Abstract
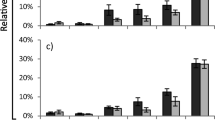
Several secondary metabolites are associated with plant resistance against herbivores. Cassava genotypes present a wide variety of metabolites with insecticidal potential, but little is known about the identity of these compounds. The present work was conducted for the identification of insecticidal molecules present in cassava genotypes. To this purpose, we firstly evaluated the development of the generalist herbivore Spodoptera frugiperda (J.E. Smith, 1797) (Lepidoptera: Noctuidae) fed with leaves from six cassava genotypes (IAC-14, IAC-90, IAC-12, IAC-Caapora, Baianinha and MEcu 72). Following bioassays, we measured the levels of two anti-herbivory related groups of compounds (phenolics and steroidal saponins) in all genotypes using colorimetric methods. The metabolic fingerprinting (GC–MS) of three contrasting cassava genotypes selected by their biological interferences over S. frugiperda was performed. The most unsuitable nutritional indices were observed for larvae fed with MEcu 72. Lower reproductive indices were observed for adults in which larval stages were fed with Baianinha or MEcu 72. Steroidal saponin content was similar in all genotypes, but phenolic content was 25% higher in MEcu 72. GC–MS metabolite fingerprinting of MEcu 72, Baianinha and IAC-Caapora resulted in the annotation of 53 metabolites in which 20 presented different relative abundance among the evaluated genotypes. Molecules such as myo-inositol-2-monophosphate, p-coumaric acid, chlorogenic acid, p-hydroxybenzoic acid, octadecadienoic acid and n-hexacosane were more abundant in MEcu 72 or Baianinha than in IAC-Caapora. The possible roles of these molecules in the development of S. frugiperda are discussed.



Similar content being viewed by others
References
Adel MM, Sehnal F, Jurzysta M (2000) Effects of alfalfa saponins on the moth Spodoptera littoralis. J Chem Ecol 26(1065–1078):2. https://doi.org/10.1023/A:1005445217004
Ainsworth EA, Gillespie KM (2007) Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin–Ciocalteu reagent. Nat Protoc 2:875–877. https://doi.org/10.1038/nprot.2007.102
Ali Q, Ashraf M, Anwar F, Al-Qurainy F (2012) Trehalose-induced changes in seed oil composition and antioxidant potential of maize grown under drought stress. J Am Oil Chem Soc 89:1485–1493. https://doi.org/10.1007/s11746-012-2032-z
Awmack CS, Leather SR (2002) Host plant quality and fecundity in herbivorous insects. Annu Rev Entomol 47:817–844. https://doi.org/10.1146/annurev.ento.47.091201.145300
Baccou JC, Lambert F, Sauvaire Y (1977) Spectrometric method for the determination of total steroidal sapogenin. Analyst 102:458–465. https://doi.org/10.1039/an9770200458
Barilli DR, Wengrat APGS, Guimarães ATB, Pietrowski V, Ringenberg R, Garcia MS (2019) Resistance of cassava genotypes to Bemisia tuberculata. Arthropod-Plant Interact 13:663–669. https://doi.org/10.1007/s11829-019-09694-z
Bellotti AC, Arias B (2001) Host plant resistance to whiteflies with emphasis on cassava as a case study. Crop Prot 20:813–823. https://doi.org/10.1016/S0261-2194(01)00113-2
Birch LC (1948) The intrinsic rate of natural increase of an insect population. J Anim Ecol 17:15–26. https://doi.org/10.2307/1605
Blanco CA, Chiaravalle W, Dalla-Rizza M, Farias JR, García-Degano MF, Gastaminza G, Mota-Sánchez D, Murúa MG, Omoto C, Pieralisi BK, Rodríguez J, Rodríguez-Maciel JC, Terán-Santofimio H, Terán-Vargas AP, Valencia SJ, Willink E (2016) Current situation of pests targeted by Bt crops in Latin America. Curr Opin Insect Sci 15:131–138. https://doi.org/10.1016/j.cois.2016.04.012
Bourgaud F, Gravot A, Milesi S, Gontier E (2001) Production of plant secondary metabolites: a historical perspective. Plant Sci 161:839–851. https://doi.org/10.1016/S0168-9452(01)00490-3
Calatayud PA, Rahbé Y, Delobel B, Khuong-Huu F, Tertuliano M, Le Rü B (1994) Influence of secondary compounds in the phloem sap of cassava on expression of antibiosis towards the mealybug Phenacoccus manihoti. Entomol Exp Appl 72:47–57. https://doi.org/10.1007/BF02382414
Capitani D, Sobolev AP, Tomassini A, Sciubba F, De Salvador FR, Mannina L, Delfini M (2013) Peach fruit: metabolic comparative analysis of two varieties with different resistances to insect attacks by NMR spectroscopy. J Agric Food Chem 61:1718–1726. https://doi.org/10.1021/jf303248z
Carabalí A, Bellotti AC, Montoya-Lerma J, Fregene M (2010) Resistance to the whitefly, Aleurotrachelus socialis, in wild populations of cassava, Manihot tristis. J Insect Sci 10:1–10. https://doi.org/10.1673/031.010.14130
Carnevale Neto F, Pilon AC, Selegato DM, Freire RT, Gu H, Raftery D, Lopes NP, Castro-Gamboa I (2016) Dereplication of natural products using GC-TOF mass spectrometry: improved metabolite identification by spectral deconvolution ratio analysis. Front Mol Biosci 3:59. https://doi.org/10.3389/fmolb.2016.00059
Casida JE (1980) Pyrethrum flowers and pyrethroid insecticides. Environ Health Perspect 34:189–202. https://doi.org/10.1289/ehp.8034189
Chen F, Liu CJ, Tschaplinski TJ, Zhao N (2009) Genomics of secondary metabolism in Populus: interactions with biotic and abiotic environments. Crit Rev Plant Sci 28:375–392. https://doi.org/10.1080/07352680903241279
Chong J, Yamamoto M, Xia J (2019) MetaboAnalystR 2.0: from raw spectra to biological insights. Metabolites 9:57. https://doi.org/10.3390/metabo9030057
Chrzanowski G, Leszczynski B, Czerniewicz P, Sytykiewicz H, Matok H, Krzyzanowski R (2012) Effect of phenolic acids from black currant, sour cherry and walnut on grain aphid (Sitobion avenae F.) development. Crop Prot 35:71–77. https://doi.org/10.1016/j.cropro.2012.01.005
Cuadros-Inostroza A, Caldana C, Redestig H, Kusano M, Lisec J, Peña-Cortés H, Willmitzer L, Hannah MA (2009) TargetSearch—a bioconductor package for the efficient preprocessing of GC-MS metabolite profiling data. BMC Bioinform 10:428. https://doi.org/10.1186/1471-2105-10-428
De Geyter E, Gellen D, Smagghe G (2007) First result on the insecticidal action of saponins. Commun Appl Biol Sci 72:645–648
Fatoretto JC, Michel AP, Silva Filho MC, Silva N (2017) Adaptive potential of fall armyworm (Lepidoptera: Noctuidae) limits Bt trait durability in Brazil. J Integr Pest Manag 8:1–10. https://doi.org/10.1093/jipm/pmx011
Felton GW, Donato K, Del Vecchio RJ, Duffey SS (1989) Activation of plant foliar oxidases by insect feeding reduces nutritive quality of foliage for noctuid herbivores. J Chem Ecol 15:2667–2694. https://doi.org/10.1007/BF01014725
Felton GW, Donato KK, Broadway RM, Duffey SS (1991) Impact of oxidized plant phenolics on the nutritional quality of dietary protein to a noctuid herbivore, Spodoptera exigua. J Insect Physiol 38:277–285. https://doi.org/10.1016/0022-1910(92)90128-Z
Ferreira D (2011) Sisvar: a computer statistical analysis system. Ciênc Agrotec 35:1039–1042. https://doi.org/10.1590/S1413-70542011000600001
Fiehn O (2002) Metabolomics—the link between genotypes and phenotypes. Plant Mol Biol 48:155–171. https://doi.org/10.1023/A:1013713905833
Gazola D, Zucareli C, Ringenberg R, Oliveira MCN, Graça JP, Nunes EO, Hoffmann-Campo CB (2018) Secondary metabolite contents in different parts of cassava plants infested by Phenacoccus manihoti Matile-Ferrero (Hemiptera: Pseudococcidae). Arthropod-Plant Interact 13:359–366. https://doi.org/10.1007/s11829-018-9649-2
Gonzalez-Coloma A, Reina M, Diaz CE, Fraga BM (2010) Natural product-based biopesticides for insect control. In: Liu H, Mander L (eds) Comprehensive natural products II. Elsevier, Oxford, pp 237–268
Grimball B, Veillon L, Calhoun T, Fronczek FR, Arceneaux E, Laine RA (2017) Cyclohexylamine inexplicably induces antennae loss in formosan subterranean termites (Coptotermes formosanus Shiraki): cyclohexylamine hydrogen phosphate salts are novel termiticides. Pest Manag Sci 73:2039–2047. https://doi.org/10.1002/ps.4610
Harborne JB (1999) Classes and functions of secondary products from plants. In: Walton NJ, Brown DE (eds) Chemicals from plants. Imperial College Press and World Scientific Publishing, London, pp 1–25. https://doi.org/10.1142/9789812817273_0001
Honek A (1993) Intraspecific variation in body size and fecundity in insects: a general relationship. Oikos 66:483–492. https://doi.org/10.2307/3544943
Ibanez S, Gallet C, Despres L (2012) Plant insecticidal toxins in ecological networks. Toxins 4:228–243. https://doi.org/10.3390/toxins4040228
Jansen JJ, Allwood W, Marsden-Edwards E, van der Putten WH, Goodacre R, van Dam NM (2009) Metabolomic analysis of the interaction between plants and herbivores. Metabolomics 5:150–161. https://doi.org/10.1007/s11306-008-0124-4
Kopka J, Schauer N, Krueger S, Birkemeyer C, Usadel B, Bergmüller E, Dörmann P, Weckwerth W, Gibon Y, Stitt M, Willmitzer L, Fernie AR, Steinhauser D (2005) GMD@CSB.DB: the Golm Metabolome Database. Bioinformatics 21:1635–1638. https://doi.org/10.1093/bioinformatics/bti236
Krebs CJ (1994) Ecology: the experimental analysis of distribution and abundance. Harper & Row, New York
Lattanzio V (2013) Phenolic compounds: introduction. In: Lattanzio V (ed) Natural products. Springer Press, Berlin, pp 1543–1580. https://doi.org/10.1007/978-3-642-22144-6_57
Leiss KA, Choi YH, Abdel-Farid IB, Verpoorte R, Klinkhamer PG (2009a) NMR metabolomics of thrips (Frankliniella occidentalis) resistance in Senecio hybrids. J Chem Ecol 35:219–229. https://doi.org/10.1007/s10886-008-9586-0
Leiss KA, Maltese F, Choi YH, Verpoorte R, Klinkhamer PG (2009b) Identification of chlorogenic acid as a resistance factor for thrips in chrysanthemum. Plant Physiol 150:1567–1575. https://doi.org/10.1104/pp.109.138131
Lunn JE, Delorge I, Figueroa CM, Dijck PV, Stitt M (2014) Trehalose metabolism in plants. Plant J 79:544–567. https://doi.org/10.1111/tpj.12509
Maag D, Erb M, Glauser G (2015) Metabolomics in plant–herbivore interactions: challenges and applications. Entomol Exp Appl 157:18–29. https://doi.org/10.1111/eea.12336
Macías FA, Galindo JLG, Galindo JCG (2007) Evolution and current status of ecological phytochemistry. Phytochemistry 68:2917–2936. https://doi.org/10.1016/j.phytochem.2007.10.010
Maia AHN, Luiz AJB, Campanhola C (2000) Statistical inference on associated fertility life table parameters using Jackknife technique: computational aspects. J Econ Entomol 93:511–518. https://doi.org/10.1603/0022-0493-93.2.511
Maluf WR, Maciel GM, Gomes LAA, Cardoso MG, Gonçalves LD, Silva EC, Knapp M (2010) Broad-spectrum arthropod resistance in hybrids between high- and low-acylsugar tomate lines. Crop Sci 50:439–450. https://doi.org/10.2135/cropsci2009.01.0045
Mithöfer A, Boland W (2012) Plant defense against herbivores: chemical aspects. Annu Rev Plant Biol 63:431–450. https://doi.org/10.1146/annurev-arplant-042110-103854
Mouden S, Klinkhamer PG, Choi YH, Leiss KA (2017) Towards eco-friendly crop protection: natural deep eutectic solvents and defensive secondary metabolites. Phytochem Rev 16:935–951. https://doi.org/10.1007/s11101-017-9502-8
Omongo CA, Kawuki R, Bellotti AC, Alicai T, Baguma Y, Maruthim MN, Bua A, Colvin J (2012) African cassava whitefly, Bemisia tabaci, resistance in African and South American cassava genotypes. J Integr Agric 11:327–336. https://doi.org/10.1016/S2095-3119(12)60017-3
Park Y, Albright KJ, Liu W, Storkson JM, Cook ME, Pariza MW (1997) Effect of conjugated linoleic acid on body composition in mice. Lipids 32:853–858. https://doi.org/10.1007/s11745-997-0109-x
Parra JRP (1991) Consumo e utilização de alimento por insetos. In: Panizzi AR, Parra JRP (eds) Ecologia nutricional de insetos e suas implicações no manejo de pragas. Manole, São Paulo, pp 9–65
Parra JRP (2001) Técnicas de criação de insetos para programas de Controle Biológico. FEALQ, Piracicaba
Paulillo LCMS, Lopes AR, Cristofoletti PT, Parra JRP, Terra WR, Silva Filho MC (2000) Changes in midgut endopeptidase activity of Spodoptera frugiperda (Lepidoptera: Noctuidae) are responsible for adaptation to soybean proteinase inhibitors. J Econ Entomol 93:892–896. https://doi.org/10.1603/0022-0493-93.3.892
Pilon AC, Carnevale Neto F, Freire RT, Cardoso P, Carneiro RL, Bolzani VS, Castro-Gamboa I (2016) Partial least squares model and design of experiments toward the analysis of the metabolome of Jatropha gossypifolia leaves: extraction and chromatographic fingerprint optimization. J Sep Sci 39:1023–1030. https://doi.org/10.1002/jssc.201500892
Ponsankar A, Vasantha-Srinivasan P, Senthil-Nathan S, Thanigaivel A, SamEdwin E, Selin-Rani S, Kalaivani K, Hunter WB, Alessandro RT, Abdel-Megeed A, Paik C, Duraipandiyan V, Al-Dhabig NA (2016) Target and non-target toxicity of botanical insecticide derived from Couroupita guianensis L. flower against generalist herbivore, Spodoptera litura Fab. and an earthworm, Eisenia foetida Savigny. Ecotoxicol Environ Saf 133:260–270. https://doi.org/10.1016/j.ecoenv.2016.06.043
Rattan RS (2010) Mechanism of action of insecticidal secondary metabolites of plant origin. Crop Prot 29:913–920. https://doi.org/10.1016/j.cropro.2010.05.008
Rheinheimer AR (2013) Resistência de variedades de mandioca à cochonilha Phenacoccus manihoti (Matile-Ferrero) e sua influência sobre o parasitoide Anagyrus lopezi (De Santis). Thesis, Universidade Estadual do Oeste do Paraná
Santos MAI, Corrêa AD, Alves APC, Simão AA, Alves DS, Oliveira RL, Saczk AA, Carvalho GA (2013) Extrato metanólico de folhas de mandioca como alternativa ao controle da lagarta-do-cartucho e de formigas cortadeiras. Semin Ciênc Agrár 34:3501–3512. https://doi.org/10.5433/1679-0359.2013v34n6Suppl1p3501
Scriber JM, Slansky F Jr (1981) The nutritional ecology of immature insects. Annu Rev Entomol 26:183–211. https://doi.org/10.1146/annurev.en.26.010181.001151
Silva MC, Ribeiro AF, Terra WR, Ferreira C (2009) Sequencing of Spodoptera frugiperda midgut trehalases and demonstration of secretion of soluble trehalase by midgut columnar cells. Insect Mol Biol 18:769–784. https://doi.org/10.1111/j.1365-2583.2009.00920.x
Silva DM, Bueno AF, Andrade K, Stecca CS, Neves PMOJ, Oliveira MCN (2017) Biology and nutrition of Spodoptera frugiperda (Lepidoptera: Noctuidae) fed on different food sources. Sci Agric 74:18–31. https://doi.org/10.1590/1678-992x-2015-0160
Silva-Brandão KL, Horikoshi RJ, Bernardi D, Omoto C, Figueira A, Brandão MM (2017) Transcript expression plasticity as a response to alternative larval host plants in the speciation process of corn and rice strains of Spodoptera frugiperda. BMC Genomics 18:792. https://doi.org/10.1186/s12864-017-4170-z
Silvera Neto S, Nakano O, Barbin D, Villa Nova NA (1976) Manual de ecologia dos insetos. Agronômica Ceres, São Paulo
Sumner LW, Amberg A, Barrett D et al (2007) Proposed minimum reporting standards for chemical analysis: Chemical Analysis Working Group (CAWG) Metabolomics Standards Initiative (MSI). Metabolomics 3:211–221. https://doi.org/10.1007/s11306-007-0082-2
Torp M, Lehrman A, Stenberg JA, Julkunen-Tiitto R, Björkman C (2013) Performance of an herbivorous leaf beetle (Phratora vulgatissima) on Salix F2 hybrids: the importance of phenolics. J Chem Ecol 39:516–524. https://doi.org/10.1007/s10886-013-0266-3
Undas AK, Weihrauch F, Lutz A, van Tol R, Delatte T, Verstappen F, Bouwmeester H (2018) The use of metabolomics to elucidate resistance markers against Damson-hop aphid. J Chem Ecol 44:711–726. https://doi.org/10.1007/s10886-018-0980-y
Waldbauer GP (1968) Consumption and utilization of food. In: Beament JWL, Treherne JE, Wigglesworth VB (eds) Advances in insect physiology. Academic Press, New York, pp 229–288
Wobeto C, Corrêa AD, Abreu CMP, Santos CD, Pereira HV (2007) Antinutrients in the cassava (Manihot esculenta Crantz) leaf powder at three ages of the plant. Ciênc Tecnol Aliment 27:108–112. https://doi.org/10.1590/S0101-20612007000100019
Wang L, Qu L, Hu J, Zhang L, Tang F, Lu M (2017) Metabolomics reveals constitutive metabolites that contribute resistance to fall webworm (Hyphantria cunea) in Populus deltoides. Environ Exp Bot 136:31–40. https://doi.org/10.1016/j.envexpbot.2017.01.002
Yamamoto I (1999) Nicotine to nicotinoids: 1962 to 1997. In: Yamamoto I, Casida JE (eds) Nicotinoid insecticides and the nicotinic acetylcholine receptor. Springer, Tokyo, pp 3–27
Yurawecz MP, Hood JK, Mossoba MM, Roach JAG, Ku Y (1995) Furan fatty acids determined as oxidation products of conjugated octadecadienoic acid. Lipids 30:595–598. https://doi.org/10.1007/BF02536995
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001.
Author information
Authors and Affiliations
Contributions
DRB performed all bioassays and participated in all data collection. IGFB and DRB analyzed GC–MS data. JLBJ and DRB performed GC–MS analysis. VSB supervised and secured funds for GC–MS analysis. ALBJ, DRB and GDR conceived and designed the research. DRB and GDR interpreted data and wrote the paper. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The research was registered in the National System for the Management of Genetic Heritage and Associated Traditional Knowledge (SisGen) under protocol ACF6CB7.
Additional information
Handling Editor: Anna-Karin Borg-Karlson.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Barilli, D.R., Budzinski, I.G.F., Bronzel Junior, J.L. et al. GC–MS analyses reveal chemical differences in the leaves of Manihot esculenta Crantz genotypes with different anti-herbivore effects. Arthropod-Plant Interactions 15, 387–398 (2021). https://doi.org/10.1007/s11829-021-09822-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-021-09822-8