Abstract
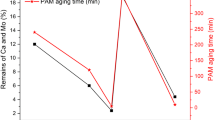
Calcium molybdate is a waste produced by the molybdenum industry, and it is very feasible to replace part of molybdenum oxide with calcium molybdate for smelting by the carbon reduction method. In this paper, through the one-factor test, we obtained the optimal conditions for the removal of impurity elements P, S, C, and Si as follows: melting temperature of 1525°C, holding time of 25 min, and alkalinity of 1.3. The contents of impurity elements P, S, C, and Si in ferromolybdenum alloy were detected as 0.046%, 0.041%, 1.43%, and 1.09%, respectively, under these conditions, and the removal rates of P and S could be up to 79.36% and 65.21%. Under this condition, three phases of Mo2C, Fe3Mo, and Fe3Mo3C exist in ferromolybdenum alloy, in which the distribution of the P, S, and C elements decrease in order. The slag system is mainly composed of a CaO-SiO2 binary system, and, under the above research conditions, P, S, C, and Si are made to enter into the slag system in different forms to achieve the decontamination effect of ferromolybdenum. The research results are of great significance for the expanded utilization of calcium molybdate and the improvement of ferromolybdenum quality.








Similar content being viewed by others
References
C. Zhu and B.-J. Wang, Hot Work. Technol. 38, 36 (2009).
Z.-Z. Zhang, D.-L. Li, X.-D. Xing, S.-F. Liu, G.-X. Zhang, and H.-H. Lu, J. Iron Steel Res. 32, 1 https://doi.org/10.13228/j.boyuan.issn1001-0963.20190169 (2020).
L. Wang, P.-M. Guo, L.-G. Luo, P. Zhao, and J.-M. Pang, Iron Steel 50, 100 https://doi.org/10.13228/j.boyuan.issn0449-749x.20150050 (2015).
C.-S. Kuang, J.-C. Cao, X.-L. Zhou, Q.-L. Yong, Y.-H. Yang, and Q.-C. Liu, Heat Treat. Met. 40, 1 https://doi.org/10.13251/j.issn.0254-6051.2015.03.001 (2015).
L.-J. Jiang, L.-P. Li, X.-H. Liu, L. Cao, and Y. Liu, China Molybd. Ind. 45, 1 https://doi.org/10.13384/j.cnki.cmi.1006-2602.2021.02.001 (2021).
M.Sun, Acta Metall. Sin. 7 (2021).
H.-Q. Chang, Y. Hou, K. He, H.-X. Wu, G.-Z. Zhou, and G.-H. Zhang, Acta Metall. Sin. 44, 1 https://doi.org/10.13384/j.cnki.cmi.1006-2602.2020.01.001 (2020).
L. Chen, Spec. Steel Technol. 25, 51 https://doi.org/10.16683/j.Cnki.Issn1674-0971.2019.3048 (2019).
H. Liu, L. Xie, and F. Yang, China Tungsten Ind. 34, 65–68 (2019).
S.-P. Yang and J.-F. Zhou, Nonferrous Met. Eng. 8, 57–60 (2018).
S.-P. Yang, J.-F. Zhou, S.-Q. Guo, P.-H. Zhang, and M. Wang, Nonferrous Met. Eng. 7, 48–53 (2017).
C. Zhang, Research on the new inhibitor of sulfide copper-molybdenum mine and its mechanism, Jiangxi University of Science and Technology (2017).
S. Huang, Research of flotation solution chemistry and regulation about a molybdenite, China University of Mining and Technology (2016).
M.-R. Chen, Preparation of SiO2 functional mesoporous materials from molybdenum tailings, Dalian Polytechnic University (2015).
B.-S. Kim, S.-B. Kim, H.-I. Lee, and Y.-Y. Choi, Mater. Trans. 52, 1288–1293 (2011).
M.H. Golmakani, J. Vahdati Khaki, and A. Babakhani, Mater. Chem. Phys. 194, 9 (2017).
M.K. Ziatdinov, I.M. Shatokhin, and L.I. Leont’ev, Steel Transl. 48, 269 (2018).
P. Aryal, S. Karki, Y. Choi, H. Kim, Y. Kim, V. Milyutin, O. Gileva, H. Park, and K. Shin, J. Mater. Cycles Waste 21, 1384 (2019).
J.W. An, Y.H. Lee, S.J. Kim, T. Tran, S.O. Lee, and M.J. Kim, Miner. Eng. 22, 1384 (2008).
W.-J. Zhang, C.-Y. Wang, and B.-Z. Ma, Trans. Nonferrous Met. Soc. China 29, 859 (2019).
S.-M. Li, Experimental research on direct alloying of liquid steel with molybdenum, nickel or chromium ore, Northeastern University (2019).
H. Huang, F.-L. Chen, Y. Jiang, H. Rong, Y.-F. Duo, and Z.-H. Yang, Yunnan Metall. 43, 66 (2014).
H.-X. Wu, L.-Y. Hong, J.-L. Tang, and X.-W. Li, Acta Metall. Sin. 42, 45 https://doi.org/10.13384/j.cnki.cmi.1006-2602.2018.02.010 (2018).
S.-P. Yang, C. Sun, and F. He, Ferro Alloys 47, 21 https://doi.org/10.16122/j.cnki.issn1001-1943.2016.01.005 (2016).
S.-P. Yang, S.-M. Liu, M. Wang, T.-T. Zhang, H.-X. Sun, and K. He, Chin. Metall. 32, 131 https://doi.org/10.13228/j.boyuan.issn1006-9356.20210717 (2022).
S.-P. Yang, Q.-S. Wei, J.-F. Zhou, B. Yang, and C. Wang, Chin. J. Proc. Eng. 18, 405 (2018).
S.-P. Yang, S.-Q. Guo, P.-H. Zhang, L. Wang, X. Zhang, K. He, X.-W. Li, and W.J. Dang, Chin. J. Proc. Eng. 16, 1016 (2016).
S.-Q. Guo, Study of microwave activated roasting of molybdenite, Xi’an University of Architecture and Technology (2017).
M. Morishita, Y. Kinoshita, H. Houshiyama, A. Nozaki, and H. Yamamoto, J. Chem. Thermodyn. 114, 30 (2017).
K. Łuczka, B. Grzmil, B. Michalkiewicz, and K. Kowalczyk, J. Ind. Eng. Chem. 23, 257 (2015).
H. Ahmed, T.S. Kumar, J. Alatalo, and B. Bjorkman, J. Mater. Res. Technol. 21, 1760 (2022).
D.R. Swinbourne and S. Arnout, Min. Proc. Ext. Met. Rev. 128, 193 (2019).
H.-K. Mao, B.-X. Guo, Z.-Y. Fan, H. Xu, F.-M. Hou, X. Cao, Q.-S. Zhao, J.-B. Hou, S.-Y. Liu, and Y. Wang, TNMSC 32, 485 (2022).
K.-P. Wang, M. Jiang, X.-H. Wang, Y. Wang, H.-Q. Zhao, and Z.-M. Cao, Metall. Mater. Tran B. 48, 2961 (2017).
S.-P. Yang, J.-Q. Liang, W.-M. Cai, C. Sun, D.-X. Liu, and L. Wang, Min. Metall. Eng. 35, 91 (2015).
M. Zakeri, M.R. Rahimipour, and A. Khanmohammadian, MSEA 492, 311 (2008).
Q.-X. Sun, Q.-J. Song, H.-F. Lu, and D.-D. Li, Phys. Test. Chem. Anal. (Part A Phys. Test.) 54, 754 (2018).
L.-P. Shu, S.-L. Zhu, W.-H. Hu, R.-Z. Luo, X.-G. Qiu, and X.-Y. Wu, Chin. Metall. 28, 43 https://doi.org/10.13228/j.boyuan.issn1006-9356.20180053 (2018).
H. Liu, Engineering Materials (Beijing Institute of Technology Press, Beijing, 2019).
L.-M. Zhang, J.-T. Wei, Y.-H. Bai, X.-D. Song, J.-F. Wang, W.-G. Su, P. Lv, Y. Zhou, G.-X. Nai, and G.-S. Yu, Fuel 326 (2022).
Acknowledgements
The authors gratefully wish to express the thanks to Shaanxi Provincial Science and Technology Plan Funded Project (2022JQ-302) for supporting this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, S., Liu, Y., Wang, M. et al. Study on Removal Rule of Impurity Elements in Preparation of Ferromolybdenum from Calcium Molybdate. JOM 76, 2491–2500 (2024). https://doi.org/10.1007/s11837-024-06396-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-024-06396-7