signaling pathway inhibition. Immunol Invest 50(8):925–938. https://doi.org/10.1080/08820139.2020.1791178
Cui M, Xiao Z, Wang Y et al (2015) Long noncoding RNA HULC modulates abnormal lipid metabolism in hepatoma cells through an miR-9-mediated RXRA signaling pathway. Cancer Res 75(5):846–857. https://doi.org/10.1158/0008-5472.CAN-14-1192
Yang W, Luo X, Liu Y et al (2020) Potential role of lncRNA HULC/miR1283p/RAC1 axis in the inflammatory response during LPSinduced sepsis in HMEC1 cells. Mol Med Rep 22(6):5095–5104. https://doi.org/10.3892/mmr.2020.11601
Yang P, Han J, Li S et al (2021) miR-128-3p inhibits apoptosis and inflammation in LPS-induced sepsis by targeting TGFBR2. Open Med (Wars) 16(1):274–283. https://doi.org/10.1515/med-2021-0222
Geovanini GR, Libby P (2018) Atherosclerosis and inflammation: overview and updates. Clin Sci (Lond) 132(12):1243–1252. https://doi.org/10.1042/CS20180306
Gimbrone MA Jr, Garcia-Cardena G (2016) Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circ Res 118(4):620–636. https://doi.org/10.1161/CIRCRESAHA.115.306301
Pan S, Cui Y, Fu Z et al (2019) MicroRNA-128 is involved in dexamethasone-induced lipid accumulation via repressing SIRT1 expression in cultured pig preadipocytes. J Steroid Biochem Mol Biol 186:185–195. https://doi.org/10.1016/j.jsbmb.2018.10.013
Chandra A, Sharma K, Pratap K et al (2021) Inhibition of microRNA-128-3p attenuates hypercholesterolemia in mouse model. Life Sci 264:118633. https://doi.org/10.1016/j.lfs.2020.118633
Yan P, Sun C, Ma J et al (2019) MicroRNA-128 confers protection against cardiac microvascular endothelial cell injury in coronary heart disease via negative regulation of IRS1. J Cell Physiol 234(8):13452–13463. https://doi.org/10.1002/jcp.28025
Zhao X, Jin Y, Li L et al (2019) MicroRNA-128-3p aggravates doxorubicin-induced liver injury by promoting oxidative stress via targeting Sirtuin-1. Pharmacol Res 146:104276. https://doi.org/10.1016/j.phrs.2019.104276
Gensini GG (1983) A more meaningful scoring system for determining the severity of coronary heart disease. Am J Cardiol 51(3):606. https://doi.org/10.1016/s0002-9149(83)80105-2
Chen X, Zhang X, Su C et al (2020) Long noncoding RNA HULC in acute ischemic stroke: association with disease risk, severity, and recurrence-free survival and relation with IL-6, ICAM1, miR-9, and miR-195. J Clin Lab Anal 34(11):e23500. https://doi.org/10.1002/jcla.23500
Ebadi N, Ghafouri-Fard S, Taheri M et al (2020) Dysregulation of autophagy-related lncRNAs in peripheral blood of coronary artery disease patients. Eur J Pharmacol 867:172852. https://doi.org/10.1016/j.ejphar.2019.172852
Ghafouri-Fard S, Gholipour M, Taheri M (2021) The emerging role of long non-coding RNAs and circular RNAs in coronary artery disease. Front Cardiovasc Med 8:632393. https://doi.org/10.3389/fcvm.2021.632393
Ma Y, Huang D, Yang F et al (2016) Long noncoding RNA highly upregulated in liver cancer regulates the tumor necrosis factor-alpha-induced apoptosis in human vascular endothelial cells. DNA Cell Biol 35(6):296–300. https://doi.org/10.1089/dna.2015.3203
Liu S, Gao S, Yang Z et al (2021) miR-128-3p reduced acute lung injury induced by sepsis via targeting PEL12. Open Med (Wars) 16(1):1109–1120. https://doi.org/10.1515/med-2021-0258
Farina FM, Hall IF, Serio S et al (2020) miR-128-3p is a novel regulator of vascular smooth muscle cell phenotypic switch and vascular diseases. Circ Res 126(12):e120–e135. https://doi.org/10.1161/CIRCRESAHA.120.316489
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Consent to participate
All subjects signed the informed consents.
Consent for publication
The Institutional Review Board had given permission for this study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhu, X., Hu, J. & Xie, L. The interplay of long noncoding RNA HULC with microRNA-128-3p and their correlations with lipid level, stenosis degree, inflammatory cytokines, and cell adhesion molecules in coronary heart disease patients. Ir J Med Sci 191, 2597–2603 (2022). https://doi.org/10.1007/s11845-021-02900-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-021-02900-z