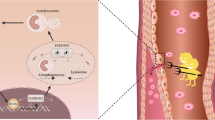
Abstract
Background:
Cardiovascular disorders pose great threat to public health. As a common type of cardiovascular disease, atherosclerosis is characterized by high morbidity and mortality/recurrence rate. However, the pathogenesis of atherosclerosis is complex and not fully understood. The aim of this study was to investigate the influences of hsa_circRNA_102541 (circ_102541) on proliferation and apoptosis of HUVEC cells and to identify the underlying mechanisms.
Methods:
RT-PCR was used to determine the expression levels of circ_102541, miR-296-5p, and PLK1 in atherosclerosis and healthy blood samples. Following the transfection with sh-circ_102541, LV-circ_102541, miR-296-5p mimics, miR-296-5p inhibitors, and si-PLK1, cell proliferation was evaluated using CCK8 assay; cell apoptosis was determined by flow cytometry; dual luciferase assay was performed to examine the interaction between abovementioned molecules. The levels of associated markers including PCNA and caspase-3 were assessed by western blotting and RT-qPCR.
Results:
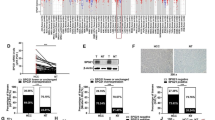
The expression of circRNA_102541 and PLK1 were significantly elevated in atherosclerosis specimens, where the level of miR-296-5p was reduced. Furthermore, circRNA_102541 could bind miR-296-5p and subsequently target PLK1. Following treatment with sh-circRNA_102541 or miR-296-5p mimics, proliferative ability and levels of PCNA were remarkably reduced in HUVEC cells, while apoptosis was significantly enhanced. Co-transfection with miR-296-5p mimics abrogated the effects induced by the overexpressed circ_102541. Additionally, treatment with si-PLK1 attenuated the biological behavior changes caused by miR-296-5p inhibitors in HUVEC cells. Moreover, transfection with LV-PLK1 reversed the effects triggered by miR-296-5p mimics.
Conclusion:
Taken together, circRNA_102541 was upregulated in atherosclerosis, and knockdown of circRNA_102541 suppressed cell proliferation while promoted apoptosis of HUVEC cells via miR-296-5p/PLK1. This novel pathway may serve essential roles on the development of atherosclerosis, and circRNA_102541 could be a promising therapeutic candidate for the treatment of atherosclerosis.




Similar content being viewed by others
Data availability
The datasets generated or analyzed during the present study are included in this published article.
Abbreviations
- HUVEC:
-
Human umbilical vein endothelial cells
- PCNA:
-
Proliferating cell nuclear antigen
- PLK1:
-
Polo-like kinase 1
References
Wong W (2017) Protected from atherosclerosis by TFEB. Science 355(6324):490
Qureshi A, Caplan L (2014) Intracranial atherosclerosis. Lancet 383(9921):984–998
Galkina E, Ley K (2009) Immune and inflammatory mechanisms of atherosclerosis. Annu Rev Immunol 27:165–197
Leischik R et al (2015) Physical activity, cardiorespiratory fitness and carotid intima thickness: sedentary occupation as risk factor for atherosclerosis and obesity. Eur Rev Med Pharmacol Sci 19:3157–3168
Memczak S et al (2013) Circular RNAs are a large class of animal RNAs with regulatory potency. Nature 495:333–338
Vicens Q, Westhof E (2014) Biogenesis of circular RNAs. Cell 159:13–14
Shen L et al (2019) CircRNA-0044073 is upregulated in atherosclerosis and increases the proliferation and invasion of cells by targeting miR-107. Mol Med Rep 19(5):3923–3932
Gong X et al (2019) Role of circular RNAs in cardiovascular disease. Exp Biol Med 244:73–82
Cui S et al (2018) Screening of up- and downregulation of circRNAs in HBV-related hepatocellular carcinoma by microarray. Oncol Lett 15:423–432
Ding S et al (2018) Circular RNAs in vascular functions and diseases. Adv Exp Med Biol 1087:287–297
Hansen T et al (2013) Natural RNA circles function as efficient microRNA sponges. Nature 495:384–388
Wang L, Yu W (2018) Association of circulating microRNA-122 with presence and severity of atherosclerotic lesions. PeerJ 6(2):e5218
He P, Zhao S (2018) Abnormally expressed miR-23b in Chinese Mongolian at high cardiovascular risk may contribute to monocyte/macrophage inflammatory reaction in atherosclerosis. Biosci Rep 38(6):BSR20180673
Li H et al (2018) MicroRNA-296: a promising target in the pathogenesis of atherosclerosis? Mol Med 24:12
Wang L, Shen C, Wang Y et al (2019) Identification of circular RNA Hsa_circ_0001879 and Hsa_circ_0004104 as novel biomarkers for coronary artery disease. Atherosclerosis 286:88–96
Fang M, Li Y, Wu Y et al (2019) MiR-185 silencing promotes the progression of atherosclerosis via targeting stromal interaction molecule 1. Cell Cycle 18:682–695
Huang R, Hu Z, Cao Y et al (2019) MiR-652-3p inhibition enhances endothelial repair and reduces atherosclerosis by promoting Cyclin D2 expression. EBioMedicine 40:685–694
Zhang F et al (2018) Comprehensive analysis of circRNA expression pattern and circRNA-miRNA-mRNA network in the pathogenesis of atherosclerosis in rabbits. Aging 10(9):2266–2283
Skuratovskaia D et al (2019) Promising directions in atherosclerosis treatment based on epigenetic regulation using microRNAs and long noncoding RNAs. Biomolecules 9(6):226
Gheghiani L et al (2017) PLK1 activation in late G2 sets up commitment to mitosis. Cell Rep 19:2060–2073
Xu C et al (2016) MiR-296-5p suppresses cell viability by directly targeting PLK1 in non-small cell lung cancer. Oncol Rep 35:497–503
Sur S et al (2016) Increased expression of phosphorylated polo-like kinase 1 and histone in bypass vein graft and coronary arteries following angioplasty. PLos One
Huang Y et al (2021) miRNA-296-5p functions as a potential tumor suppressor in human osteosarcoma by targeting SND1. Chin Med J (Engl) 134(5):564–572
Chen K et al (2012) MicroRNAs in atherosclerosis. Kaohsiung J Med Sci 28(12):631–640
De Carcer G et al (2018) Plk1 overexpression induces chromosomal instability and suppresses tumor development. Nat Commun
Yang Y, Xu X (2021) Bioinformatic identification of hub genes and related transcription factors in low shear stress treated endothelial cells. BMC Med Genomics
Author information
Authors and Affiliations
Contributions
DY initiated the present study. ND, ML, and DY carried out the experiments and data analysis. All the authors drafted the manuscript and approved the final version.
Corresponding author
Ethics declarations
Ethics approval
The present study was approved by the Ethics Committee of the First Affiliated Hospital of Jinzhou Medical University (Jinzhou, China).
Consent to participate
Written informed consents were signed by our patients.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Du, N., Li, M. & Yang, D. Hsa_circRNA_102541 regulates the development of atherosclerosis by targeting miR-296-5p/PLK1 pathway. Ir J Med Sci 191, 1153–1159 (2022). https://doi.org/10.1007/s11845-021-02708-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-021-02708-x