Abstract
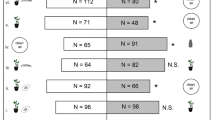
Herbivore-induced plant volatiles (HIPVs) are regarded as an indirect plant defense against herbivores, since they attract natural enemies to infested plants. On the other hand, HIPVs also affect behavioral responses of herbivores. In a previous study, Lytopylus rufipes, a parasitoid of the oriental fruit moth (Grapholita molesta), showed positive responses to pear HIPVs, but the influence of pear HIPVs on G. molesta is still unclear. To better understand these responses, we first tested oviposition preferences of G. molesta to uninfested and infested pear shoots (US and IS, respectively) and uninfested mature leaves (ML) in dual-choice bioassays using a cylindrical tube. Then, preferences to volatile components were also assessed. G. molesta females preferred US-, IS-, and ML-treated areas compared to the control area (no leaves), respectively. Subsequently, G. molesta females preferred IS over US, and US more than ML in bioassays. Furthermore, G. molesta females did not show any significant preference for individual volatile components, but more eggs were laid in an area treated with a synthetic pear HIPV blend, compared to the hexane-treated area (control). These results indicate that G. molesta females do not avoid infested pear shoots nor HIPVs. Moreover, HIPVs may recruit not only natural enemies, but also G. molesta females to the release point of HIPVs. Thus, it is important to examine responses of G. molesta females to HIPVs in the field before employing them for pest management.



Similar content being viewed by others
References
Allen HW, Yetter WP (1949) Bassus diversus, an oriental fruit moth parasite established in the United States. J Econ Entomol 42:540. https://doi.org/10.1093/jee/42.3.540
Allen BW (1962) Parasites of the oriental fruit moth in the eastern United States. USDA Tech Bull No. 1265
Bernasconi ML, Turlings TCJ, Ambrosetti L, Bassetti P, Dorn S (1998) Herbivore-induced emissions of maize volatiles repel the corn leaf aphid, Rhopalosiphum maidis. Entomol Exp Appl 87:133–142. https://doi.org/10.1046/j.1570-7458.1998.00315.x
CABI (2022), current year. Grapholita molesta. In: Invasive Species Compendium. Wallingford, UK: CAB International. www.cabi.org/isc/datasheet/29904. Accessed 5 Aug 2022
Chen LH, Tian K, Wang GR, Xu XL, He KH, Liu W, Wu JX (2019) The general odorant receptor GmolOR9 from Grapholita molesta (Lepidoptera: Tortricidae) is mainly tuned to eight host-plantvolatiles. Insect Sci 27:1233–1243. https://doi.org/10.1111/1744-7917.12725
Collatz J, Dorn S (2013) Tritrophic consequences arising from a host shift between apple and walnut in an oligophagous herbivore. Biol Control 65:330–337. https://doi.org/10.1016/j.biocontrol.2013.03.011
De Lame FM, Hong J, Shearer PW, Brattsten LB (2001) Sex-related differences in the tolerance of Oriental fruit moth (Grapholita molesta) to organophosphate insecticides. Pest Manag Sci 57:827–832. https://doi.org/10.1002/ps.368
De Moraes CM, Mescher MC, Tumlinson JH (2001) Caterpillar-induced nocturnal plant volatiles repel conspecific females. Nature 410:577–580. https://doi.org/10.1038/35069058
Dicke M, Hilker M (2003) Induced plant defences: from molecular biology to evolutionary ecology. Basic Appl Ecol 4:3–14. https://doi.org/10.1078/1439-1791-00129
Dicke M, Van Beek TA, Posthumus MA, Ben Dom N, Van Bokhoven H, De Groot A (1990) Isolation and identification of volatile kairomone that affects acarine predator–prey interactions involvement of host plant in its production. J Chem Ecol 16:381–396. https://doi.org/10.1007/BF01021772
El-Sayed AM, Knight AL, Byers JA, Judd GJR, Suckling DM (2016) Caterpillar-induced plant volatiles attract conspecific adults in nature. Sci Rep 6:37555. https://doi.org/10.1038/srep37555
El-Sayed AM, Knight AL, Basoalto E, Suckling DM (2018) Caterpillar-induced plant volatiles attract conspecific herbivores and a generalist predator. J Appl Entomol 142:495–503. https://doi.org/10.1111/jen.12495
El-Sayed AM, Sporle A, Gemeno C, Jósvai JK, Simmons GS, Suckling DM (2019) Leafroller-induced phenylacetonitrile and acetic acid attract adult Lobesia botranain European vineyards. J Biosci 74:161–165. https://doi.org/10.1515/znc-2018-0163
Feeny P (1976) Plant apparency and chemical defense. In: Wallace JW, Mansell RL (eds) Biochemical interaction between plants and insects. Springer, New York, pp 1–40. https://doi.org/10.1007/978-1-4684-2646-5_1
Giacomuzzi V, Cappellin L, Khomenko I, Biasioli F, Schütz S, Tasin M, Knight AI, Angeli S (2016) Emission of volatile compounds from apple plants infested with Pandemis heparana larvae, antennal response of conspecific adults, and preliminary field trial. J Chem Ecol 42:1265–1280. https://doi.org/10.1007/s10886-016-0794-8
Giacomuzzi V, Matthias J, Basoalto E, Knight AL (2017) Survey of conspecific herbivore-induced volatiles from apple as possible attractants for Pandemis pyrusana (Lepidoptera: Tortricidae). Pest Manag Sci 73:1837–1845. https://doi.org/10.1002/ps.4548
Halitschke R, Stenberg JA, Kessler D, Kessler A, Baldwin T (2008) Shared signals ‘alarm calls’ from plant increase apparency to herbivores and their enemies in nature. Ecol Lett 11:24–34. https://doi.org/10.1111/j.1461-0248.2007.01123.x
Il’ichev AL, Kugimiya S, Williams DG, Takabayashi J (2009) Volatile compounds from young peach shoots attract males of oriental fruit moth in the field. J Plant Interact 4:289–294. https://doi.org/10.1080/17429140903267814
Kanga LHB, Pree DJ, van Lier JL, Walker GM (2003) Management of insecticide resistance in oriental fruit moth (Grapholita molesta; Lepidoptera: Tortricidae) populations from Ontario. Pest Manag Sci 59:921–927. https://doi.org/10.1002/ps.702
Kirk H, Dorn S, Mazzi D (2013) Worldwide population genetic structure of the oriental fruit moth (Grapholita molesta), a globally invasive pest. BMC Ecol 13:12. https://doi.org/10.1186/1472-6785-13-12
Kurokura T, Mimida N, Battey NH, Hytönen T (2013) The regulation of seasonal flowering in the Rosaceae. J Exp Bot 64:4131–4141. https://doi.org/10.1093/jxb/ert233
Landolt PJ (1993) Effects of host plant leaf damage on cabbage looper moth attraction and oviposition. Entomol Exp Appl 67:79–85
Leopold EJ (1986) Selective hydroboration of a 1,3,7-triene: homogeraniol. Org Synth 64:164–164. https://doi.org/10.1002/0471264180.os064.25
Liu CM, Matsuyama S, Kainoh Y (2019) Synergistic effects of volatiles from host-infested plants on host-searching behavior in the parasitoid wasp Lytopylus rufipes (Hymenoptera: Braconidae). J Chem Ecol 45:684–692. https://doi.org/10.1007/s10886-019-01088-y
Lu P, Qiao H (2020) Peach volatile emission and attractiveness of different host plant volatiles blends to Cydia molesta in adjacent peach and pear orchards. Sci Rep 10:13658. https://doi.org/10.1038/s41598-020-70685-9
Mills NJ, Heimpel GE (2018) Could increased understanding of foraging behavior help to predict the success of biological control? Curr Opin Insect Sci 27:26–31. https://doi.org/10.1016/j.cois.2018.02.013
Mumm R, Dicke M (2010) Variation in natural plant products and the attraction of 360bodyguards involved in indirect plant defense. Can J Zool 88:628–667
Najar-Rodriguez A, Orschel B, Dorn S (2013) Season-long volatile emissions from peach and pear trees in situ, overlapping profiles, and olfactory attraction of an oligophagous fruit moth in the laboratory. J Chem Ecol 39:418–429
Navarro-Roldán MA, Avilla J, Bosch D, Valls J, Gemeno C (2017) Comparative effect of three neurotoxic insecticides with different modes of action on adult males and females of three tortricid moth pests. J Econ Entomol 110:1740–1749. https://doi.org/10.1093/jee/tox113
Pastori PL, Arioli CJ, Botton M, Monteiro LB, Stoltman L, Mafra-Neto A (2012) Integrated control of two tortricids (Lepidoptera) pests in apple orchards with sex pheromones and insecticides. Rev Colomb Entomol 38:224–230
Piyasaengthong N, Sato Y, Kinoshita N, Kainoh Y (2016) Oviposition preference for leaf age in the smaller tea tortrix Adoxophyes honmai (Lepidoptera: Tortricidae) as related to performance of neonates. Appl Entomol Zool 51:363–371
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rings RW (1970) Economic aspects of the biology and control of the Oriental Fruit moth, Grapholitha molesta Busck, in the United States. Ohio J Sci 70:58–61
Rothschild GHL, Vickers RA (1991) Biology, ecology and control of the oriental fruit moth. In: van der Geest LPS, Evenhuis HH (eds) Totricid pests: their biology, natural enemies, and control. Elsevier Science Publishers, Amsterdam, pp 389–412
Rusch A, Delbac L, Muneret L, Thiéry D (2015) Organic farming and host density affect parasitism rates of tortricid moths in vineyards. Agric Ecosyst Environ 214:46–53
Schoonhoven LM, van Loon JJA, Dicke M (2005) Insect-plant biology, 2nd edn. Oxford University Press, Oxford
Trimble RM, Pree DJ, Carter NJ (2001) Integrated control of oriental fruit moth (Lepidoptera: Tortricidae) in peach orchards using insecticide and mating disruption. J Econ Entomol 94:476–485. https://doi.org/10.1603/0022-0493-94.2.476
Vallat A, Dorn S (2005) Changes in volatile emissions from apple trees and associated response of adult female codling moths over the fruit-growing season. J Agric Food Chem 53:4083–4090. https://doi.org/10.1021/jf048499u
Yildizhan S, Schulz S (2011) Easy access to (E)-β-ocimene. Synlett 2011:2831–2833. https://doi.org/10.1055/s-0031-12898
Yoneya K, Miki T (2015) Co-evolution of foraging behaviour in herbivores and their natural enemies predicts multifunctionality of herbivore-induced plant volatiles. Funct Ecol 29:451–461
Acknowledgements
We appreciate the kind help of the following researchers: Mr. Masao Kaneko (Nagano Fruit Tree Experiment Station) for providing the moth colony, Prof. Sumiko Sugaya (Faculty of Life and Environmental Sciences, University of Tsukuba) for providing the pear trees and advice on their care, and Ms. Kai Han and Ms. Yu Hsin Yao for preparing materials and insect rearing for bioassays.
Author information
Authors and Affiliations
Contributions
CML, SM, and YK conceived the study. SM carried out chemical analyses and synthesis. CML designed the bioassay and carried out chemical and statistical analyses. CML and SP carried out bioassays. CML drafted the manuscript with assistance from SM and YK, and WA provided technical suggestions. All authors edited the manuscript and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors did not receive support from any organization for the submitted work and have no conflicts of interest to declare. All coauthors have seen and agreed with the contents of the manuscript and there was no financial interest to report.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, CM., Phukhahad, S., Auamcharoen, W. et al. Oviposition preferences of the oriental fruit moth Grapholita molesta (Lepidoptera: Tortricidae) to pear HIPVs. Arthropod-Plant Interactions 16, 517–523 (2022). https://doi.org/10.1007/s11829-022-09914-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-022-09914-z