Abstract
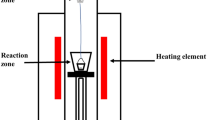
In this work, graphite flakes were used to reduce fayalite slag originated from the pyrometallurgical copper extraction process. Experiments were conducted with a significantly different contact area between graphite and slag at two temperatures, 1300°C and 1400°C. The process was continuously monitored via the concentration change of CO and CO2 in off-gas. Reduction rate values in experiments where 150-micron-diameter graphite flakes were submerged into the slag and left to float slowly to the top are about four times higher compared with when graphite flakes were dispersed at the top surface of liquid slag. The activation energy for instigating reduction was 302.61 kJ mol−1 and 306.67 kJ mol−1 in the case where graphite flakes were submerged into the slag and dispersed at the surface, respectively. The reduction process is characterized by two distinctive periods: an initial steep increase in the concentration of CO and CO2 controlled by the Boudouard reaction and a subsequent slow decrease of CO and CO2 concentrations in the off-gas controlled by mass transfer of reducible oxides from bulk to the gas–slag interface.




Similar content being viewed by others
References
The Future of Non-Ferrous Slag: Market Forecasts to 2027 (Smithers Apex, 2017). http://www.smithersapex.com/market-reports/the-future-of-non-ferrous-slag-market-forecasts-to. Accessed 12 July 2017.
L.E. Vargas, N.R.R. Reyes, and E. Estupiñán, Facultad de Ingeniería 26, 59 (2017).
S. Hosseini, S.M. Soltani, P. Fennell, T. Choong, and M. Aroua, Environ. Technol. Rev. 5, 1 (2016).
Coal Industry Advisory Board, The Global Value of Coal (OECD/IEA, 2012). https://www.scribd.com/document/135299346/Global-Value-of-Coal. Accessed 25 March 2017.
J. Mróz, Metall. Mater. Trans. B 32, 821 (2001).
D. Min and R. Fruehan, Metall. Trans. B 23B, 29 (1992).
R.K. Paramguru, R. Galgali, and H. Ray, Metall. Mater. Trans. B 28B, 805 (1997).
A. Warczok and T. Utigard, Can. Metall. Q. 37, 27 (1998).
D. Min, J. Han, and W. Chung, Metall. Mater. Trans. B 30B, 215 (1999).
T. Utigard, G. Sunchez, J. Manriquez, A. Lurashi, C. Diaz, D. Cordero, and E. Almendras, Metall. Mater. Trans. B 28, 821 (1997).
M. Barati and K. Coley, Metall. Mater. Trans. B 36, 169 (2005).
S. Sun and G. Belton, Metall. Mater. Trans. B 29, 137 (1998).
C. Liu, S. Huang, P. Wollants, B. Blanpain, and M. Guo, Metall. Mater. Trans. B 48, 1602 (2017).
Y. Sun, Y. Han, P. Gao, and Y. Li, ISIJ Int. 56, 1697 (2016).
B. L’vov, Thermochim. Acta 360, 109 (2000).
K. Takahashi, M. Amatatsu, and T. Soma, Tetsu-to-Hagane 61, 2525 (1975).
Y. Sasaki and T. Soma, Tetsu-to-Hagane 64, 376 (1978).
C. Borgranni, Ironwork. Steelmak. 5, 61 (1978).
Acknowledgements
The authors are grateful to Prof. Torstein Utigard for his guidance and assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mitrašinović, A. Effect of Temperature and Graphite Immersion Method on Carbothermic Reduction of Fayalite Slag. JOM 69, 1682–1687 (2017). https://doi.org/10.1007/s11837-017-2455-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-017-2455-y