Abstract
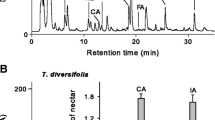
Several studies on floral nectar demonstrated that the behaviour of visit performed by pollinators is influenced by nectar chemistry. Biogenic amines act as neurotransmitters in invertebrates and recently have been reported in the floral nectar of 15 plant species for the first time. However, both their occurrence in floral nectar and the effects of their nectar-like concentrations on bee behaviour remains largely unsubstantiated. To increase knowledge on the role of biogenic amines on plant–pollinator interactions, here we (i) investigated the biogenic amine composition of Echium vulgare nectar in relation to its floral sexual phases, and (ii) studied how an artificial solution enriched with nectar-like concentrations of tyramine affects the visit on flowers of bumblebees under semi-controlled conditions. The chemical analysis reported the presence of tyramine in E. vulgare nectar and no difference in concentration between the two sexual phases. To explore potential effects of tyramine on bee behaviour, we designed a new method consisting in zucchini flowers emptied of their natural nectar and refilled with artificial tyramine-enriched nectar, and we used bumblebee workers as pollinator model. We found that bees fed tyramine-enriched solution spent less time foraging on a single flower than those fed control solution, suggesting that their behaviour of visit was overall more dynamic. Our results highlight the importance of addressing further investigations on this emerging class of nectar compounds on insect cognition and behaviour, other than on its occurrence and distribution in nectar of other species.


Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article [and its supplementary information files].
References
Alkema MJ, Hunter-Ensor M, Ringstad N, Horvitz HR (2005) Tyramine functions independently of octopamine in the Caenorhabditis elegans nervous system. Neurons 46:247–260. https://doi.org/10.1016/j.neuron.2005.02.024
Baker HG, Baker I (1977) Intraspecific constancy of floral nectar amino acid complements. Bot Gaz 138:183–191. https://doi.org/10.1086/336914
Baker HG, Baker I (1986) The occurrence and significance of amino acids in floral nectars. Plant Syst Evol 151:175–186. https://doi.org/10.1007/BF02430273
Barberis M, Bogo G, Bortolotti L, Conte L, Alessandrini M, Nepi M, Galloni M (2021) Gender-biased nectar targets different behavioural traits of flower visitors. Plant Ecol 222:233–246. https://doi.org/10.1007/s11258-020-01101-5
Barberis M, Calabrese D, Galloni M, Nepi M (2023) Secondary metabolites in nectar-mediated plant-pollinator relationships. Plants 12(3):550. https://doi.org/10.3390/plants12030550
Barlow SE, Wright GA, Ma C, Barberis M, Farrell IW, Marr EC, Brankin A, Pavlik BM, Stevenson PC (2017) Distasteful nectar deters floral robbery. Curr Biol 27:2552–2558. https://doi.org/10.1016/j.cub.2017.07.012
Barron AB, Maleszka R, Vander Meer RK, Robinson GE (2007) Octopamine modulates honey bee dance behavior. PNAS 104(5):1703–1707. https://doi.org/10.1073/pnas.0610506104
Blenau W, Baumann A (2001) Molecular and pharmacological properties of insect biogenic amine receptors: lessons from Drosophila melanogaster and Apis mellifera. Arch Insect Biochem Physiol 48:13–38. https://doi.org/10.1002/arch.1055
Bogo G, Bortolotti L, Sagona S, Felicioli A, Galloni M, Barberis M, Nepi M (2019) Effects of non protein amino acids in nectar on bee survival and behavior. J Chem Ecol 45:278–285. https://doi.org/10.1007/s10886-018-01044-2
Bogo G, Fisogni A, Rabassa-Juvanteny J, Bortolotti L, Nepi M, Guarnieri M, Conte L, Galloni M (2021) Nectar chemistry is not only a plant’s affair: floral visitors affect nectar sugar and amino acid composition. Oikos 103:1180–1192. https://doi.org/10.1111/oik.08176
Boppré M (2011) The ecological context of pyrrolizidine alkaloids in food, feed and forage: an overview. Food Addit Contam Part A 28(3):260–281. https://doi.org/10.1080/19440049.2011.555085
Corbet SA (1978) Bee visits and the nectar of Echium vulgare L. and Sinapis alba L. Ecol Entomol 3:25–37. https://doi.org/10.1111/j.1365-2311.1978.tb00900.x
Cresswell JE (1999) The influence of nectar and pollen availability on pollen transfer by individual flowers of oil-seed rape (Brassica napus) when pollinated by bumblebees (Bombus lapidarius). J Ecol 87:670–677. https://doi.org/10.1046/j.1365-2745.1999.00385.x
Felicioli A, Sagona S, Galloni M, Bortolotti L, Bogo G, Guarnieri M, Nepi M (2018) Effects of nonprotein amino acids on survival and locomotion of Osmia bicornis. Insect Mol Biol 27(5):556–563. https://doi.org/10.1111/imb.12496
Fisogni A, Cristofolini G, Rossi M, Galloni M (2011) Pollinator directionality as a response to nectar gradient: promoting outcrossing while avoiding geitonogamy. Plant Biol 13:848–856. https://doi.org/10.1111/j.1438-8677.2011.00453.x
Fussnecker BL, Smith BH, Mustard JA (2006) Octopamine and tyramine influence the behavioral profile of locomotor activity in the honey bee (Apis mellifera). J Insect Physiol 52:1083–1092. https://doi.org/10.1016/j.jimsphys.2006.07.008
Galen C, Plowright RC (1984) The effects of nectar level and flower development on pollen carry-over in inflorescences of fireweed (Epilobium angustifolium) (Onagraceae). Can J Bot 63:488–481. https://doi.org/10.1139/b85-060
Hammer M, Menzel R (1998) Multiple sites of associative odor learning as revealed by local brain microinjections of octopamine in honeybees. Learn Mem 5:146–156. https://doi.org/10.1101/lm.5.1.146
Harder LD (1983) Flower handling efficiency of bumble bees: morphological aspects of probing time. Oecologia 57:274–280. https://doi.org/10.1007/BF00379591
Hardie SL, Zhang JX, Hirsh J (2007) Trace amines differentially regulate adult locomotor activity, cocaine sensitivity, and female fertility in Drosophila melanogaster. Dev Neurobiol 67(10):1396–1405. https://doi.org/10.1002/dneu.20459
Hartmann T, Witte L (1995) Chemistry, biology and chemoecology of the pyrrolizidine alkaloids. In: Pelletier SW (ed) Alkaloids: chemical and biological perspectives, vol 9. Pergamon Press, Oxford, pp 155–233
Kessler D, Bhattacharya S, Diezel C, Rothe E, Gase K, Schöttner M, Baldwin IT (2012) Unpredictability of nectar nicotine promotes outcrossing by hummingbirds in Nicotiana attenuata. Plant J 71(4):529–538. https://doi.org/10.1111/j.1365-313X.2012.05008.x
Kutsukake M, Komatsu A, Yamamoto D, Ishiwa-Chigusa S (2000) A tyramine receptor gene mutation causes a defective olfactory behavior in Drosophila melanogaster. Gene 245:31–42
Lange AB (2009) Tyramine: from octopamine precursor to neuroactive chemical in insects. Gen Comp Endocrinol 162(1):18–26. https://doi.org/10.1016/j.ygcen.2008.05.021
Laverty TM (1994) Bumble bee learning and flower morphology. Anim Behav 47:531–545. https://doi.org/10.1006/anbe.1994.1077
Lucchetti MA, Glauser G, Kilchenmann V, Dübecke A, Beckh G, Praz C, Kast C (2016) Pyrrolizidine alkaloids from Echium vulgare in honey originate primarily from floral nectar. J Agric Food Chem 64:5267–5273. https://doi.org/10.1021/acs.jafc.6b02320
Melser C, Rademaker MCJ, Klinkhamer PGL (1997) Selection on pollen donors by Echium vulgare (Boraginaceae). Sex Plant Reprod 10:305–312. https://doi.org/10.1007/s004970050103
Mercer AR, Menzel R (1982) The effect of biogenic amines on conditioned and unconditioned responses to olfactory stimuli in the honeybee Apis mellifera. J Comp Physiol A 145:363–368. https://doi.org/10.1007/BF00619340
Mustard JA (2020) Neuroactive nectar: compounds in nectar that interact with neurons. Arthropod Plant Interact 14:151–159. https://doi.org/10.1007/s11829-020-09743-y
Muth F, Philbin CS, Jeffrey CS, Leonard AS (2022) Discovery of octopamine and tyramine in nectar and their effects on bumblebee behavior. Iscience 25(8):104765. https://doi.org/10.1016/j.isci.2022.104765
Nagaya Y, Kutsukake M, Chigusa SI, Komatsu A (2002) A trace amine, tyramine, functions as a neuromodulator in Drosophila melanogaster. Neurosci Lett 329:324–328. https://doi.org/10.1016/s0304-3940(02)00596-7
Nepi M (2017) New perspectives in nectar evolution and ecology: simple alimentary reward or a complex multiorganism interaction? Acta Agrobot 70(1):1704. https://doi.org/10.5586/aa.1704
Nepi M, Cresti L, Guarnieri M, Pacini E (2011) Dynamics of nectar production and nectar homeostasis in male flowers of Cucurbita pepo L. Int J Plant Sci 172(2):183–190. https://doi.org/10.1086/657648
Nepi M, Grasso DA, Mancuso S (2018) Nectar in plant–insect mutualistic relationships: from food reward to partner manipulation. Front Plant Sci 9:1063. https://doi.org/10.3389/fpls.2018.01063
Peng T, Schroeder M, Grüter C (2020) Octopamine increases individual and collective foraging in a neotropical stingless bee. Biol Lett 16:20200238. https://doi.org/10.1098/rsbl.2020.0238
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2020) linear and non-linear mixed effects models. https://CRAN.R-project.org/package=nlme
Pleasants JM (1981) Bumblebee response to variation in nectar availability. Ecology 62:1648–1661. https://doi.org/10.2307/1941519
Pyke GH (2016) Floral nectar: pollinator attraction or manipulation? Trends Ecol Evol 31(5):339–341. https://doi.org/10.1016/j.tree.2016.02.013
Real RA, Rathcke BJ (1991) Individual variation in nectar production and its effects on fitness in Kalmia latifolia. Ecology 72:149–155. https://doi.org/10.2307/1938910
Rhoades DF, Bergdahl JC (1981) Adaptive significance of toxic nectar. Am Nat 117:798–803
Roeder T (2005) Tyramine and octopamine: ruling behavior and metabolism. Annu Rev Entomol 50:447–477. https://doi.org/10.1146/annurev.ento.50.071803.130404
Schulz DJ, Robinson GE (2001) Octopamine influences division of labor in honey bee colonies. J Comp Physiol A 187:53–61. https://doi.org/10.1007/s003590000177
Sgolastra F, Medrzycki P, Bortolotti L, Renzi MT, Tosi S, Bogo G, Teper D, Porrini C, Molowny-Horas R, Bosch J (2017) Synergistic mortality between a neonicotinoid insecticide and an ergosterol biosynthesis-inhibiting fungicide in three bee species. Pest Manag Sci 73:1236–1243. https://doi.org/10.1002/ps.4449
Shykoff JA, Bucheli E (1995) Pollinator visitation patterns, floral rewards and the probability of transmission of Microbotryum violaceum, a veneral disease plant. J Ecol 83:189–198. https://doi.org/10.2307/2261557
Thomson JD, Draguleasa MA, Guorui Tan M (2015) Flowers with caffeinated nectar receive more pollination. Arthropod-Plant Interact 9:1–7. https://doi.org/10.1007//s11829-014-9350-z
Venables WN, Ripley BD (2002) Modern applied statistics with S, 4th edn. Springer, New York
Wright GA, Baker DD, Palmer MJ, Stabler D, Mustard A, Power EF, Borland AM, Stevenson PC (2013) Caffeine in floral nectar enhances a pollinator’s memory of reward. Science 339:1202–1204. https://doi.org/10.1126/science.1228806
Acknowledgements
Thanks to Salvatore Cozzolino for scientific advice, Rosa Ranalli and Laura Zavatta for their help in the realization of the study, and Michela Boi for helping with data management. We are grateful to Fausto Bonafede and WWF Bologna Metropolitana for allowing field surveys in the protected area.
Funding
This work was partly supported by the Project BeeNet (Italian National Fund under FEASR 2014–2020) from the Italian Ministry of Agriculture, Food Sovereignty and Forestry (MASAF). Marta Barberis was supported by a PhD Grant from the University of Bologna.
Author information
Authors and Affiliations
Contributions
Conceptualization: MG, MN, MB, AF, GB and LB; Methodology and Investigation: MB, GB and MG; Statistical Analyses: MB, GB; Writing—Original Draft Preparation: MB; Writing—Review and Editing: GB, LB, MG, MN and AF.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Isabel Alves dos Santos.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Barberis, M., Bogo, G., Bortolotti, L. et al. Nectar tyramine decreases the duration of bumblebee visits on flowers. Arthropod-Plant Interactions 17, 563–569 (2023). https://doi.org/10.1007/s11829-023-09976-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-023-09976-7