Abstract
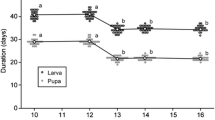
According to the theory of optimal foraging and preference–performance hypothesis, herbivores usually select plant hosts that benefit themselves or their offspring. We investigated the hypothesis that gravid females of the cucurbit beetle Diabrotica speciosa use volatiles derived from non-infested maize plants and herbivore-infested plants under attack by their conspecific immatures and adults in selecting a host for oviposition. The response of D. speciosa females to volatiles was quantified for the following treatments: (i) non-infested plants; (ii) plants infested by larvae (root-infested); (iii) plants infested by adults (leaf-infested); and (iv) plants infested by larvae and adults (leaf + root-infested). We also assessed if females were able to identify conditions of competition and host adequacy for offspring by means of chemical cues emitted by these plants. The results indicated that the presence of D. speciosa conspecifics on maize plants triggered the emission of volatiles, which influenced host selection for oviposition. Gravid females avoided both plants infested by larvae and plants infested by larvae and adults. Leaf-infested plants did not affect the host-selection behavior of females. Weight gain was higher in larvae kept on non-infested and leaf-infested plants than on root-infested or root + leaf-infested plants, indicating that female preference is related to its offspring performance.
Graphical Abstract
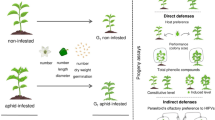
Synthesis of the effects of volatile organic compounds (VOCs) produced by uninfested, leaf-infested, root-infested and leaf+root-infested corn plants on the host-choice behavior of adults and performance of Diabrotica speciosa larvae





Similar content being viewed by others
Data availability
The dataset supporting the results of this study is available upon request to the corresponding author (caroline_rech@outlook.com).
References
Arce CCM, Theepan V, Shimmel BCJ, Jaffuel G, Erb M, Machado RAR (2021) Plant-associated CO2 mediates long-distance host location and foraging behavior of a root herbivore. Elife. https://doi.org/10.7554/eLife.65575
Ávila CJ, Tabai ACP, Parra JRP (2000) Rearing techniques of Diabrotica specisoa (Germar) (Coleoptera: Chrysomelidae) on natural and artificial diets. Na Soc Entomol Bras 29:257–267. https://doi.org/10.1590/S0301-80592000000200007
Baur R, Koštál V, Städler E (1996) Root damage by conspecific larvae induces preference for oviposition in cabbage root flies. Entomol Exp Appl 80:224–227. https://doi.org/10.1007/978-94-009-1720-0_51
Bezemer TM, Wagenaar R, van Dam NM, Wäckers FL (2003) Interactions between above- and belowground insect herbivores as mediated by the plant defense system. Oikos 101:555–562. https://doi.org/10.1034/j.1600-0706.2003.12424.x
Boiça Junior AL, Costa EN, Nogueira L, Ribeiro ZA (2022) Evaluation of maize genotypes on ovipositiion preference of Diabrotica speciosa (Germar). Arthtopod Plant Interact 16:691–698. https://doi.org/10.1007/s11829-022-09928-7
Boriani M, Agosti M, Kiss J, Edwards CR (2006) Sustainable management of the western corn rootworm, Diabrotica virgifera virgifera LeConte (Coleoptera: Chrysomelidae), in infested areas: experiences in Italy, Hungary and the USA. EPPO Bull 36:531–537. https://doi.org/10.1111/j.1365-2338.2006.01055.x
Bretz F, Hothorn T, Westfall P (2010) Multiple comparisons using R, 1st edn. Chapman and Hall, New York
Carvalho MRM, Vasconcellos-Neto J (2021) Host plant selection and larval performance in the Neotropical butterfly Mechanitis polymnia casabranca. Entomol Exp Appl 169:255–263. https://doi.org/10.1111/eea.13011
Clark KE, Hartley SE, Johnson SN (2011) Does mother know best? The preference-performance hypothesis and parent-offspring conflict in aboveground-belowground herbivore life cycles. Ecol Entomol 36:117–124. https://doi.org/10.1111/j.1365-2311.2010.01248.x
Costa EN, Fernandes MG, Reis LC, Martins LO, Foresti AC, de Paula Quintão Scalon S, (2022) Above-and belowground resistance in Brazilian maize varieties under attack of Spodoptera frugiperda and Diabrotica speciosa. Entomol Exp Appl 170(8):718–726
Demétrio CGB, Hinde J, Moral RA (2014) Models for overdispersed fata in Entomology. In: Ferreira C, Godoy W (eds) Ecological modelling applied to Entomology. Springer International Publishing Switzerland, Entomology in Focus, pp 219–259
Dicke M, van Loon JJA (2000) Multitrophic effects of herbivore-induced plant volatiles in an evolutionary context. Entomol Exp Appl 97:237–249. https://doi.org/10.1046/j.1570-7458.2000.00736.x
Erb M, Robert CAM, Hibbard BE, Turlings TCJ (2011) Sequence of arrival determines plant-mediated interactions between herbivores. J Ecol 99:7–15. https://doi.org/10.1111/j.1365-2745.2010.01757.x
Fernandes P, Hilker M (2007) Host plant location by Chysomelidae. Basic Appl Ecol 8:97–116. https://doi.org/10.1016/j.baae.2006.05.001
Gripenberg S, Mayhew PJ, Parnell M, Roslin T (2010) A meta-analysis of preference-performance relationships in phytophagous insects. Ecol Lett 13:383–393. https://doi.org/10.1111/j.1461-0248.2009.01433.x
Hammack L (1997) Attractiveness of synthetic corn volatiles to northern and western corn rootworm beetles (Coleoptera: Chrysomelidae). Environ Entomol 26:311–317. https://doi.org/10.1093/ee/26.2.311
Hammack L (2001) Single and blended maize volatiles as attractants for diabroticite corn rootworm beetles. J Chem Ecol 27:1373–1390. https://doi.org/10.1023/A:1010365225957
Hammack L, Hibbard BE, Holyoke CW, Kline M, Leva DM (1999) Behavioral response of corn rootworm adults to host plant volatiles perceived by western corn rootworm (Coleoptera: Chrysomelidae). Environ Entomol 28:961–967. https://doi.org/10.1093/ee/28.6.961
Hiltpold I, Hibbard BE (2016) Neonate larvae of the specialist herbivore Diabrotica virgifera virgifera do not exploit the defensive volatile (E)-β-caryophyllene in locating maize roots. J Pest Sci 89:853–858. https://doi.org/10.1007/s10340-015-0714-7
Jaenike J (1978) On optimal oviposition behavior in phytophagous insects. Theor Popul Biol 14:350–356. https://doi.org/10.1016/0040-5809(78)90012-6
Johnson SN, Gregory PJ (2006) Chemically-mediated host-plant location and selection by root-feeding insects. Physiol Entomol 31 (1):1–13. https://doi.org/10.1111/j.1365-3032.2005.00487.x
Johnson SN, Rasmann S (2015) Root-feeding insects and their interactions with organisms in the rhizosphere. Annu Rev Entomol 60:517–535. https://doi.org/10.1146/annurev-ento-010814-020608
Johnson SN, Erb M, Hartley SE (2016) Roots under attack: contrasting plant responses to below- and aboveground insect herbivory. New Phytol 210:413–418. https://doi.org/10.1111/nph.13807
Jones LC, Rafter MA, Walter GH (2019) Insects allocate eggs adaptively across their native host plants. Arthropod Plant Interact 13:181–191. https://doi.org/10.1007/s11829-019-09688-x
Kaplan I, Halitschke R, Kessler A, Rehill BJ, Sardanelli S, Denno RF (2008) Physiological integration of roots and shoots in plant defense strategies links above- and belowground herbivory. Ecol Lett 11:841–851. https://doi.org/10.1111/j.1461-0248.2008.01200.x
Karssemeijer PN, Reichelt M, Gershenzon J, Van Loon J, Dicke M (2020) Foliar herbivory by caterpillars and aphids differentially affects phytohormonal signaling on roots and plant defence to a root herbivore. Plant Cell Environ 43:775–789. https://doi.org/10.1111/pce.13707
Karssemeijer PN, Weiger L, Van Loon JJA, Dicke M (2022) Leaf-chewing herbivores affect preference and performance of a specialist root herbivore. Oecologia 199:243–255. https://doi.org/10.1007/s00442-022-05132-9
Kohandani F, Le Goff GJ, Hance T (2017) Does insect mother know under what conditions it will make their offspring live? Insect Sci 24:141–149. https://doi.org/10.1111/1744-7917.12300
Marti G, Erb M, Boccard J, Glauser G, Doyen GR, Villard N, Robert CAM, Turlings TCJ, Rudaz S, Wolfender JL (2013) Metabolomics reveals herbivore-induced metabolites of resistance and susceptibility in maize leaves and roots. Plant, Cell Environ 36(3):621–639. https://doi.org/10.1111/pce.12002
Mayhew PJ (1997) Adaptive patterns of host-plant selection by phytophagous insects. Oikos 79:417–428. https://doi.org/10.2307/3546884
Menacer K, Cortesero NM, Hervé MR (2021) Challenging the Preference-Performance Hypothesis in an above-belowground insect. Oecologia 197:179–187. https://doi.org/10.1007/s00442-021-05007-5
Midega CAO, Khan ZR, Pickett JA, Nylin S (2011) Host plant selection behaviour of Chilo partekkus and its implication for effectiveness of a trap crop. Entomol Exp Appl 138:40–47. https://doi.org/10.1111/j.1570-7458.2010.01073.x
Mithöfer A, Wanner G, Boland W (2005) Effects of feeding Spodoptera littoralis on lima bean leaves. II. Continuous mechanical wounding resembling insect feeding is sufficient to elicit herbivory-related volatile emission. Plant Physiol 137(3):1160–1168. https://doi.org/10.1104/pp.401.054460
Murphy SM (2004) Enemy-free space maintains swallowtail butterfly host shift. Proceedings National Academy Sci 101(52): 18048–18052. https://doi.org/10.1073/pnas.0406490102
Pearman PB, Wilbur HM (1990) Changes in population dynamics resulting from oviposition in a subdivided habitat. Am Nat 135:706–723. https://doi.org/10.1086/285070
Pereira RV, Filgueiras CC, Willett DS, Peñaflor MFGV (2020) Sight unseen: Belowground feeding influences the distribution of an aboveground herbivore. Ecosphere 11(9):e03163. https://doi.org/10.1002/ecs2.3163
Randlkofer B, Bermaier EO, Meiners T (2007) Mother’s choice of the oviposition site: balancing risk of egg parasitism and need of food supply for the progeny with an infochemical shelter? Chemoecology 17:177–186. https://doi.org/10.1007/s00049-007-0377-9
Rasmann S, Turlings TCJ (2007) Simultaneous feeding by aboveground and belowground herbivores attenuates plant-mediated attraction on their respective natural enemies. Ecol Lett 10:926–936. https://doi.org/10.1111/j.1461-0248.2007.01084.x
Robert CAM, Erb M, Duployer M, Zwahlen C, Doyen GR, Turlings TCJ (2012a) Herbivore-induce plant volatiles mediate host selection by a root herbivore. New Phytol 194:1061–1069. https://doi.org/10.1111/j.1469-8137.2012.04127.x
Robert CA, Erb M, Hibbard BE, Wade French B, Zwahlen C, Turlings TC (2012b) A specialist root herbivore reduces plant resistance and uses an induced plant volatile to aggregate in a density-dependent manner. Funct Ecol 26(6):1429–1440. https://doi.org/10.1111/j.1365-2435.2012.02030.x
Rostás M, Cripps MG, Silcock P (2015) Aboveground endophyte affects root volatile emission and host plant selection of a belowground insect. Oecologia 177:487–497. https://doi.org/10.1007/s00442-014-3104-6
Schoonhoven LM, van Loon B, van Loon JJ, Dicke M (2005) Insect-plant biology. Oxford University Press, Oxford
Shikano I, Akhtar Y, Isman MB (2010) Relationship between adult and larval host plant selection and larval performance in the generalist moth, Trichoplusia ni. Arthropod Plant Interact 4:197–205. https://doi.org/10.1007/s11829-010-9100-9
Soler R, Harvey JA, Kamp AFD, Vet LEM, van der Putten WH, van Dam NM, Steufer JF, Gols R, Hordjk CA, Bezemer TM (2007) Root herbivores influence the behaviour of an aboveground parasitoid through changes in plant-volatile signals. Oikos 116:367–376. https://doi.org/10.1111/j.0030-1299.2007.15501.x
Soler R, Schaper SV, Bezemer TM, Cortesero AM, Hoffmeister TS, van der Putten WH, Vet LEM, Harvey JA (2009) Influence of presence and spatial arrangement of belowground insects on host-plant selection of aboveground insects: a field study. Ecol Entomol 34:339–345. https://doi.org/10.1111/j.1365-2311.2008.01082.x
Thompson JN (1988) Evolutionary ecology of the relationship between oviposition preference and performance of offspring in phytophagous insects. Entomol Exp Appl 47:3–14. https://doi.org/10.1111/j.1570-7458.1988.tb02275.x
Thompson MN, Grunseich JM, Marmolejo LO, Aguirre NM, Bradicich PA, Behmer ST, Suh CPC, Helms AM (2022) Undercover operation: belowground insect herbivory modifies systemic plant defense and repels aboveground foraging insect herbivores. Front Ecol Evol 10:1033730. https://doi.org/10.3389/fevo.2022.1033730
Toepfer S, Kuhlmann U (2006) Constructing life-tables for the invasive maize pest Diabrotica virgifera virgifera (Col. Chrysomelidae) in Europe. J Appl Entomol 130(4):193–205. https://doi.org/10.1111/j.1439-0418.2006.01060.x
Turlings TCJ, Lengwiler UB, Bernasconi ML, Wechsler D (1998) Timing of induced volatile emissions in maize seedlings. Planta 207:146–152. https://doi.org/10.1007/s004250050466
Valladares G, Lawton JH (1991) Host-plant selection in the holly leaf-miner: does mother know best? J Anim Ecol 60:227–240. https://doi.org/10.2307/5456
Van Dam NM, Harvey JA, Wäckers FL, Bezemer TM, Van Der Putten WH, Vet LEM (2003) Interactions between aboveground and belowground induced responses against phytophages. Basic Appl Ecol 4:63–77. https://doi.org/10.1078/1439-1791-00133
Walsh GC (2003) Host range and reproductive traits of Diabrotica speciosa (Germar) and Diabrotica viridula (F.) (Coleoptera: Chrysomelidae), two species of South America pest rootworms, with notes on other species of Diabroticina. Environ Entomol 32:276–285. https://doi.org/10.1603/0046-225X-32.2.276
Walsh GC, Ávila CJ, Cabrera N, Nava DE, Sene PA (2020) Biology and management of pest Diabrotica species in South America. InSects 11:421–439. https://doi.org/10.3390/insects11070421
Wang M, Biere A, Van der Putten WH, Bezemer TM (2014) Sequential effects of root and foliar herbivory on aboveground and belowground induced plant defense responses and insect performance. Oecologia 175:187–198. https://doi.org/10.1007/s00442-014-2885-y
Wetzel WC, Strong DR (2015) Host selection by an insect herbivore with spatially variable density dependence. Oecologia 179 (3):777–784. https://doi.org/10.1007/s00442-015-3378-3
Ye W, Bustos-Segura C, Degen T, Erb M, Turlings TC (2022) Belowground and aboveground herbivory differentially affect the transcriptome in roots and shoots of maize. Plant Direct 6(7):e426. https://doi.org/10.1002/pld3.426
Funding
This research was supported by the National Institute of Science and Technology – Semiochemicals in Agriculture (Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) and Conselho Nacional de Desenvolvimento Científico e Tecnológico grants #2014/50871-0 and #465511/2014-7, respectively). Fundação de Amparo à Pesquisa do Estado de São Paulo, #2014/50871-0, Crisitane Nardi, Conselho Nacional de Desenvolvimento Científico e Tecnológico, #465511/2014-7, Crisitane Nardi
Author information
Authors and Affiliations
Contributions
Methods, checking, and original draft preparation, CN, MFGVP, and FS; revision and editing, CR and JRFO; statistical analyses, FS; formal analyses and revision, MFGVP and JMSB.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflicts of interest.
Additional information
Communicated by Jarmo K. Holopainen.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nardi, C., Rech, C., Oliveira, J.R.F. et al. Preference-performance hypothesis and host oviposition selection of Diabrotica speciosa: aboveground female avoids belowground conspecific larvae in maize. Arthropod-Plant Interactions 17, 629–638 (2023). https://doi.org/10.1007/s11829-023-09972-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-023-09972-x