Abstract
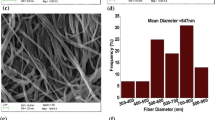
In this study, thin electrospun solvent-free electrolytes based on polyethylene oxide (PEO) incorporated with silicon dioxide (SiO2), aluminum oxide (Al2O3) and clay nanoparticles were prepared and characterized. Lithium perchlorate and ethylene carbonate were used as salt and plasticizer, respectively. The morphological properties were investigated using scanning electron microscopy, energy-dispersive spectroscopy, Fourier transform infrared spectroscopy and x-ray diffraction patterns. The obtained results confirmed an increment of the fraction of free ions and amorphous regions by incorporation of the fillers into the electrospun electrolytes. Introduction of the fillers into the PEO matrix significantly improved the ion conductivity. The highest ion conductivities of 0.033 mS cm−1, 0.059 mS cm−1 and 0.016 mS cm−1 were obtained by the addition of SiO2, Al2O3 and clay nanoparticles into the as-spun electrolytes, respectively. The electrospun electrolytes showed superior ion conductivities compared with polymeric electrolytes synthesized through a standard solution-casting method. In addition, the activation energy decreased with the addition of fillers into the electrospun fibres. The as-spun electrolytes displayed low cycling durability. Furthermore, tensile properties implied that tensile strength could be improved by loading an optimum ratio of the fillers. This investigation presents the great potential of electrospun membranes as electrolytes applicable for solid-state lithium-ion batteries.







Similar content being viewed by others
References
J.W. Fergus, J. Power Sources 195, 4554 (2010).
W.H. Meyer, J. Adv. Mater. 10, 439 (1998).
R. Agrawal and G. Pandey, J. Phys. D 41, 223001 (2008).
M.Z.A. Munshi, Solid polymer electrolytes, ed: Google Patents, 2003.
J.-M. Tarascon and M. Armand, Nature 414, 359 (2001).
J. Song, Y. Wang, and C.C. Wan, J. Power Sources 77, 183 (1999).
Z. Xue, D. He, and X. Xie, J. Mater. Chem. A 3, 19218 (2015).
R. M. Formato, R. F. Kovar, P. Osenar, N. Landrau, and L. S. Rubin, Composite solid polymer electrolyte membranes, ed: Google Patents, 2001.
M. Armand, Solid State Ionics 9, 745 (1983).
Z. Gadjourova, Y.G. Andreev, D.P. Tunstall, and P.G. Bruce, Nature 412, 520 (2001).
S. Chung, K. Such, W. Wieczorek, and J. Stevens, J. Polym. Sci. B 32, 2733 (1994).
M.A. Ratner and D.F. Shriver, Chem. Rev. 88, 109 (1988).
M. Jacob, S. Prabaharan, and S. Radhakrishna, Solid State Ionics 104, 267 (1997).
A.M. Stephan and K. Nahm, Polym. J. 47, 5952 (2006).
W. Liu, N. Liu, J. Sun, P.-C. Hsu, Y. Li, and H.-W. Lee, et al., Nano Lett. 15, 2740 (2015).
F. Yuan, H.-Z. Chen, H.-Y. Yang, H.-Y. Li, and M. Wang, Mater. Chem. Phys. 89, 390 (2005).
S.S. Zhang, J. Power Sources 162, 1379 (2006).
W. Wieczorek, Z. Florjanczyk, and J. Stevens, Electrochim. Acta 40, 2251 (1995).
E.M. Masoud, Polym. Test. 56, 65 (2016).
E.M. Masoud, A.-A. El-Bellihi, W.A. Bayoumy, and E.A. Mohamed, J. Mol. Liq. 260, 237 (2018).
E.M. Masoud, M.E. Hassan, S.E. Wahdaan, S.R. Elsayed, and S.A. Elsayed, Polym. Test. 56, 277 (2016).
K. Wimalaweera, V. Seneviratne, and M. Dissanayake, Procedia Eng. 215, 109 (2017).
N. Byrne, J. Efthimiadis, D. MacFarlane, and M. Forsyth, J. Mater. Chem. 14, 127 (2004).
Y. Yap, A. You, L. Teo, and H. Hanapei, Int. J. Electrochem. Sci. 8, 2154 (2013).
L. Fan, C.-W. Nan, and S. Zhao, Solid State Ionics 164, 81 (2003).
Y. Ma, L. Li, G. Gao, X. Yang, and Y. You, Electrochim. Acta 187, 535 (2016).
C. Tang, K. Hackenberg, Q. Fu, P.M. Ajayan, and H. Ardebili, Nano Lett. 12, 1152 (2012).
H.W. Chen, C.Y. Chiu, and F.C. Chang, J. Polym. Sci. B 40, 1342 (2002).
E.M. Masoud, Ionics 25, 2645 (2019).
S.N. Banitaba, D. Semnani, B. Rezaei, and A.A. Ensafi, Polym. Int. 68, 746 (2019).
K. Freitag, P. Walke, T. Nilges, H. Kirchhain, R. Spranger, and L. van Wüllen, J. Power Sources 378, 610 (2018).
K.M. Freitag, H. Kirchhain, L.V. Wüllen, and T. Nilges, Inorg. Chem. 56, 2100 (2017).
S. N. Banitaba, D. Semnani, E. Heydari-Soureshjani, B. Rezaei, and A. A. Ensafi, Mater. Res. Express, (2019)
S. N. Banitaba, D. Semnani, B. Rezaei, and A. A. Ensafi, Polym. Adv. Technol., (2019).
D. Hambali, Z. Zainuddin, I. SUPA, and Z. Osman, Sains Malays. 45, 1697 (2016).
M.R. Johan, O.H. Shy, S. Ibrahim, S.M.M. Yassin, and T.Y. Hui, Solid State Ionics 196, 41 (2011).
A. León, P. Reuquen, C. Garín, R. Segura, P. Vargas, and P. Zapata, et al., J. Appl. Sci. 7, 49 (2017).
C. Drew, X. Wang, L.A. Samuelson, and J. Kumar, J. Macromol. Sci. A 40, 1415 (2003).
A. Arya and A. Sharma, J. Phys. D 50, 443002 (2017).
C. Bhatt, R. Swaroop, A. Arya, and A. Sharma, J. Mater. Sci. Eng. B 5, 418 (2015).
J.O. Kweon and S.T. Noh, J. Appl. Polym. Sci. 81, 2471 (2001).
W. Liu, Multilayer Composite Solid Electrolytes for Lithium Ion Batteries, Dissertations (2016).
H.R. Pant, M.P. Bajgai, K.T. Nam, Y.A. Seo, D.R. Pandeya, and S.T. Hong, et al., J. Hazard. Mater. 185, 124 (2011).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Banitaba, S.N., Semnani, D., Heydari-Soureshjani, E. et al. Electrospun Polyethylene Oxide-Based Membranes Incorporated with Silicon Dioxide, Aluminum Oxide and Clay Nanoparticles as Flexible Solvent-Free Electrolytes for Lithium-Ion Batteries. JOM 71, 4537–4546 (2019). https://doi.org/10.1007/s11837-019-03810-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-019-03810-3