Abstract
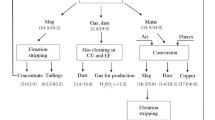
At the Kennecott Utah copper smelter in Magna, Utah, bleed streams from the refinery tankhouse and precious-metals plant are combined with smelter weak acid and electrostatic precipitator dust to produce leach solutions containing copper and impurities. Copper and arsenic are precipitated from the solutions as sulfides in a two-stage continuous process that enables excess arsenic to be removed from the circuit and routed to hazardous waste disposal as a highly concentrated material.
Similar content being viewed by others
References
P.J. Gabb et al., “The Kennecott Smelter Hydromet allurgical Impurities Process,” Proceedings of Copper ’95-Cobre ’95 International Conference, vol. 3, ed. W.C. Cooper et al. (Montreal, Canada: The Metallurgical Society of CIM, 1995), pp. 591–606.
R.H. Davies et al., “MTDATA—The NPL Databank for Metallurgical Thermochemistry,” User Aspects of Phase Diagrams: Proceedings of the International Conference, ed F.H. Hayes (London: Institute of Metals, 1990), pp. 140–152.
Author information
Authors and Affiliations
Additional information
For more information, contact P.J. Gabb, Rio Tinto Technical Services, P.O. Box 50, Castlemead, Lower Castle Street, Bristol BS99 7YR, United Kingdom; telephone 44-117-927-6407; fax 44-117-927-3317; e-mail phil.gabb@riotinto.com.
Rights and permissions
About this article
Cite this article
Gabb, P.J., Davies, A.L. The industrial separation of copper and arsenic as sulfides. JOM 51, 18–19 (1999). https://doi.org/10.1007/s11837-999-0151-2
Issue Date:
DOI: https://doi.org/10.1007/s11837-999-0151-2