Abstract
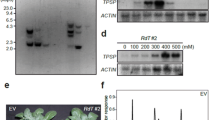
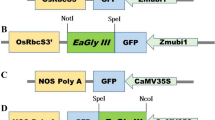
Transgenic potato (Solanum tuberosum L. cv. Desiree) plants expressing components of a novel cyanobacterial photorespiratory glycolate catabolism pathway were developed. Transgenic plant expressing glcD1 (glycolate dehydrogenase I) gene was referred to as synGDH and transgenic plants expressing gcl (glyoxylate carboligase) and tsr (tartronic semialdehyde reductase) genes simultaneously were designated as synGT. Both synGDH and synGT plants showed stable gene transformation, integration and expression. Enhanced glyoxylate contents in synGDH plants were detected as compared to synGT and non-transgenic (NT) plants. Phenotypic evaluation revealed that synGDH plants accumulated 11 % higher dry weight, while, tuber weight was 38 and 16 % higher than NT and synGT, respectively. Upon challenging the plants in high temperature and high light conditions synGDH plants maintained higher Fv/Fm and showed less bleaching of chlorophyll as compared to synGT and NT plants. These results indicate that genetic transformation of complete pathway in one plant holds promising outcomes in terms of biomass accumulation to meet future needs for food and energy.




Similar content being viewed by others
References
Ahmad R, Kim MD, Back KH, Kim HS, Lee HS, Kwon SY, Murata N, Chung WI, Kwak SS (2008) Stress-induced expression of choline oxidase in potato plant chloroplasts confers enhanced tolerance to oxidative, salt, and drought stresses. Plant Cell Rep 27:687–698. doi:10.1007/s00299-007-0479-4
Anderson LE (1971) Chloroplast and cytoplasmic enzymes. II. Pea leaf triose phosphate isomerase. Biochim Biophys Acta 235:237–244
Bauwe H, Hagemann M, Fernie AR (2010) Photorespiration: players, partners and origin. Trends Plant Sci 15:330–336. doi:10.1016/j.tplants.2010.03.006
Bouallagui H, Touhami Y, Hanafi N, Ghariani A, Hamdi M (2013) Performances comparison between three technologies for continuous ethanol production from molasses. Biomass Bioenergy 48:25–32. doi:10.1016/j.biombioe.2012.10.018
Bowes G, Ogren WL, Hageman RH (1971) Phosphoglycolate production catalyzed by ribulosediphosphate carboxylase. Biochem Biophys Res Commun 45:716–722. doi:10.1016/0006-291X(71)90475-X
Brown RH, Hattersley PW (1989) Leaf anatomy of C3-C4 species as related to evolution of C4 photosynthesis. Plant Physiol 91:1543–1550. doi:10.1104/91.4.1543
Douce R, Neuburger M (1999) Biochemical dissection of photorespiration. Curr Opin Plant Biol 2:214–222. doi:10.1016/S1369-5266(99)80038-7
Eisenhut M, Ruth W, Maya H, Bauwe H, Kaplan A, Hagemann M (2008) The photorespiratory glycolate metabolism is essential for cyanobacteria and might have been conveyed endosymbiontically to plants. Proc Natl Acad Sci 105:17199–17204. doi:10.1073/pnas.0807043105
Gonzalez-Moro B, Lacuesta M, Becerril JM, Gonza´lelz-Murua C, Mun˜oz-Rueda A (1997) Glycolate accumulation causes a decrease of photosynthesisby inhibiting RUBISCO activity in maize. J Plant Physiol 150:388–394. doi:10.1016/S0176-1617(97)80087-9
Kebeish R, Niessen M, Thiruveedhi K, Bari R, Hirsch HJ, Rosenkranz R, Staebler N, Schoenfeld B, Kreuzaler F, Peterhaensel C (2007) Chloroplastic photorespiratory bypass increases photosynthesis and biomass production in Arabidopsis thaliana. Nat Biotech 25:593–599. doi:10.1038/nbt1299
Kim SH, Hamada T (2005) Rapid and reliable method of extracting DNA and RNA from sweetpotato, Ipomoea batatas (L). Lam. Biotechnol Lett 23:1841–1845. doi:10.1007/s10529-005-3891-2
Kozaki A, Takeba G (1996) Photorespiration protects C3 plants from photooxidation. Nature 384:557–560. doi:10.1038/384557a0
Oliver DJ (1978) Effect of glyoxylate on the sensitivity of net photosynthesis tooxygen (the Warburg effect) in tobacco. Plant Physiol 62:938–940. doi:10.1104/62.6.938
Oliver DJ (1980) The effect of glyoxylate on photosynthesis and photorespiration by isolated soybean mesophyll cells. Plant Physiol 65:888–892. doi:10.1104/91.4.1543
Park YI, Choi SB, Lie JR (2009) Transgenic plants with cyanobacterial genes. Plant Biotech Rep 3:267–275. doi:10.1007/s11816-009-0100-0
Peterhansel C, Maurino VG (2010) Photorespiration redesigned. Plant Physiol 155:49–55. doi:10.1104/pp.110.165019
Rojano-Delgado AM, Ruiz-Jiménez J, de Castro MD, De-Prado R (2010) Determination of glyphosate and its metabolites in plant material by reversed-polarity CE with indirect absorptiometric detection. Electrophoresis 8:1423–1430. doi:10.1002/elps.200900583
Sharkey TD (2001) Photorespiration. Encycl Life Sci. doi:10.1038/npg.els.0001292
Wendler C, Putzer A, Wild A (1992) Effect of glufosinate (phosphinothricin) and inhibitors of photorespiration on photosynthesis and ribulose-1,5-bisphosphate carboxylase activity. J Plant Physiol 139:666–671
Zelitch I, Schultes NP, Peterson RB, Brown P, Brutnell TP (2009) High glycolate oxidase activity is required for survival of maize in normal air. Plant Physiol 149:195–204. doi:10.1104/pp.108.128439
Zhu XG, Long SP, Ort DR (2010) Improving photosynthetic efficiency for greater yield. Annu Rev Plant Biol 61:235–261. doi:10.1146/annurev-arplant-042809-112206
Acknowledgments
This work was supported by Grants from the Technology Development Program for Agriculture and Forestry, Ministry of Agriculture, Food and Rural Affairs, Republic of Korea, and the KRIBB Research Initiative Program.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ahmad, R., Bilal, M., Jeon, JH. et al. Improvement of biomass accumulation of potato plants by transformation of cyanobacterial photorespiratory glycolate catabolism pathway genes. Plant Biotechnol Rep 10, 269–276 (2016). https://doi.org/10.1007/s11816-016-0403-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11816-016-0403-x