Abstract
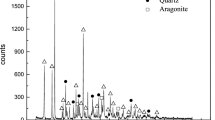
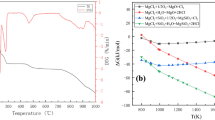
The typical method for the preparation of chromium-magnesia is by energy-consuming grinding, but the pollution of Cr6+ ions is a problem. However, the chromium-magnesia refractory is still irreplaceable due to its outstanding performance. In this regard, low-chromium magnesia was prepared by the alkaline chromium precipitation method from cheap light-burned magnesia. The impurities in the light-burned magnesia was removed in order to avoid the formation of Cr6+. The chromium was precipitated directly on the light-burned magnesia surface without the grinding process. The low-chromium magnesia was obtained after the calcination. The reaction temperature, solid content, reaction time and stirring speed were examined and low-chromium magnesia with Cr3+ 0.4%, MgO 98.5% was obtained. The effect of temperature on the surface tomography of low-chromium magnesia was also discussed. The present novel alkaline method should be a promising way for the preparation of low-chromium magnesia.





Similar content being viewed by others
References
D.Y. Kim, S.Y. Yoo, S.M. Kim, C.S. Ha, and J.M. Park, Am. Ceram. Soc. Bull. 84, 9201 (2005).
J.P. Bennett, K.S. Kwong, C.P. Powell, H. Thomas, and A.V. Petty Jr, In 20th Annual Conference on Fossil Energy Materials, (Knoxville, TN, US, 2007), pp. 200–206.
J. Li, H. Zhao, P. Zhao, J. Cui, S. Mu, and Y. Lv, Ceram. Int. 42, 18579 (2016). https://doi.org/10.1016/j.ceramint.2016.08.200.
H. Li, J. Liu, H. Feng, and L. Zhang, China’s Refract. 4, 2 (2014). https://doi.org/10.3969/j.issn.1004-4493.2014.04.001
B. Sahin and C. Aksel, J. Eur. Ceram. Soc. 32, 49 (2012). https://doi.org/10.1016/j.jeurceramsoc.2011.07.024.
S. Ritwik, K.D. Samir, and B. Goutam, J. Eur. Ceram. Soc. 22, 1243 (2002). https://doi.org/10.1016/s0955-2219(01)00446-0.
N.N. Tupotilov, V.V. Ostrikov, and A.Y. Kornev, Chem. Technol. Fuels Oils 44, 29 (2008). https://doi.org/10.1007/s10553-008-0012-7.
S. Fellahi, N. Chikhi, and M. Bakar, J. Appl. Polym. Sci. 82, 861 (2001). https://doi.org/10.1002/app.1918.
N.M. Deraz, Ceram. Int. 38, 511 (2012). https://doi.org/10.1016/j.ceramint.2011.07.036.
X. Liu, Y. Feng, H. Li, P. Zhang, and P. Wang, J. Cent. South Univ. (Sci. Technol.) 42, 3912 (2011).
H. Xu, Y. Cai, X. Shi, and G. Pi, J. Nat. Sci. Hunan Norm. Univ. 29, 52 (2006). https://doi.org/10.3969/j.issn.1000-2537.2006.01.013.
H. Xu, Y. Cai, B. Chen, and Y. Su, J. Cent. South Univ. 37, 698 (2006). https://doi.org/10.3969/j.issn.1672-7207.2006.04.014.
H. Xu, W. Liu, X. Yang, X. Shi, S. Chen, and L. Yu, J. Cent. South Univ. (Sci. Technol.) 42, 2204 (2011).
W. Liu, H.Xu, X. Shi, and X. Yang, In XVII Balkan Mineral Processing Congress (BMPC-2017) (Antalya, Turkey, 2017), pp. 513–521.
W. Liu, H. Xu, X. Yang, X. Chang, and Y. Chen, J. Cent. South Univ. 19, 2751 (2012). https://doi.org/10.1007/s11771-012-1337-2.
H.R. Zargar, C. Oprea, G. Oprea, and T. Troczynski, Ceram. Int. 38, 6235 (2012). https://doi.org/10.1016/j.ceramint.2012.04.077.
J.W. Nelson and I.B. Cutler, J. Am. Ceram. Soc. 41, 406 (1958). https://doi.org/10.1111/j.1151-2916.1958.tb13512.x.
X. Shi, F. Wang, and S. Xiong, Nonferrous Met. Sci. Eng. 3, 54 (2016). https://doi.org/10.13264/j.cnki.ysjskx.2016.03.010.
Y. Deng, H. Wang, and H. Zhao, Ceram. Int. 34, 573 (2008). https://doi.org/10.1016/j.ceramint.2006.12.002.
W. Ren, B. Xue, C. Lu, Z. Zhang, Y. Zhang, and L. Jiang, J. Clean. Prod. 135, 214 (2016). https://doi.org/10.1016/j.jclepro.2016.06.118.
H. Xu, W. Liu, R. Dong, X. Yang, X. Shi, and N. Zhao, Nonferrous Met. (Extr. Metall.) 20 (2011). https://doi.org/10.3969/j.issn.1007-7545.2011.01.006.
Y. Liang, Y. Yang, and W. Mao, Rock Miner. Anal. 26, 73 (2007). https://doi.org/10.3969/j.issn.0254-5357.2007.01.018.
B. Müller, ChemEQL V3.2, (Duebendorf: Swiss Federal Institute for Environmental Science and Technology, 1996), pp. 1–6.
Y.C. Akira, In Supplement of China’s Refractories—Proceeding of the Fifth International Symposium on Refractories (2007), pp. 28–30.
W. Liu, H. Xu, X. Shi, X. Yang, Y. Chen, J. Cheng, and G. Li, Chin. J. Nonferrous Met. 22, 2656 (2012).
W. Liu, H. Xu, J. Cheng, G. Li, X. Shi, and X. Yang, Mater. Rev. 26, 313 (2012). https://doi.org/10.3969/j.issn.1005-023X.2012.z1.084.
H. Xu, J. Cheng, G. Li, X. Yang, and W. Liu, Mater. Rev. 27, 104 (2013). https://doi.org/10.3969/j.issn.1005-023X.2013.16.028.
T.A. Clancy, University of Missouri-Rolla, 4 (1968).
Y. Sun, H. Qian, J. Liu, and B. Yu, Non Met. Mines 28, 54 (2005). https://doi.org/10.3969/j.issn.1000-8098.2005.04.020.
D.J. Bellamy and P.H. Clarke, Nature 218, 1180 (1968). https://doi.org/10.1038/2181180a0.
D. Hou and H. Li, J. Salt Lake Res. 16, 45 (2008).
K.P. Ananthapadmanabhan and P. Somasundaran, Colloids Surf. 13, 151 (1985). https://doi.org/10.1016/0166-6622(85)80014-7.
D.P. Rai, B.M. Sass, and D.A. Moore, Inorg. Chem. 26, 345 (1987). https://doi.org/10.1021/ic00250a002.
L.R. Owen, E.J. Pickering, H.Y. Playford, H.J. Stone, M.G. Tucker, and N.G. Jones, Acta Mater. 122, 11 (2017). https://doi.org/10.1016/j.actamat.2016.09.032.
R.S. Mishra, N. Kumar, and M. Komarasamy, Mater. Sci. Technol. 31, 1259 (2015). https://doi.org/10.1179/1743284715Y.0000000050.
Acknowledgements
Funding for this research by National Natural Science Foundation of China (51574286) and China Postdoctoral Science Foundation (2016M592448) are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wang, C., Xu, H., Liu, W. et al. Novel Alkaline Method for the Preparation of Low-Chromium Magnesia. JOM 72, 333–339 (2020). https://doi.org/10.1007/s11837-018-3284-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-018-3284-3