Abstract
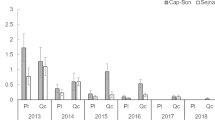
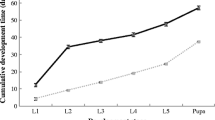
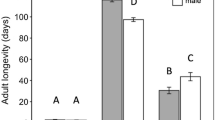
The outbreak species, Orgyia trigotephras caused significant defoliation in northeastern Tunisia in 2005. This work aims to elucidate the population cycle of this pest by testing a wide range of variation of fecundity and population growth. The recorded fecundity was at its highest peak with an average of 153 eggs/egg batch during outbreaks. The action of the complex of egg parasitoid/predator associated with O. trigotephras varies over time. The action of Aprostocetus sp. and Coccidiphila rungsella was at its utmost during the collapse phase of the insect in 2007 and 2014. Only these two species were recorded over our 17-year study. A low proportion of dried eggs versus a high proportion of unfertilized eggs were observed due to the poor quality of foliage consumed by the mother larva during its development. The correlation between fecundity and unfertilized eggs was highly significant. Fecundity change indicators of O. trigotephras are indirect, namely defoliation by high densities of larvae reducing leaf quality for the next generation. The abundance of the host species makes it easily found by parasitoids or vulnerable to parasitism. This strategy is used by O. trigotephras since the action of natural enemies is very low. Yet, the insect was not observed since 2018. It will be important to conduct large research in the region to see whether the insect is still present in the localities or relocate to other ones near the studied site.



Similar content being viewed by others
Data availability
We admit that the results in the paper will be archived in an appropriate public repository.
References
Bella S, Longo S, Sidoti A (2011) Indagini su Teia trigotephras defogliatore del lentisco nella Sicilia sud-orientale. Atti XXIII Congresso Nazionale Italiano di Entomologia Genova, 13–16-giugno: 107.
Ben Jamâa ML, M’nara S, Villemant C, Khaldi A, (2002) Lymantria dispar L. (Lepidoptera, Lymantriidae) en Tunisie: état actuel des connaissances et perspectives de recherche. IOBC-WPRS Bulletin 25:101–108
Berryman AA (1996) What causes population cycles of forest Lepidoptera? Trends Ecol Evol 11:28–32. https://doi.org/10.1016/0169-5347(96)81066-4
Campbell RW, Sloan RJ, Biazak CE (1977) Sources of mortality among late instar gypsy moth larvae in sparse populations. Environ Entomol 6:865–871
Cappuccino N, Lavertu D, Bergeron Y, Regniere R (1998) Spruce budworm impact, abundance and parasitism rate in a patchy landscape. Oecologia 114:236–242
Direction Générale des Forêts (1995) Forêt domaniale De Béni Oulid: Plan d’aménagement 1996–2015. SOGET Maghreb, Tunisia
Duan JJ, Bauer LS, Abell KJ, Lelito JP, Van Driesche R (2013) Establishment and abundance of Tetrastichus planipennisi (Hymenoptera: Eulophidae) in Michigan: Potential for success in classical biocontrol of the invasive emerald ash borer (Coleoptera: Buprestidae). J Eco Entomol 106:1145–1154. https://doi.org/10.1603/EC13047
Elton C (1927) Animal ecology. Sidgwick and Jackson, London
Ezzine O, Ben Jamâa ML, M’nara S, Nouira S (2010) Bioécologie d’Orgyia trigotephras (Boisduval, 1829), (Lepidoptera, Lymantriidae) à Jebel Abderrahmane (Nord Est, Tunisie). IOBC/WPRS Bull 57:123–127
Ezzine O, Branco M, Villemant C, Schmidt S, Nouira S, BenJamâa ML (2015) Host use in Orgyia trigotephras (Erebidae, Lymantriinae) during the outbreak: effects on larval performance and egg mortality. Anna Forest Sci AFS72:561–568. https://doi.org/10.1007/s13595-015-0484-7
Ezzine O, Dhahri S, Hammami S, LaajimiO MS, Ben Jamâa ML (2020) Pupa parasitoids of Casama innotata (Lepidoptera, Erebidae), defoliator of Acacia horrida in Tunisia. IOBC-WPRS Bull 151:83–88
Ezzine O, Dhahri S, Hammami S, Bourouguaoui A, Ben Jamâa ML (2021) Occurrence of a new pest Casama innotata (Walker 1855) (Lepidoptera, Erebidae) on a nonnative host plant in an arid environment. J Arid Environ 188:104450. https://doi.org/10.1016/j.jaridenv.2021.104450
Ezzine O (2016) Interactions insectes/plantes-hôtes: cas d’Orgyia trigotephras Boisduval (1829) (Lepidoptera, Erebidae) en Tunisie. PhD thesis, Faculté des Sciences Mathématiques, Physiques et Naturelles de Tunis.
Hammami S, Ezzine O, Dhahri S, Villemant C, Schmidt S, Ben Jamâa ML (2019) Pupae mortality of Orgyia trigotephras Boisduval 1829 (Erebidae, Lymantriinae) in Tunisia. Redia 102:107–111. https://doi.org/10.19263/REDIA-102.19.16
Harrison S (1997) Persistent, localized outbreaks in the western tussock moth Orgyia vetusta: the roles of resource quality, predation, and poor dispersal. Ecol Entomol 22:158–16. https://doi.org/10.1046/j.1365-2311.1997.00053.x
Hoch G, Zubrik M, Novotny J, Schopf A (2001) The natural enemy complex of the gypsy moth, Lymantria dispar (Lep., Lymantriidae) in different phases of its population dynamics in eastern Austria and Slovakia- a comparative study. J Appl Entomol 125:217–227. https://doi.org/10.1046/j.1439-0418.2001.00540.x
Krugner R (2014) Suitability of non-fertilized eggs of Homalodisca vitripennis for the egg parasitoid Gonatocerus morrilli. BioControl 59:167–174. https://doi.org/10.1007/s10526-014-9562-2
Maron JL, Harrison S (1997) Spatial pattern formation in an insect host-parasitoid system. Science 278:1619–1621. https://doi.org/10.1126/science.278.5343.1619
Maron JL, Harrison S, Greaves M (2001) Origin of an insect outbreak: escape in space or time from natural enemies? Oecologia 126:595–602. https://doi.org/10.1007/S004420000558
May RM, Hassell MP (1988) Population dynamics and biological control. R Soc 318:129–169
Montgomery ME, Wallner WE (1988) The gypsy moth: a westward migrant. In: Berryman AA (ed) Dynamics of forest insect populations: patterns, causes, implications. Plenum Press, New York, pp 353–376
Münster-Swendsen M (1985) A simulation study of primary-, clepto and hyper-parasitism in Epinotia tedella (Lepidoptera: Tortricidae). J Anim Ecol 54:683–695
Myers JH, Cory JS (2013) Population cycles in forest lepidoptera revisited. Annu Rev Ecol Evol Syst 44:565–592. https://doi.org/10.1146/annurev-ecolsys-110512-135858
Rossiter MC (1994) Maternal effects hypothesis of herbivore outbreak. BioScience 44:752–63. https://doi.org/10.2307/1312584
Sampson BJ, Roubos CR, Stringer SJ, Marshall D, Liburd OE (2013) Biology and efficacy of Aprostocetus (Eulophidae: Hymenoptera) as a parasitoid of the blueberry gall midge complex: Dasineura oxycoccana and Prodiplosis vaccinii (Diptera: Cecidomyiidae). J Econ Entomol 106:73–79. https://doi.org/10.1603/ec12404
Tanhuanpää M, Ruohomäki K, Turchin P, Ayres MP, Bylund H, Kaitaniemi P, Tammaru T, Haukioja E (2002) Population cycles of the autumnal moth in Fennoscandia. In: Berryman AA (ed) Population cycles: the case for trophic interactions. Oxford University Press, New York, pp 142–164
Ticehurst M, Fusco RA, Kling RP, Unger J (1978) Observations on parasites of gypsy moth in first cycle infestations in Pennsylvania from 1974–1977. Environ Entomol 7:355–358
Viggiani G (2021) Biological notes on some egg parasitoids of Phaneroptera nana Fieber, 1853 (Orthoptera, Tettigoniidae) with a description of a new species of Aprostocetus Westwood, 1833 (Hymenoptera, Eulophidae) from Italy. Biodivers J 12:289–295. https://doi.org/10.31396/Biodiv.Jour.2021.12.2.289.295
Villemant C, Fraval A (1992) Les ennemis des œufs de Porthetria dispar (L.) (Lép.: Lymantriidae) au Maroc: inventaire et problèmes relatifs à l’évaluation de leur impact. Bulletin De L’institut Scientifique Rabat 16:160–172
Voegele JM (1989) Biological control of Brontispa longissima in Western Samoa: an ecological and economic evaluation. Agric Ecosyst Environ 27:315–329
Wang Y, Zou ZP, Hou YY, Yang XB, Wang S, Dai HJ, Xu YY, Zang LS (2020) Manually-extracted unfertilized eggs of Chinese oak silkworm, Antheraea pernyi, enhance mass production of Trichogramma parasitoids. Entomol Gen 40:397–406. https://doi.org/10.1127/entomologia/2020/1060
Wang J, Chen YM, Yang XB, Lv RE, Desneux N, Zang LS (2021) Parasitism and suitability of Aprostocetus brevipedicellus on Chinese Oak silkworm, Antheraea pernyi, a dominant factitious host. Insects 12:694. https://doi.org/10.3390/insects12080694
Wyckhuys KAG, O’Neil RJ (2006) Population dynamics of Spodoptera frugiperda Smith (Lepidoptera: Noctuidae) and associated arthropod natural enemies in Honduran subsistence maize. Crop Prot 25:1180–1190. https://doi.org/10.1016/j.cropro.2006.03.003
Yang ZQ, Yao YX, Cao LM (2015) Chalcidoidea parasitizing forest defoliators (Hymenoptera). Science Press, Beijing, China
Acknowledgements
Thanks to our forest technicians for their valuable help in the field. We wish to thank Emna DARGHOUTHI (mannouta@msn.com) for her assistance with the language editing.
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ezzine, O., Hammami, S. & Ben Jamâa, M.L. The role of natural enemies in regulating the population of the outbreak species Orgyia trigotephras (Lepidoptera: Erebidae) in North Africa. Arthropod-Plant Interactions 16, 469–475 (2022). https://doi.org/10.1007/s11829-022-09915-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-022-09915-y